Abstract
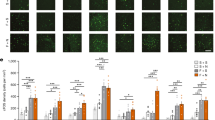
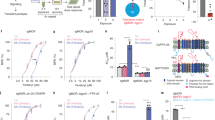
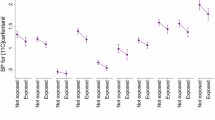
Fentanyl and its analogues are the most commonly used synthetic opioid analgesics in clinical practice, but their abuse is a significant concern. Drug-paired environmental cues often trigger memory retrieval, leading to relapse, complicating treatment and overdose prevention. In this study we investigated μ-opioid receptor-related molecular mechanisms underlying the retrieval of fentanyl contextual addiction memory in mice. A conditional place preference (CPP) model was established in mice by citrate injections of fentanyl (0.1 mg/kg) for 4 days. By performing whole-brain screening using c-Fos immunofluorescence staining, we found that the paraventricular thalamus (PVT) was dramatically activated. We conducted Western blotting, co-immunoprecipitation and proteomics to evaluate the proteins interacting with μ-opioid receptors on the membrane, and found marked externalization of μ-opioid receptors on the membrane in PVT neurons. We revealed that μ-opioid receptors trafficking in PVT was regulated by the extent of binding of Ap2a1 to the membrane μ-opioid receptors. By conditional knockdown and chemogenetic manipulation, we demonstrated the contribution of μ-opioid receptors to the retrieval of fentanyl contextual memory via modulating the neuronal activity in PVT. In conclusion, this study suggests that Ap2a1-mediated trafficking of μ-opioid receptors underlies the retrieval of fentanyl contextual addiction memory through regulating the neuronal activity in PVT.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Rollman JE, Heyward J, Olson L, Lurie P, Sharfstein J, Alexander GC. Assessment of the FDA risk evaluation and mitigation strategy for transmucosal immediate-release fentanyl products. JAMA. 2019;321:676–85.
Muijsers RB, Wagstaff AJ. Transdermal fentanyl: an updated review of its pharmacological properties and therapeutic efficacy in chronic cancer pain control. Drugs. 2001;61:2289–307.
Paech MJ, Lim CB, Banks SL, Rucklidge MW, Doherty DA. A new formulation of nasal fentanyl spray for postoperative analgesia: a pilot study. Anaesthesia. 2003;58:740–4.
Friedman J, Shover CL. Charting the fourth wave: Geographic, temporal, race/ethnicity and demographic trends in polysubstance fentanyl overdose deaths in the United States, 2010-2021. Addiction. 2023;118:2477–85.
Samet JH, El-Bassel N, Winhusen TJ, Jackson RD, Oga EA, Chandler RK, et al. Community-based cluster-randomized trial to reduce opioid overdose deaths. N Engl J Med. 2024;391:989–1001.
Temple C, Hendrickson RG. Increasing exposure of young children to illicit fentanyl in the United States. N Engl J Med. 2024;390:956–7.
Kuehn BM. Fentanyl drives startling increases in adolescent overdose deaths. JAMA. 2023;329:280–1.
Bogdan R, Hatoum AS, Johnson EC, Agrawal A. The genetically informed neurobiology of addiction (GINA) model. Nat Rev Neurosci. 2023;24:40–57.
Bijoch, Klos J, Pawłowska M, Wiśniewska J, Legutko D, Szachowicz U, et al. Whole-brain tracking of cocaine and sugar rewards processing. Transl Psychiatry. 2023;13:20.
Crombag HS, Bossert JM, Koya E, Shaham Y. Review. Context-induced relapse to drug seeking: a review. Philos Trans R Soc Lond B Biol Sci. 2008;363:3233–43.
Zhuang Y, Wang Y, He B, He X, Zhou XE, Guo S, et al. Molecular recognition of morphine and fentanyl by the human μ-opioid receptor. Cell. 2022;185:4361–75.e19.
Plens GM, Droghi MT, Alcala GC, Pereira SM, Wawrzeniak IC, Victorino JA, et al. Expiratory muscle activity counteracts positive end-expiratory pressure and is associated with fentanyl dose in patients with acute respiratory distress syndrome. Am J Respir Crit Care Med. 2024;209:563–72.
Vo QN, Mahinthichaichan P, Shen J, Ellis CR. How μ-opioid receptor recognizes fentanyl. Nat Commun. 2021;12:984.
Darcq E, Kieffer BL. Opioid receptors: drivers to addiction? Nat Rev Neurosci. 2018;19:499–514.
Zhang Y, Xiong W, Lin X, Ma X, Yu LC. Receptor trafficking induced by mu-opioid-receptor phosphorylation. Neurosci Biobehav Rev. 2009;33:1192–7.
Hou G, Jiang S, Chen G, Deng X, Li F, Xu H, et al. Opioid Receptors modulate firing and synaptic transmission in the paraventricular nucleus of the thalamus. J Neurosci. 2023;43:2682–95.
George SR, Zastawny RL, Briones-Urbina R, Cheng R, Nguyen T, Heiber M, et al. Distinct distributions of mu, delta and kappa opioid receptor mRNA in rat brain. Biochem Biophys Res Commun. 1994;205:1438–44.
Zhu Y, Nachtrab G, Keyes PC, Allen WE, Luo L, Chen X. Dynamic salience processing in paraventricular thalamus gates associative learning. Science. 2018;362:423–9.
Zhou K, Zhu Y. The paraventricular thalamic nucleus: A key hub of neural circuits underlying drug addiction. Pharmacol Res. 2019;142:70–6.
Giannotti G, Gong S, Fayette N, Heinsbroek JA, Orfila JE, Herson PS, et al. Extinction blunts paraventricular thalamic contributions to heroin relapse. Cell Rep. 2021;36:109605.
Keyes PC, Adams EL, Chen Z, Bi L, Nachtrab G, Wang VJ, et al. Orchestrating opiate-associated memories in thalamic circuits. Neuron. 2020;107:1113–23.e4.
Du K, Wang Z, Zhang H, Zhang Y, Su H, Wei Z, et al. Levo-tetrahydropalmatine attenuates the acquisition of fentanyl-induced conditioned place preference and the changes in ERK and CREB phosphorylation expression in mice. Neurosci Lett. 2021;756:135984.
Barattini AE, Montanari C, Edwards KN, Edwards S, Gilpin NW, Pahng AR. Chronic inflammatory pain promotes place preference for fentanyl in male rats but does not change fentanyl self-administration in male and female rats. Neuropharmacology. 2023;231:109512.
Williams JT, Ingram SL, Henderson G, Chavkin C, von Zastrow M, Schulz S, et al. Regulation of μ-opioid receptors: desensitization, phosphorylation, internalization, and tolerance. Pharmacol Rev. 2013;65:223–54.
Corder G, Tawfik VL, Wang D, Sypek EI, Low SA, Dickinson JR, et al. Loss of μ opioid receptor signaling in nociceptors, but not microglia, abrogates morphine tolerance without disrupting analgesia. Nat Med. 2017;23:164–73.
Dam DHM, Jelsma SA, Yu JM, Liu H, Kong B, Paller AS. Flotillin and AP2A1/2 promote IGF-1 receptor association with clathrin and internalization in primary human keratinocytes. J Invest Dermatol. 2020;140:1743–52.e4.
White JR, Thompson DT, Koch KE, Kiriazov BS, Beck AC, van der Heide DM, et al. AP-2α-mediated activation of E2F and EZH2 drives melanoma metastasis. Cancer Res. 2021;81:4455–70.
Ritter B, Murphy S, Dokainish H, Girard M, Gudheti MV, Kozlov G, et al. NECAP 1 regulates AP-2 interactions to control vesicle size, number, and cargo during clathrin-mediated endocytosis. PLoS Biol. 2013;11:e1001670.
Chen Z, Tang Y, Tao H, Li C, Zhang X, Liu Y. Dynorphin activation of kappa opioid receptor reduces neuronal excitability in the paraventricular nucleus of mouse thalamus. Neuropharmacology. 2015;97:259–69.
Chaudun F, Python L, Liu Y, Hiver A, Cand J, Kieffer BL, et al. Distinct µ-opioid ensembles trigger positive and negative fentanyl reinforcement. Nature. 2024;630:141–8.
Dang VC, Christie MJ. Mechanisms of rapid opioid receptor desensitization, resensitization and tolerance in brain neurons. Br J Pharmacol. 2012;165:1704–16.
Wu Y, Perez-Rosello T, Awatramani R, Surmeier DJ. Presynaptic Mu Opioid receptors suppress the functional connectivity of ventral tegmental area dopaminergic neurons with aversion-related brain regions. J Neurosci. 2025;45:e1194242025.
Paniccia JE, Vollmer KM, Green LM, Grant RI, Winston KT, Buchmaier S, et al. Restoration of a paraventricular thalamo-accumbal behavioral suppression circuit prevents reinstatement of heroin seeking. Neuron. 2024;112:772–85.e9.
Macé G, Miaczynska M, Zerial M, Nebreda AR. Phosphorylation of EEA1 by p38 MAP kinase regulates mu opioid receptor endocytosis. EMBO J. 2005;24:3235–46.
Bohn LM, Gainetdinov RR, Lin FT, Lefkowitz RJ, Caron MG. Mu-opioid receptor desensitization by beta-arrestin-2 determines morphine tolerance but not dependence. Nature. 2000;408:720–3.
Raehal KM, Schmid CL, Groer CE, Bohn LM. Functional selectivity at the μ-opioid receptor: implications for understanding opioid analgesia and tolerance. Pharmacol Rev. 2011;63:1001–19.
Zhang Y, Zhou P, Wang Z, Chen M, Fu F, Su R. Hsp90β positively regulates μ-opioid receptor function. Life Sci. 2020;252:117676.
Siuda ER, Copits BA, Schmidt MJ, Baird MA, Al-Hasani R, Planer WJ, et al. Spatiotemporal control of opioid signaling and behavior. Neuron. 2015;86:923–35.
Land BB, Bruchas MR, Schattauer S, Giardino WJ, Aita M, Messinger D, et al. Activation of the kappa opioid receptor in the dorsal raphe nucleus mediates the aversive effects of stress and reinstates drug seeking. Proc Natl Acad Sci USA. 2009;106:19168–73.
Di Chiara G, Imperato A. Drugs abused by humans preferentially increase synaptic dopamine concentrations in the mesolimbic system of freely moving rats. Proc Natl Acad Sci USA. 1988;85:5274–8.
Shippenberg TS, Zapata A, Chefer VI. Dynorphin and the pathophysiology of drug addiction. Pharmacol Ther. 2007;116:306–21.
Acknowledgements
This work was supported in part by the National Natural Science Foundation of China (82371533, 82301677); the Natural Science Foundation of Shanghai (22ZR1449300); and the Program of Shanghai Academic/Technology Research Leader (22XD1402400).
Author information
Authors and Affiliations
Contributions
YJL, RSC, TFY, and ZQL designed the experiments; YJL, XYH, and YCS performed the experiments. Statistical data analysis was performed by WJD and XYH. YJL prepared the figures. WJD, YJL, and RSC wrote the manuscript. WJD, GYZ, and BZ provided critical comments on the study design. GYZ, BZ, and ZQL reviewed the manuscript. TFY and ZQL supervised the study.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, Yj., Chen, Rs., Zan, Gy. et al. Adapter protein 2-modulated \({{{\rm{\mu }}}}\)-opioid receptor trafficking in paraventricular thalamus contributes to fentanyl contextual addiction memory in mice. Acta Pharmacol Sin 47, 827–835 (2026). https://doi.org/10.1038/s41401-025-01684-7
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41401-025-01684-7