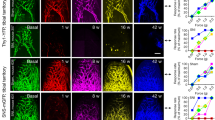
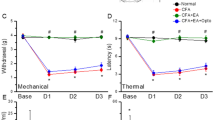
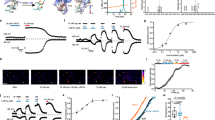
Abstract
Mechanical hyperalgesia is a primary symptom of clinical pain; there remains a therapeutic challenge for severe mechanical and chronic forms of neuropathic pain. The 34-amino-acid neuropeptide GsMTx4, isolated from Tarantula spatulata, was identified as a selective inhibitor of mechanosensitive channels and shown to reduce mechanical hyperalgesia and neuropathic pain in rats. We previously reported two types (type I and type II) of short peptides derived from GsMTx4 that mimicked the inhibitory action of GsMTx4 on a mechanosensitive BK (SAKca) channel. In this study, we investigated whether these short peptides alleviate mechanical hypersensitivity, a major symptom of neuropathic pain. The synthetic type I peptide 01 (Pept 01) was derived from loop2+loop3 of GsMTx4, whereas type II Pept 02, Pept 03 and, Pept 04 were derived from loop 2 of GsMTx4. Carrageenan-induced inflammatory pain was induced in rats and mice, while the chronic constriction nerve injury (CCI) model was established in rats. We showed that administration of short peptides (270 μg/kg, i.p.) selectively inhibited mechanical pain in rats but failed to impact thermal or cold hyperalgesia. Interestingly, the antihyperalgesic effects of these peptides were comparable to those of morphine; however, they were resistant to the μ-opioid receptor antagonist naloxone and lacked morphine-induced side effects, e.g., tolerance and conditioned place preference (CPP). Among them, Pept 03, which contained only one Trp (Trp1) at the head and an additional Arg (Arg11, corresponding to Arg18 in GsMTx4) at the end of the peptide, most potently alleviated mechanical hyperalgesia. Genetic deletion of the TRPV4 gene in mice mostly abolished the analgesic effect of Pept 03. In oocytes expressing TRPV4 channels, application of Pept 03 or Pept 01 inhibited GSK101- or hypotonicity-activated TRPV4 currents in a dose-dependent manner. These results suggest that TRPV4 channels may serve as a direct target for the short peptides in alleviating mechanical pain. This study identifies several natural toxin-based peptides as promising non-opioid analgesics, paving the way for the development of selective and potent painkillers to treat mechanical pain.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout









Similar content being viewed by others
References
Costigan M, Scholz J, Woolf CJ. Neuropathic pain: a maladaptive response of the nervous system to damage. Annu Rev Neurosci. 2009;32:1–32.
Murthy SE, Loud MC, Daou I, Marshall KL, Schwaller F, Kühnemund J, et al. The mechanosensitive ion channel Piezo2 mediates sensitivity to mechanical pain in mice. Sci Transl Med. 2018;10:eaat9897.
Bates D, Schultheis BC, Hanes MC, Jolly SM, Chakravarthy KV, Deer TR, et al. A comprehensive algorithm for management of neuropathic pain. Pain Med. 2019;20:S2–12.
Hu X, Du L, Liu S, Zhou L, Kaikai Z, Jing F, et al. A TRPV4-dependent neuroimmune axis in the spinal cord promotes neuropathic pain. J Clin Invest. 2023;133:e161507.
Szczot M, Liljencrantz J, Ghitani N, Barik A, Lam R, Thompson LH, et al. PIEZO2 mediates injury-induced tactile pain in mice and humans. Sci Transl Med. 2018;10:eaat9892
Fernandez-Trillo, Florez-Paz J, Inigo-Portugues D, Íñigo-Portugués A, González-González A, Del O1, et al. Piezo2 mediates low-threshold mechanically evoked pain in the cornea. J Neurosci. 2020;40:8976–93.
Caterina MJ, Schumacher MA, Tominaga M, Rosen TA, Levine JD, Julius D. The capsaicin receptor: a heat-activated ion channel in the pain pathway. Nature. 1997;389:816–24.
McKemy DD, Neuhausser WM, Julius D. Identification of a cold receptor reveals a general role for TRP channels in thermosensation. Nature. 2002;416:52–8.
Story GM, Peier AM, Reeve AJ, Eid SR, Mosbacher J, Hricik TR, et al. ANKTM1, a TRP-like channel expressed in nociceptive neurons, is activated by cold temperatures. Cell. 2003;112:819–29.
Grace MS, Bonvini SJ, Belvisi MG, McIntyre P. Modulation of the TRPV4 ion channel as a therapeutic target for disease. Pharmacol Ther. 2017;177:9–22.
Alessandri-Haber N, Dina OA, Yeh JJ, Parada CA, Reichling DB, Levine JD. Transient receptor potential vanilloid 4 is essential in chemotherapy-induced neuropathic pain in the rat. J Neurosci. 2004;24:4444–52.
Alessandri-Haber N, Joseph E, Dina OA, Liedtke W, Levine JD. TRPV4 mediates pain-related behavior induced by mild hypertonic stimuli in the presence of inflammatory mediator. Pain. 2005;118:70–9.
Alessandri-Haber N, Yeh JJ, Boyd AE, Parada CA, Chen X, Reichling DB, et al. Hypotonicity induces TRPV4-mediated nociception in rat. Neuron. 2003;39:497–511.
Choi G, Yang TJ, Yoo S, Choi SI, Lim JY, Cho PS, et al. TRPV4-mediated anti-nociceptive effect of suberanilohydroxamic acid on mechanical pain. Mol Neurobiol. 2019;56:444–53.
Li H, Xu J, Shen ZS, Wang GM, Tang M, Du XR, et al. The neuropeptide GsMTx4 inhibits a mechanosensitive BK channel through the voltage-dependent modification specific to mechano-gating. J Biol Chem. 2019;294:11892–909.
Suchyna TM, Tape SE, Koeppe RE 2nd, Andersen OS, Sachs F, et al. Bilayer-dependent inhibition of mechanosensitive channels by neuroactive peptide enantiomers. Nature. 2004;430:235–40.
Lee W, Leddy HA, Chen Y, Lee SH, Zelenski NA, McNulty AL, et al. Synergy between Piezo1 and Piezo2 channels confers high-strain mechanosensitivity to articular cartilage. Proc Natl Acad Sci USA. 2014;111:E5114–22.
Suchyna TM, Johnson JH, Hamer K, Leykam JF, Gage DA, Clemo HF, et al. Identification of a peptide toxin from Grammostola spatulata spider venom that blocks cation-selective stretch-activated channels. J Gen Physiol. 2000;115:583–98.
Park SP, Kim BM, Koo JY, Cho H, Lee CH, Kim M, et al. A tarantula spider toxin, GsMTx4, reduces mechanical and neuropathic pain. Pain. 2008;137:208–17.
Alessandri-Haber N, Dina OA, Chen X, Levine JD. TRPC1 and TRPC6 channels cooperate with TRPV4 to mediate mechanical hyperalgesia and nociceptor sensitization. J Neurosci. 2009;29:6217–28.
Zhou N, Li H, Xu J, Shen ZS, Tang M, Wang XH, et al. Two types of peptides derived from the neurotoxin GsMTx4 inhibit a mechanosensitive potassium channel by modifying the mechano-gate. J Biol Chem. 2022;298:102326.
Ke SX, Dong P, Mei Y, Wang J, Tang M, Su A, et al. A synthetic peptide, derived from neurotoxin GsMTx4, acts as a non-opioid analgesic to alleviate mechanical and neuropathic pain through the TRPV4 channel. Acta Pharm Sin B. 2025;15:1447–62.
Percie du Sert N, Hurst V, Ahluwalia A, Alam S, Avey MT, Baker M, et al. The ARRIVE guidelines 2.0: updated guidelines for reporting animal research. BMJ Open Sci. 2020;4:e100115.
Lilley E, Stanford SC, Kendall DE, Alexander SPH, Cirino G, Docherty JR, et al. ARRIVE 2.0 and the British Journal of Pharmacology: Updated guidance for 2020. Br J Pharmacol. 2020;177:3611–6.
Mizuno A, Matsumoto N, Imai M, Suzuki M. Impaired osmotic sensation in mice lacking TRPV4. Am J Physiol-Cell Ph. 2003;285:C96–C101.
Zou Y, Zhang M, Wu Q, Zhao N, Chen M, Yang C, et al. Activation of transient receptor potential vanilloid 4 is involved in pressure overload-induced cardiac hypertrophy. Elife. 2022;11:e74519.
Diochot S, Baron A, Salinas M, Douguet D, Scarzello S, Dabert-Gay AS, et al. Black mamba venom peptides target acid-sensing ion channels to abolish pain. Nature. 2012;490:552–5.
Bennett GJ, Xie YK. A peripheral mononeuropathy in rat that produces disorders of pain sensation like those seen in man. Pain. 1988;33:87–107.
Kiso T, Sekizawa T, Uchino H, Tsukamoto M, Kakimoto S. Analgesic effects of ASP3662, a novel 11beta-hydroxysteroid dehydrogenase 1 inhibitor, in rat models of neuropathic and dysfunctional pain. Br J Pharmacol. 2018;175:3784–96.
Zhang Y, Huang L, Kozlov SA, Rubini P, Tang Y, Illes P. Acupuncture alleviates acid- and purine-induced pain in rodents. Br J Pharmacol. 2020;177:77–92.
Liu D, Tang QQ, Yin C, Song Y, Liu Y, Yang JX, et al. Brain-derived neurotrophic factor-mediated projection-specific regulation of depressive-like and nociceptive behaviors in the mesolimbic reward circuitry. Pain. 2018;159:175.
Pan Z, Li GF, Sun ML, Sun ML, Zhang M, Hao LY, et al. MicroRNA-1224 splicing CircularRNA-Filip1l in an Ago2-dependent manner regulates chronic inflammatory pain via targeting Ubr5. J Neurosci. 2019;39:2125–43.
Brenner DS, Golden JP, Gereau. RWt. A novel behavioral assay for measuring cold sensation in mice. PLoS One. 2012;7:e39765.
Ehlers VL, Sadler KE, Stucky CL. Peripheral transient receptor potential vanilloid type 4 hypersensitivity contributes to chronic sickle cell disease pain. Pain. 2023;164:1874–86.
Tian JH, Zhang W, Fang Y, Xu W, Grandy DK, Han JS. Endogenous orphanin FQ: evidence for a role in the modulation of electroacupuncture analgesia and the development of tolerance to analgesia produced by morphine and electroacupuncture. Br J Pharmacol. 1998;124:21–6.
Deng M, Chen SR, Chen H, Pan HL. Alpha2delta-1-bound N-methyl-D-aspartate receptors mediate morphine-induced hyperalgesia and analgesic tolerance by potentiating glutamatergic input in rodents. Anesthesiology. 2019;130:804–19.
Portugal GS, Al-Hasani R, Fakira AK, Gonzalez-Romero JL, Melyan Z, McCall JG, et al. Hippocampal long-term potentiation is disrupted during expression and extinction but is restored after reinstatement of morphine place preference. J Neurosci. 2014;34:527–38.
Portugal GS, Gould TJ. Nicotine withdrawal disrupts new contextual learning. Pharmacol Biochem Behav. 2009;92:117–23.
Rodriguez-Menchaca AA, Adney SK, Tang QY, Meng XY, Rosenhouse-Dantsker A, Cui M, et al. PIP2 controls voltage-sensor movement and pore opening of Kv channels through the S4-S5 linker. Proc Natl Acad Sci USA. 2012;109:E2399–2408.
Humphrey W, Dalke A, Schulten K. VMD: visual molecular dynamics. J Mol Graph. 1996;14:33–38. 27-38.
Nishizawa K, Nishizawa M, Gnanasambandam R, Sachs F, Sukharev SI, Suchyna TM. Effects of Lys to Glu mutations in GsMTx4 on membrane binding, peptide orientation, and self-association propensity, as analyzed by molecular dynamics simulations. Biochim Biophys Acta. 2015;1848:2767–78.
Tang QY, Zhang Z, Meng XY, Cui M, Logothetis DE. Structural determinants of phosphatidylinositol 4,5-bisphosphate (PIP2) regulation of BK channel activity through the RCK1 Ca2+ coordination site. J Biol Chem. 2014;289:18860–72.
Tang QY, Zhang Z, Xia J, Ren D, Logothetis DE. Phosphatidylinositol 4,5-bisphosphate activates Slo3 currents and its hydrolysis underlies the epidermal growth factor-induced current inhibition. J Biol Chem. 2010;285:19259–66.
Tang QY, Larry T, Hendra K, Yamamoto E, Bell J, Cui M, et al. Mutations in nature conferred a high affinity phosphatidylinositol 4,5-bisphosphate-binding site in vertebrate inwardly rectifying potassium channels. J Biol Chem. 2015;290:16517–29.
Xu J, Lv YT, Zhao XY, Wang JJ, Shen ZS, Li J, et al. Identification of sodium- and chloride-sensitive sites in the Slack channel. J Neurosci. 2023;43:2665–81.
Loukin S, Zhou X, Su Z, Saimi Y, Kung C. Wild-type and brachyolmia-causing mutant TRPV4 channels respond directly to stretch force. J Biol Chem. 2010;285:27176–81.
Zhang Q, Gao SH, Shen ZS, Wang Y, Hu SW, Duan GB, et al. The Slack channel regulates anxiety-like behaviors via basolateral amygdala glutamatergic projections to ventral hippocampus. J Neurosci. 2022;42:3049–64.
Tang QY, Zhang FF, Xu J, Wang R, Chen J, Logothetis DE, et al. Epilepsy-related Slack channel mutants lead to channel over-activity by two different mechanisms. Cell Rep. 2016;14:129–39.
Xu XY, Zhang FF, Gan J, Zhang MY, Shen ZS, Guo Q, et al. Identification of the acid-sensitive site critical for chloral hydrate (CH) activation of the proton-activated chloride channel. J Neurosci. 2023;43:526–39.
Wang GM, Zhong ZG, Du XR, Zhang FF, Guo Q, Liu Y, et al. Cloning and characterization of the rat Slo3 (KCa 5.1) channel: from biophysics to pharmacology. Br J Pharmacol. 2020;177:3552–67.
Tang QY, Zhang Z, Xia XM, Lingle CJ. Block of mouse Slo1 and Slo3 K+ channels by CTX, IbTX, TEA, 4-AP and quinidine. Channels (Austin). 2010;4:22–41.
Curtis MJ, Alexander S, Cirino G, Docherty JR, George CH, Giembycz MA, et al. Experimental design and analysis and their reporting II: updated and simplified guidance for authors and peer reviewers. Br J Pharmacol. 2018;175:987–93.
Kuo YL, Cheng JK, Hou WH, Chang YC, Du PH, Jian JJ, et al. K+ channel modulatory subunits KChIP and DPP participate in Kv4-mediated mechanical pain control. J Neurosci. 2017;37:4391–404.
Escoubas P, De Weille JR, Lecoq A, Diochot S, Waldmann R, Champigny G, et al. Isolation of a tarantula toxin specific for a class of proton-gated Na+ channels. J Biol Chem. 2000;275:25116–21.
Oswald RE, Suchyna TM, McFeeters R, Gottlieb P, Sachs F. Solution structure of peptide toxins that block mechanosensitive ion channels. J Biol Chem. 2002;277:34443–50.
Chen K, Wang T, Li Y, Wu J, Zhao CX, Liu S, et al. Rhodojaponin VI indirectly targets Cav2.2 channels via N-ethylmaleimide-sensitive fusion protein to alleviate neuropathic pain. Acta Pharm Sin B. 2023;13:1326–36.
Gnanasambandam R, Ghatak C, Yasmann A, Nishizawa K, Sachs F, Ladokhin AS, et al. GsMTx4: mechanism of inhibiting mechanosensitive ion channels. Biophys J. 2017;112:31–45.
Bohn LM, Gainetdinov RR, Lin FT, Lefkowitz RJ, Caron MG. Mu-opioid receptor desensitization by beta-arrestin-2 determines morphine tolerance but not dependence. Nature. 2000;408:720–3.
Swain SM, Romac JM, Vigna SR, Liddle RA. Piezo1-mediated stellate cell activation causes pressure-induced pancreatic fibrosis in mice. JCI Insight. 2022;7:e158288.
Tang QY, Kolanos R, De Felice LJ, Glennon RA. Structural analysis of dopamine- and amphetamine-induced depolarization currents in the human dopamine transporter. ACS Chem Neurosci. 2015;6:551–8.
Han Z, Shen Z, Pei J, You Q, Zhang Q, Wang L. Transformation of peptides to small molecules in medicinal chemistry: challenges and opportunities. Acta Pharm Sin B. 2024;14:4243–65.
Hayashi Y, Morinaga S, Zhang J, Satoh Y, Meredith AL, Nakata T, et al. BK channels in microglia are required for morphine-induced hyperalgesia. Nat Commun. 2016;7:11697.
Alsaloum M, Higerd GP, Effraim PR, Waxman SG. Status of peripheral sodium channel blockers for non-addictive pain treatment. Nat Rev Neurol. 2020;16:689–705. Dec.
Perez de Vega MJ, Ferrer-Montiel A, Gonzalez-Muniz R. Recent progress in non-opioid analgesic peptides. Arch Biochem Biophys. 2018;660:36–52.
Acknowledgements
We would like to thank Raymond Zhang for his help with the pain behavior test, data analysis, and grammar check for the final version. We thank Prof. Atsuko Mizuno (Jichi Medical University, Japan) for Trpv4 KO mice, Dr. Fan Yang (Zhejiang University, China) for providing the TRPV4 cDNA plasmids. We are also grateful to Dr. Linda Boland (University of Richmond, USA) for the good suggestion and English editing at the earlier stage. This work was supported by NSFC grants (81450064, 82371233) to QYT and NSFC grants (81471314, 81671090) to ZZ, Luzhou Science and Technology Bureau (2021-SYF-28) and Southwest Medical University of China (2021ZKMS033) grants to MXT, Key Project in Sichuan province department of education to MXT (16ZA0196), and Jiangsu specially appointed professorship to QYT and ZZ.
Author information
Authors and Affiliations
Contributions
PD, QKF, SXK, JQW, YM, JW, YTD, YYX, JJW, CC, and XHW performed research and data analysis; XJ, NZ, and MC contributed to MD simulations. YMD provides the TRPV4-KO mice. MS, MXT, ZZ, and QYT designed the experiment and supervised experiments. MXT, ZZ, and QYT wrote the original draft.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests. Qiong-yao Tang, Zhe Zhang, Ming-xi Tang, Ping Dong, and Shao-xi Ke have applied for two patents related to this study.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Dong, P., Fan, Qk., Ke, Sx. et al. Discovery of non-opioid peptides that selectively relieve mechanical pain in rodents through inhibition of TRPV4 channels. Acta Pharmacol Sin (2026). https://doi.org/10.1038/s41401-025-01698-1
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41401-025-01698-1