Abstract
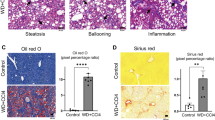
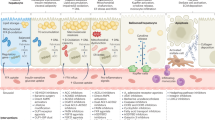
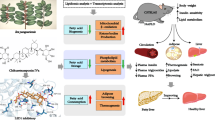
Metabolic dysfunction-associated steatohepatitis (MASH), an inflammatory subtype of metabolic dysfunction-associated fatty liver disease (MAFLD), drives hepatic dysfunction and poses a significant health burden. Lipophagy dysfunction disrupts lipid droplet degradation and induces lysosomal damage, which is closely linked to MASH progression; thus, targeting lipophagy-lysosomal activation has emerged as a promising therapeutic strategy for the therapy of MASH. β-Sitosterol (β-SIT) derived from Polygonum hydropiper L. is structurally similar to cholesterol, and exhibits neuroprotective, antidiabetic and anti-obesity bioactivities. In this study, we explored the therapeutic potential of β-SIT for MASH. The mouse models of MASH were established by feeding a choline-deficient, L-amino acid-defined high-fat diet (CDAHFD) for 10 weeks, or high-fat diet (HFD) for 12 weeks. For in vitro experiments, AML-12 cells were treated with FFA mixture (OA:PA molar ratio = 2:1) to mimic lipid overload condition. MASH mice were administered β-SIT (10 or 20 mg·kg−1 d−1, i.g.) for 10 weeks. We showed that β-SIT treatment dose-dependently alleviated MASH by enhancing the lipophagy-lysosomal pathway in vivo and in vitro. In FFA-stimulated AML-12 cells, we demonstrated that β-SIT (20 μM) activated autophagic flux, promoted lysosomal biogenesis, and enhanced lysosome-lipid droplet interactions, as revealed by transmission electron microscopy, multi-SIM real-time fluorescence monitoring, and lipophagy-related marker detection. By integrated approaches including bioinformatics, molecular dynamics, CETSA and functional assays, we found that β-SIT inhibited mTOR pathway activation by directly targeting Ras-related C3 botulinum toxin substrate 1 (RAC1) in MASH mice. By conducting imaging/3D reconstruction, co-immunoprecipitation, immunofluorescence colocalization, lysosomal fractionation, and biochemical analyses in FFA-stimulated AML-12 cells, we confirmed that β-SIT modulated RAC1/mTOR interactions on lysosomes to restore lipophagy function. Critically, β-SIT promoted transcription factor EB (TFEB) nuclear translocation by modulating the RAC1-mTOR axis, thereby repairing lipophagy-lysosomal defects and attenuating MASH progression. Our results suggest that targeting the RAC1-mTOR-TFEB axis is a novel mechanism of β-SIT-driven lipophagy-lysosomal regulation, and highlight β-SIT as a potential candidate for the treatment of MASH.

This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Feng G, Valenti L, Wong VW, Fouad YM, Yilmaz Y, Kim W, et al. Recompensation in cirrhosis: unravelling the evolving natural history of nonalcoholic fatty liver disease. Nat Rev Gastroenterol Hepatol. 2024;21:46–56.
Younossi Z, Tacke F, Arrese M, Chander Sharma B, Mostafa I, Bugianesi E, et al. Global perspectives on nonalcoholic fatty liver disease and nonalcoholic steatohepatitis. Hepatology. 2019;69:2672–82.
Younossi ZM, Koenig AB, Abdelatif D, Fazel Y, Henry L, Wymer M. Global epidemiology of nonalcoholic fatty liver disease-meta-analytic assessment of prevalence, incidence, and outcomes. Hepatology. 2016;64:73–84.
Tao L, Yi Y, Chen Y, Zhang H, Orning P, Lien E, et al. RIP1 kinase activity promotes steatohepatitis through mediating cell death and inflammation in macrophages. Cell Death Differ. 2021;28:1418–33.
Powell EE, Wong VW, Rinella M. Non-alcoholic fatty liver disease. Lancet. 2021;397:2212–24.
El-Agroudy NN, Kurzbach A, Rodionov RN, O’Sullivan J, Roden M, Birkenfeld AL, et al. Are lifestyle therapies effective for NAFLD treatment?. Trends Endocrinol Metab. 2019;30:701–9.
Petroni ML, Brodosi L, Bugianesi E, Marchesini G. Management of non-alcoholic fatty liver disease. BMJ. 2021;372:m4747.
Li QP, Dou YX, Huang ZW, Chen HB, Li YC, Chen JN, et al. Therapeutic effect of oxyberberine on obese non-alcoholic fatty liver disease rats. Phytomedicine. 2021;85:153550.
Nandi S, Nag A, Khatua S, Sen S, Chakraborty N, Naskar A, et al. Anticancer activity and other biomedical properties of β-sitosterol: bridging phytochemistry and current pharmacological evidence for future translational approaches. Phytother Res. 2024;38:592–619.
Ayaz M, Junaid M, Ullah F, Subhan F, Sadiq A, Ali G, et al. Anti-Alzheimer’s studies on β-sitosterol isolated from Polygonum hydropiper L. Front Pharmacol. 2017;8:697.
Wang H, Liu J, Zhang Z, Peng J, Wang Z, Yang L, et al. β-Sitosterol targets ASS1 for Nrf2 ubiquitin-dependent degradation, inducing ROS-mediated apoptosis via the PTEN/PI3K/AKT signaling pathway in ovarian cancer. Free Radic Biol Med. 2024;214:137–57.
Hah YS, Lee WK, Lee S, Kim EJ, Lee JH, Lee SJ, et al. β-Sitosterol attenuates dexamethasone-induced muscle atrophy via regulating FoxO1-dependent signaling in C2C12 cell and mice model. Nutrients. 2022;14:2894.
Jayaraman S, Devarajan N, Rajagopal P, Babu S, Ganesan SK, Veeraraghavan VP, et al. β-Sitosterol circumvents obesity induced inflammation and insulin resistance by down-regulating IKKβ/NF-κB and JNK signaling pathway in adipocytes of type 2 diabetic rats. Molecules. 2021;26:2101.
Filali-Mouncef Y, Hunter C, Roccio F, Zagkou S, Dupont N, Primard C, et al. The ménage à trois of autophagy, lipid droplets and liver disease. Autophagy. 2022;18:50–72.
Jung EJ, Sung KW, Bae TH, Kim HY, Choi HR, Kim SH, et al. The N-degron pathway mediates lipophagy: the chemical modulation of lipophagy in obesity and NAFLD. Metabolism. 2023;146:155644.
Martinez-Lopez N, Garcia-Macia M, Sahu S, Athonvarangkul D, Liebling E, Merlo P, et al. Autophagy in the CNS and periphery coordinate lipophagy and lipolysis in the brown adipose tissue and liver. Cell Metab. 2016;23:113–27.
Minami Y, Hoshino A, Higuchi Y, Hamaguchi M, Kaneko Y, Kirita Y, et al. Liver lipophagy ameliorates nonalcoholic steatohepatitis through extracellular lipid secretion. Nat Commun. 2023;14:4084.
Liu K, Qiu D, Liang X, Huang Y, Wang Y, Jia X, et al. Lipotoxicity-induced STING1 activation stimulates MTORC1 and restricts hepatic lipophagy. Autophagy. 2022;18:860–76.
Rawnsley DR, Diwan A. Lysosome impairment as a trigger for inflammation in obesity: the proof is in the fat. EBioMedicine. 2020;56:102824.
Du J, Ji X, Xu B, Du Q, Li Y, Zhou B, et al. Ubiquitination of cytoplasmic HMGB1 by RNF186 regulates hepatic lipophagy in non-alcoholic fatty liver disease. Metabolism. 2024;152:155769.
Sanyal AJ, Friedman SL, McCullough AJ, Dimick-Santos L. Challenges and opportunities in drug and biomarker development for nonalcoholic steatohepatitis: findings and recommendations from an American Association for the Study of Liver Diseases-U.S. Food and Drug Administration Joint Workshop. Hepatology. 2015;61:1392–405.
Pacheco Y, Lim CX, Weichhart T, Valeyre D, Bentaher A, Calender A. Sarcoidosis and the mTOR, Rac1, and autophagy triad. Trends Immunol. 2020;41:286–99.
Saci A, Cantley LC, Carpenter CL. Rac1 regulates the activity of mTORC1 and mTORC2 and controls cellular size. Mol Cell. 2011;42:50–61.
Du X, Di Malta C, Fang Z, Shen T, Niu X, Chen M, et al. Nuciferine protects against high-fat diet-induced hepatic steatosis and insulin resistance via activating TFEB-mediated autophagy-lysosomal pathway. Acta Pharm Sin B. 2022;12:2869–86.
Li W, Li Y, Guan Y, Du Y, Zhao M, Chen X, et al. TNFAIP8L2/TIPE2 impairs autolysosome reformation via modulating the RAC1-MTORC1 axis. Autophagy. 2021;17:1410–25.
Sharma M, Urano F, Jaeschke A. Cdc42 and Rac1 are major contributors to the saturated fatty acid-stimulated JNK pathway in hepatocytes. J Hepatol. 2012;56:192–8.
Marei H, Malliri A. Rac1 in human diseases: the therapeutic potential of targeting Rac1 signaling regulatory mechanisms. Small GTPases. 2017;8:139–63.
Choi SS, Sicklick JK, Ma Q, Yang L, Huang J, Qi Y, et al. Sustained activation of Rac1 in hepatic stellate cells promotes liver injury and fibrosis in mice. Hepatology. 2006;44:1267–77.
Zhang C, Duan Y, Xia M, Dong Y, Chen Y, Zheng L, et al. TFEB mediates immune evasion and resistance to mTOR inhibition of renal cell carcinoma via induction of PD-L1. Clin Cancer Res. 2019;25:6827–38.
Medina DL, Fraldi A, Bouche V, Annunziata F, Mansueto G, Spampanato C, et al. Transcriptional activation of lysosomal exocytosis promotes cellular clearance. Dev Cell. 2011;21:421–30.
Malik N, Ferreira BI, Hollstein PE, Curtis SD, Trefts E, Weiser Novak S, et al. Induction of lysosomal and mitochondrial biogenesis by AMPK phosphorylation of FNIP1. Science. 2023;380:eabj5559.
Li H, Gong Y, Wang Y, Sang W, Wang C, Zhang Y, et al. β-Sitosterol modulates osteogenic and adipogenic balance in BMSCs to suppress osteoporosis via regulating mTOR-IMP1-Adipoq axis. Phytomedicine. 2025;138:156400.
de Moraes Arnoso BJ, Magliaccio FM, de Araújo CA, de Andrade Soares R, Santos IB, de Bem GF, et al. Açaí seed extract (ASE) rich in proanthocyanidins improves cardiovascular remodeling by increasing antioxidant response in obese high-fat diet-fed mice. Chem Biol Interact. 2022;351:109721.
Li B, Xiao Q, Zhao H, Zhang J, Yang C, Zou Y, et al. Schisanhenol ameliorates non-alcoholic fatty liver disease via inhibiting miR-802 activation of AMPK-mediated modulation of hepatic lipid metabolism. Acta Pharm Sin B. 2024;14:3949–63.
Xu R, Xiao X, Zhang S, Pan J, Tang Y, Zhou W, et al. The methyltransferase METTL3-mediated fatty acid metabolism revealed the mechanism of cinnamaldehyde on alleviating steatosis. Biomed Pharmacother. 2022;153:113367.
Matsumoto M, Hada N, Sakamaki Y, Uno A, Shiga T, Tanaka C, et al. An improved mouse model that rapidly develops fibrosis in non-alcoholic steatohepatitis. Int J Exp Pathol. 2013;94:93–103.
Zhu W, Hong Y, Tong Z, He X, Li Y, Wang H, et al. Activation of hepatic adenosine A1 receptor ameliorates MASH via inhibiting SREBPs maturation. Cell Rep Med. 2024;5:101477.
Dixon ED, Claudel T, Nardo AD, Riva A, Fuchs CD, Mlitz V, et al. Inhibition of ATGL alleviates MASH via impaired PPARα signalling that favours hydrophilic bile acid composition in mice. J Hepatol. 2025;82:658–75.
Zhang C, Tong Q, Liu K, Mao T, Song Y, Qu Y, et al. Morroniside delays the progression of non-alcoholic steatohepatitis by promoting AMPK-mediated lipophagy. Phytomedicine. 2024;129:155703.
Zhang H, Lu J, Liu H, Guan L, Xu S, Wang Z, et al. Ajugol enhances TFEB-mediated lysosome biogenesis and lipophagy to alleviate non-alcoholic fatty liver disease. Pharmacol Res. 2021;174:105964.
Schulze RJ, Krueger EW, Weller SG, Johnson KM, Casey CA, Schott MB, et al. Direct lysosome-based autophagy of lipid droplets in hepatocytes. Proc Natl Acad Sci USA. 2020;117:32443–52.
Kim YC, Guan KL. mTOR: a pharmacologic target for autophagy regulation. J Clin Invest. 2015;125:25–32.
Settembre C, Zoncu R, Medina DL, Vetrini F, Erdin S, Erdin S, et al. A lysosome-to-nucleus signalling mechanism senses and regulates the lysosome via mTOR and TFEB. EMBO J. 2012;31:1095–108.
Yang H, Jiang X, Li B, Yang HJ, Miller M, Yang A, et al. Mechanisms of mTORC1 activation by RHEB and inhibition by PRAS40. Nature. 2017;552:368–73.
Das D, Sharma M, Gahlot D, Nia SS, Gain C, Mecklenburg M, et al. VPS4A is the selective receptor for lipophagy in mice and humans. Mol Cell. 2024;84:4436–53.e8.
Yin L, Zhou J, Li T, Wang X, Xue W, Zhang J, et al. Inhibition of the dopamine transporter promotes lysosome biogenesis and ameliorates Alzheimer’s disease-like symptoms in mice. Alzheimers Dement. 2023;19:1343–57.
Wang T, Qin Y, Ye Z, Jing DS, Fan GX, Liu MQ, et al. A new glance at autophagolysosomal-dependent or -independent function of transcriptional factor EB in human cancer. Acta Pharmacol Sin. 2023;44:1536–48.
Zhou W, Yan X, Zhai Y, Liu H, Guan L, Qiao Y, et al. Phillygenin ameliorates nonalcoholic fatty liver disease via TFEB-mediated lysosome biogenesis and lipophagy. Phytomedicine. 2022;103:154235.
Pan A, Jin J, Wu Y, Zhang Q, Chen H, Hu Y, et al. Carabrone attenuates metabolic dysfunction-associated steatohepatitis by targeting STAT3 in mice. MedComm. 2025;6:e70145.
Sancak Y, Bar-Peled L, Zoncu R, Markhard AL, Nada S, Sabatini DM. Ragulator-Rag complex targets mTORC1 to the lysosomal surface and is necessary for its activation by amino acids. Cell. 2010;141:290–303.
Huang P, Dong RY, Wang P, Xu M, Sun X, Dong XP. MCOLN/TRPML channels in the regulation of MTORC1 and autophagy. Autophagy. 2024;20:1203–4.
Hosios AM, Wilkinson ME, McNamara MC, Kalafut KC, Torrence ME, Asara JM, et al. mTORC1 regulates a lysosome-dependent adaptive shift in intracellular lipid species. Nat Metab. 2022;4:1792–811.
Goul C, Peruzzo R, Zoncu R. Author Correction: The molecular basis of nutrient sensing and signalling by mTORC1 in metabolism regulation and disease. Nat Rev Mol Cell Biol. 2023;24:934.
Wang Z, Fayngerts S, Wang P, Sun H, Johnson DS, Ruan Q, et al. TIPE2 protein serves as a negative regulator of phagocytosis and oxidative burst during infection. Proc Natl Acad Sci USA. 2012;109:15413–8.
Tan A, Prasad R, Lee C, Jho EH. Past, present, and future perspectives of transcription factor EB (TFEB): mechanisms of regulation and association with disease. Cell Death Differ. 2022;29:1433–49.
Chen M, Liu G, Fang Z, Gao W, Song Y, Lei L, et al. Buddleoside alleviates nonalcoholic steatohepatitis by targeting the AMPK-TFEB signaling pathway. Autophagy. 2025;21:1316–34.
Roczniak-Ferguson A, Petit CS, Froehlich F, Qian S, Ky J, Angarola B, et al. The transcription factor TFEB links mTORC1 signaling to transcriptional control of lysosome homeostasis. Sci Signal. 2012;5:ra42.
Yang J, Yuan L, Li L, Liu F, Liu J, Chen Y, et al. Trehalose activates autophagy to alleviate cisplatin-induced chronic kidney injury by targeting the mTOR-dependent TFEB signaling pathway. Theranostics. 2025;15:2544–63.
Zhou XY, Lai YH, Zhang J, Li Y, Wu XM, Yang YZ, et al. Advanced oxidation protein products attenuate the autophagy-lysosome pathway in ovarian granulosa cells by modulating the ROS-dependent mTOR-TFEB pathway. Cell Death Dis. 2024;15:161.
Acknowledgements
This work received partial financial support from the National Natural Science Foundation of China (grant number: 82073851).
Author information
Authors and Affiliations
Contributions
YW, YS and CYW performed the experiment; YW drafted the article; MC and RH collected and interpreted the data; SW performed the analysis with constructive discussions; HJS designed the study, obtained funding, supervised the study, drafted and critically revised the paper. All the authors have read and approved the final paper. All data were generated in-house, and no paper mill was used.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, Y., Sun, Y., Wang, Cy. et al. β-Sitosterol ameliorates metabolic dysfunction-associated steatohepatitis by targeting the RAC1/mTOR/TFEB axis thus activating lipophagy-lysosomal pathway. Acta Pharmacol Sin 47, 946–963 (2026). https://doi.org/10.1038/s41401-025-01702-8
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41401-025-01702-8