Abstract
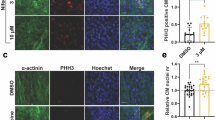
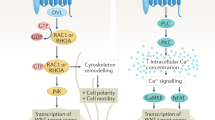
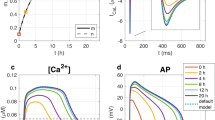
The adult human heart is incapable of regeneration after myocardial infarction (MI) injury. One potential therapeutic strategy is to enhance the proliferation of resident cardiomyocytes (CMs). In this study, we developed a high-content screening assay based on DNA synthesis in human pluripotent stem cell-derived cardiomyocytes (hPSC-CMs) to identify small molecules that could promote CM proliferation. In the primary screening, we found that L-type calcium channel (LTCC) blockers induced DNA synthesis of hPSC-CMs. Among the 6 clinically approved calcium channel blockers tested in secondary screening and confirmatory experiments, nimodipine (NM) consistently enhanced CM proliferation both in vitro and in vivo. RNA-Seq analysis revealed that NM activated the canonical Wnt signaling pathway, while inhibiting Wnt signaling blunted the proliferative effect of NM. Lrp5, a co-receptor for Wnt ligands known to interact with LTCC, was found to mediate the effect of NM to promote nuclear localization of β-catenin and CM proliferation. In the MI mouse model established by ligating the left anterior descending coronary artery, administration of NM (10 mg/kg, i.p.) for 7 consecutive days significantly improved cardiac contractile function and enhanced resident CM proliferation, which was attenuated by co-treatment with Wnt inhibitor Wnt-C59 (10 mg/kg, i.p.). Our data suggest that L-type calcium channel blockers that induce CM proliferation may be potentially used in the treatment of MI and heart failure to promote cardiac regeneration.

This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
All data associated with this study are present in the paper or the Supplementary Materials. RNA-seq data generated in this study have been deposited into the Gene Expression Omnibus (GEO) database (GSE185542).
References
Sadek H, Olson EN. Toward the goal of human heart regeneration. Cell Stem Cell. 2020;26:7–16.
Poss KD, Wilson LG, Keating MT. Heart regeneration in zebrafish. Science. 2002;298:2188–90.
Porrello ER, Mahmoud AI, Simpson E, Hill JA, Richardson JA, Olson EN, et al. Transient regenerative potential of the neonatal mouse heart. Science. 2011;331:1078–80.
Xin M, Kim Y, Sutherland LB, Murakami M, Qi X, McAnally J, et al. Hippo pathway effector Yap promotes cardiac regeneration. Proc Natl Acad Sci USA. 2013;110:13839–44.
Mohamed TMA, Ang YS, Radzinsky E, Zhou P, Huang Y, Elfenbein A, et al. Regulation of cell cycle to stimulate adult cardiomyocyte proliferation and cardiac regeneration. Cell. 2018;173:104–116 e112.
Aguirre A, Montserrat N, Zacchigna S, Nivet E, Hishida T, Krause MN, et al. In vivo activation of a conserved microrna program induces mammalian heart regeneration. Cell Stem Cell. 2014;15:589–604.
Wei K, Serpooshan V, Hurtado C, Diez-Cunado M, Zhao M, Maruyama S, et al. Epicardial fstl1 reconstitution regenerates the adult mammalian heart. Nature. 2015;525:479–85.
Bassat E, Mutlak YE, Genzelinakh A, Shadrin IY, Baruch Umansky K, Yifa O, et al. The extracellular matrix protein agrin promotes heart regeneration in mice. Nature. 2017;547:179–84.
Liu X, De la Cruz E, Gu X, Balint L, Oxendine-Burns M, Terrones T, et al. Lymphoangiocrine signals promote cardiac growth and repair. Nature. 2020;588:705–11.
Vujic A, Natarajan N, Lee RT. Molecular mechanisms of heart regeneration. Semin Cell Dev Biol. 2020;100:20–8.
Eulalio A, Mano M, Dal Ferro M, Zentilin L, Sinagra G, Zacchigna S, et al. Functional screening identifies miRNAs inducing cardiac regeneration. Nature. 2012;492:376–81.
Uosaki H, Magadum A, Seo K, Fukushima H, Takeuchi A, Nakagawa Y, et al. Identification of chemicals inducing cardiomyocyte proliferation in developmental stage-specific manner with pluripotent stem cells. Circ Cardiovasc Genet. 2013;6:624–33.
Lian X, Hsiao C, Wilson G, Zhu K, Hazeltine LB, Azarin SM, et al. Robust cardiomyocyte differentiation from human pluripotent stem cells via temporal modulation of canonical Wnt signaling. Proc Natl Acad Sci USA. 2012;109:E1848–1857.
Dai DF, Danoviz ME, Wiczer B, Laflamme MA, Tian R. Mitochondrial maturation in human pluripotent stem cell derived cardiomyocytes. Stem Cells Int. 2017;2017:5153625.
Sharma A, Burridge PW, McKeithan WL, Serrano R, Shukla P, Sayed N, et al. High-throughput screening of tyrosine kinase inhibitor cardiotoxicity with human induced pluripotent stem cells. Sci Transl Med 2017; 9:eaaf2584.
da Rocha AM, Campbell K, Mironov S, Jiang J, Mundada L, Guerrero-Serna G, et al. Hipsc-cm monolayer maturation state determines drug responsiveness in high throughput pro-arrhythmia screen. Sci Rep. 2017;7:13834.
Diez-Cunado M, Wei K, Bushway PJ, Maurya MR, Perera R, Subramaniam S, et al. Mirnas that induce human cardiomyocyte proliferation converge on the Hippo pathway. Cell Rep. 2018;23:2168–74.
Woo LA, Tkachenko S, Ding M, Plowright AT, Engkvist O, Andersson H, et al. High-content phenotypic assay for proliferation of human iPSC-derived cardiomyocytes identifies L-type calcium channels as targets. J Mol Cell Cardiol. 2019;127:204–14.
Hagan R, Rex E, Woody D, Milewski M, Glaza T, Maher MP, et al. Development of phenotypic assays for identifying novel blockers of L-type calcium channels in neurons. Sci Rep. 2021;11:456.
Xu L, Sun L, Xie L, Mou S, Zhang D, Zhu J, et al. Advances in L-type calcium channel structures, functions and molecular modeling. Curr Med Chem. 2021;28:514–24.
Ye D, Tester DJ, Zhou W, Papagiannis J, Ackerman MJ. A pore-localizing cacna1c-e1115k missense mutation, identified in a patient with idiopathic QT prolongation, bradycardia, and autism spectrum disorder, converts the L-type calcium channel into a hybrid nonselective monovalent cation channel. Heart Rhythm. 2019;16:270–8.
Napolitano C, Antzelevitch C. Phenotypical manifestations of mutations in the genes encoding subunits of the cardiac voltage-dependent L-type calcium channel. Circ Res. 2011;108:607–18.
Fukuyama M, Ohno S, Wang Q, Kimura H, Makiyama T, Itoh H, et al. L-type calcium channel mutations in Japanese patients with inherited arrhythmias. Circ J. 2013;77:1799–806.
Zhang Q, Chen J, Qin Y, Wang J, Zhou L. Mutations in voltage-gated L-type calcium channel: Implications in cardiac arrhythmia. Channels. 2018;12:201–18.
Godfraind T. Discovery and development of calcium channel blockers. Front Pharmacol. 2017;8:286.
Lian X, Bao X, Zilberter M, Westman M, Fisahn A, Hsiao C, et al. Chemically defined, albumin-free human cardiomyocyte generation. Nat Methods. 2015;12:595–6.
Carpenter AE, Jones TR, Lamprecht MR, Clarke C, Kang IH, Friman O, et al. Cellprofiler: Image analysis software for identifying and quantifying cell phenotypes. Genome Biol. 2006;7:R100.
Eldridge S, Guo L, Mussio J, Furniss M, Hamre J 3rd, Davis M, et al. Examining the protective role of erbb2 modulation in human-induced pluripotent stem cell-derived cardiomyocytes. Toxicol Sci. 2014;141:547–59.
Traister A, Li M, Aafaqi S, Lu M, Arab S, Radisic M, et al. Integrin-linked kinase mediates force transduction in cardiomyocytes by modulating SERCA2a/PLN function. Nat Commun. 2014;5:4533.
Scott CW, Zhang XY, Abi-Gerges N, Lamore SD, Abassi YA, Peters MF. An impedance-based cellular assay using human iPSC-derived cardiomyocytes to quantify modulators of cardiac contractility. Toxicol Sci. 2014;142:331–8.
Salic A, Mitchison TJ. A chemical method for fast and sensitive detection of DNA synthesis in vivo. Proc Natl Acad Sci USA. 2008;105:2415–20.
Artap S, Manderfield LJ, Smith CL, Poleshko A, Aghajanian H, See K, et al. Endocardial hippo signaling regulates myocardial growth and cardiogenesis. Dev Biol. 2018;440:22–30.
Mahmoud AI, Kocabas F, Muralidhar SA, Kimura W, Koura AS, Thet S, et al. Meis1 regulates postnatal cardiomyocyte cell cycle arrest. Nature. 2013;497:249–53.
Pertea M, Pertea GM, Antonescu CM, Chang TC, Mendell JT, Salzberg SL. Stringtie enables improved reconstruction of a transcriptome from RNA-seq reads. Nat Biotechnol. 2015;33:290–5.
Robinson MD, McCarthy DJ, Smyth GK. Edger: A bioconductor package for differential expression analysis of digital gene expression data. Bioinformatics. 2010;26:139–40.
Subramanian A, Tamayo P, Mootha VK, Mukherjee S, Ebert BL, Gillette MA, et al. Gene set enrichment analysis: A knowledge-based approach for interpreting genome-wide expression profiles. Proc Natl Acad Sci USA. 2005;102:15545–50.
Wang S, Ye L, Li M, Liu J, Jiang C, Hong H, et al. Gsk-3β inhibitor chir-99021 promotes proliferation through upregulating β-catenin in neonatal atrial human cardiomyocytes. J Cardiovasc Pharmacol. 2016;68:425–32.
Chen H, Vandorpe DH, Xie X, Alper SL, Zeidel ML, Yu W. Disruption of cav1.2-mediated signaling is a pathway for ketamine-induced pathology. Nat Commun. 2020;11:4328.
Janjua N, Mayer SA. Cerebral vasospasm after subarachnoid hemorrhage. Curr Opin Crit Care. 2003;9:113–9.
Kopljar I, Hermans AN, Teisman A, Gallacher DJ, Lu HR. Impact of calcium-sensitive dyes on the beating properties and pharmacological responses of human iPS-derived cardiomyocytes using the calcium transient assay. J Pharmacol Toxicol Methods. 2018;91:80–6.
Orourke RA. Current status of calcium-channel blockers - foreword. Curr Probl Cardiol. 1994;19:641–88.
Rascol O, Clanet M, Montastruc JL. Calcium antagonists and the vestibular system: A critical review of flunarizine as an antivertigo drug. Fundam Clin Pharmacol. 1989;3:79s–87s.
Tang L, Gamal El-Din TM, Swanson TM, Pryde DC, Scheuer T, Zheng N, et al. Structural basis for inhibition of a voltage-gated Ca2+ channel by Ca2+ antagonist drugs. Nature. 2016;537:117–21.
Ji N, Middelkoop TC, Mentink RA, Betist MC, Tonegawa S, Mooijman D, et al. Feedback control of gene expression variability in the Caenorhabditis elegans Wnt pathway. Cell. 2013;155:869–80.
Nusse R, Clevers H. Wnt/beta-catenin signaling, disease, and emerging therapeutic modalities. Cell. 2017;169:985–99.
Veeman MT, Slusarski DC, Kaykas A, Louie SH, Moon RT. Zebrafish prickle, a modulator of noncanonical Wnt/Fz signaling, regulates gastrulation movements. Curr Biol. 2003;13:680–5.
Daskalopoulos EP, Blankesteijn WM. Effect of interventions in Wnt signaling on healing of cardiac injury: A systematic review. Cells 2021;10:207.
Liang D, Wu Y, Zhou L, Chen Y, Liu H, Xie D, et al. Lrp5 controls cardiac qt interval by modulating the metabolic homeostasis of L-type calcium channel. Int J Cardiol. 2019;275:120–8.
Nguyen NUN, Canseco DC, Xiao F, Nakada Y, Li S, Lam NT, et al. A calcineurin-hoxb13 axis regulates growth mode of mammalian cardiomyocytes. Nature. 2020;582:271–6.
Paul MA, Wainwright CL, Hector EE, Ryberg E, Leslie SJ, Walsh SK. Short-term oral administration of the porcupine inhibitor, wnt-c59, improves the structural and functional features of experimental HFPEF. Pharmacol Res Perspect. 2025;13:e70054.
Blyszczuk P, Muller-Edenborn B, Valenta T, Osto E, Stellato M, Behnke S, et al. Transforming growth factor-beta-dependent Wnt secretion controls myofibroblast formation and myocardial fibrosis progression in experimental autoimmune myocarditis. Eur Heart J. 2017;38:1413–25.
Jang J, Song J, Sim I, Kwon YV, Yoon Y. Wnt-signaling inhibitor wnt-c59 suppresses the cytokine upregulation in multiple organs of lipopolysaccharide-induced endotoxemic mice via reducing the interaction between beta-catenin and NF-kappaB. Int J Mol Sci 2021;22:6249.
Zhao Z, Liu H, Li Y, Tian J, Deng S. Wnt-c59 attenuates pressure overload-induced cardiac hypertrophy via interruption of wnt pathway. Med Sci Monit. 2020;26:e923025.
Steinhart Z, Angers S. Wnt signaling in development and tissue homeostasis. Development. 2018;145:dev146589.
Qyang Y, Martin-Puig S, Chiravuri M, Chen S, Xu H, Bu L, et al. The renewal and differentiation of isl1+ cardiovascular progenitors are controlled by a Wnt/beta-catenin pathway. Cell Stem Cell. 2007;1:165–79.
Kwon C, Arnold J, Hsiao EC, Taketo MM, Conklin BR, Srivastava D. Canonical Wnt signaling is a positive regulator of mammalian cardiac progenitors. Proc Natl Acad Sci USA. 2007;104:10894–9.
Quaife-Ryan GA, Mills RJ, Lavers G, Voges HK, Vivien CJ, Elliott DA, et al. Beta-catenin drives distinct transcriptional networks in proliferative and nonproliferative cardiomyocytes. Development 2020;147:dev193417.
Fan Y, Ho BX, Pang JKS, Pek NMQ, Hor JH, Ng SY, et al. Wnt/β-catenin-mediated signaling re-activates proliferation of matured cardiomyocytes. Stem Cell Res Ther. 2018;9:338.
Tseng AS, Engel FB, Keating MT. The GSK-3 inhibitor bio promotes proliferation in mammalian cardiomyocytes. Chem Biol. 2006;13:957–63.
Duan J, Gherghe C, Liu D, Hamlett E, Srikantha L, Rodgers L, et al. Wnt1/betacatenin injury response activates the epicardium and cardiac fibroblasts to promote cardiac repair. EMBO J. 2012;31:429–42.
Pereira C, Schaer DJ, Bachli EB, Kurrer MO, Schoedon G. Wnt5a/camkii signaling contributes to the inflammatory response of macrophages and is a target for the antiinflammatory action of activated protein c and interleukin-10. Arterioscler Thromb Vasc Biol. 2008;28:504–10.
Barandon L, Couffinhal T, Ezan J, Dufourcq P, Costet P, Alzieu P, et al. Reduction of infarct size and prevention of cardiac rupture in transgenic mice overexpressing frza. Circulation. 2003;108:2282–9.
Min JK, Park H, Choi HJ, Kim Y, Pyun BJ, Agrawal V, et al. The Wnt antagonist dickkopf2 promotes angiogenesis in rodent and human endothelial cells. J Clin Invest. 2011;121:1882–93.
Xiang FL, Fang M, Yutzey KE. Loss of beta-catenin in resident cardiac fibroblasts attenuates fibrosis induced by pressure overload in mice. Nat Commun. 2017;8:712.
Moon J, Zhou H, Zhang LS, Tan W, Liu Y, Zhang S, et al. Blockade to pathological remodeling of infarcted heart tissue using a porcupine antagonist. Proc Natl Acad Sci USA. 2017;114:1649–54.
Wo D, Peng J, Ren DN, Qiu L, Chen J, Zhu Y, et al. Opposing roles of wnt inhibitors igfbp-4 and dkk1 in cardiac ischemia by differential targeting of lrp5/6 and beta-catenin. Circulation. 2016;134:1991–2007.
Fu WB, Wang WE, Zeng CY. Wnt signaling pathways in myocardial infarction and the therapeutic effects of wnt pathway inhibitors. Acta Pharmacol Sin. 2019;40:9–12.
Hou C, Liu Q, Zhang H, Wang W, Wang B, Cui X, et al. Nimodipine attenuates early brain injury by protecting the glymphatic system after subarachnoid hemorrhage in mice. Neurochem Res. 2022;47:701–12.
Korte N, Barkaway A, Wells J, Freitas F, Sethi H, Andrews SP, et al. Inhibiting Ca2+ channels in alzheimer’s disease model mice relaxes pericytes, improves cerebral blood flow, and reduces immune cell stalling and hypoxia. Nat Neurosci. 2024;27:2086–100.
Ansari MA, Iqubal A, Ekbbal R, Haque SE. Effects of nimodipine, vinpocetine and their combination on isoproterenol-induced myocardial infarction in rats. Biomed Pharmacother. 2019;109:1372–80.
Kaur AH, Singh J, Srivastava RK, Mathur SK. Effect of nitrendipine, nimodipine and nisoldipine on experimentally induced myocardial infarction in rats. Indian J Exp Biol. 1995;33:420–3.
Gregorieff A, Clevers H. Wnt signaling in the intestinal epithelium: from endoderm to cancer. Genes Dev. 2005;19:877–90.
Xie S, Fu W, Yu G, Hu X, Lai KS, Peng X, et al. Discovering small molecules as Wnt inhibitors that promote heart regeneration and injury repair. J Mol Cell Biol. 2020;12:42–54.
Peng X, Lai KS, She P, Kang J, Wang T, Li G, et al. Induction of Wnt signaling antagonists and p21-activated kinase enhances cardiomyocyte proliferation during zebrafish heart regeneration. J Mol Cell Biol. 2021;13:41–58.
Wu Y, Zhou L, Liu H, Duan R, Zhou H, Zhang F, et al. Lrp6 downregulation promotes cardiomyocyte proliferation and heart regeneration. Cell Res. 2021;31:450–62.
Hallhuber M, Burkard N, Wu R, Buch MH, Engelhardt S, Hein L, et al. Inhibition of nuclear import of calcineurin prevents myocardial hypertrophy. Circ Res. 2006;99:626–35.
Martinez-Martinez S, Lozano-Vidal N, Lopez-Maderuelo MD, Jimenez-Borreguero LJ, Armesilla AL, Redondo JM. Cardiomyocyte calcineurin is required for the onset and progression of cardiac hypertrophy and fibrosis in adult mice. FEBS J. 2019;286:46–65.
Molkentin JD. Calcineurin-NFAT signaling regulates the cardiac hypertrophic response in coordination with the mapks. Cardiovasc Res. 2004;63:467–75.
Acknowledgements
This work was funded by Key Research and Development Program, Ministry of Science and Technology of China (2017YFA0105600, 2018YFA0800104), National Natural Science Foundation of China (31771613, 32070823, 92168205, 82400339, 82470404), Natural Science Foundation of Shanghai (24SF1900600, 24ZR1457600), Chinese Society of Cardiology Foundation (CSCF2023B02), Fundamental Research Funds for the Central Universities (22120200411, 22120210073, 22120250374) and Tongji University Medicine-X Interdisciplinary Research Initiative (2025-0553-ZD-10). The graphical abstract was created with BioRender.com. The authors thank the Peak Disciplines (Type IV) of Institutions of Higher Learning in Shanghai, and the Frontier Science Research Center for Stem Cells, Ministry of Education, for their support. And we thank Yang Dong and Mei-hui Zhang for their technical assistance, Zhen Zhang and Tao P Zhong for their helpful discussions.
Author information
Authors and Affiliations
Contributions
KW, MYF, and YST conceived the study. DCX, GHG, and KW supervised the experiments. MYF, SY, and YST performed a high-content screen. MYF, YST, and RRH performed experiments on hPSC-CMs, NRVCs and mice. ZHOY, SQH, RJ, and MY performed NRVC experiments. BLL and YST performed experiments on mice. HJZ performed bioinformatics analysis. KW, MYF, and YST wrote the manuscript with input from all authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Feng, My., Tang, Ys., Yao, S. et al. Inhibiting L-type calcium channel promotes cardiomyocyte proliferation through activating the canonical Wnt signaling pathway. Acta Pharmacol Sin 47, 917–931 (2026). https://doi.org/10.1038/s41401-025-01704-6
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41401-025-01704-6