Abstract
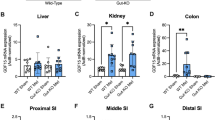
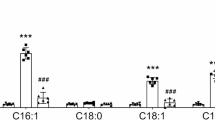
Metformin, the most prescribed drug for the treatment of type 2 diabetes mellitus, increases the circulating levels of the metabolic regulator growth differentiation factor 15 (GDF15) via transcriptional regulation, with the kidneys being responsible for this increase. Since peroxisome proliferator-activated receptor (PPAR)β/δ agonists mimic many of the effects of metformin, including the rise in circulating GDF15 levels, we herein investigated whether the metformin-mediated antidiabetic effects and GDF15 upregulation were dependent on this nuclear receptor. Male Ppard−/− and wild-type (WT) mice received a western-type high-fat diet (HFD) for 12 weeks and were treated with metformin (200 mg ·kg−1 ·d−1, i.g.) in the last 3 weeks. At the end of the treatment, the mice were sacrificed, and the skeletal muscle, kidney, and liver samples were collected for analyses. We showed that metformin treatment ameliorated glucose intolerance and increased hepatic and circulating GDF15 levels in WT mice, but not in Ppard−/− mice fed a HFD. In the kidneys, metformin treatment increased the expression levels of phosphorylated AMPK and GDF15 in the WT mice, which was abolished in the Ppard−/− mice. Both β-arrestin 1 and proprotein convertase subtilisin/kexin type 6 (PCSK6) are involved in the posttranslational maturation of GDF15. Likewise, metformin treatment increased the levels of β-arrestin 1 and PCSK6 in the kidneys of WT mice, but not Ppard−/− mice. Furthermore, treatment of mice with a PPARβ/δ activator, GW501516 (3 mg· kg−1 ·d−1, i.g., for 7 days), increased the levels of these proteins in the kidneys and liver. In contrast, a PPARβ/δ antagonist GSK0660 (50 µM) prevented the increase in GDF15, β-arrestin 1, and PCSK6 levels caused by metformin in cultured podocytes. Collectively, these data uncover a regulatory axis wherein metformin, via PPARβ/δ, orchestrates glucose tolerance, AMPK activity, and GDF15 maturation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Patel S, Alvarez-Guaita A, Melvin A, Rimmington D, Dattilo A, Miedzybrodzka EL, et al. GDF15 provides an endocrine signal of nutritional stress in mice and humans. Cell Metab. 2019;29:707–18.e708.
Breit SN, Brown DA, Tsai VW. The GDF15–GFRAL pathway in health and metabolic disease: friend or foe?. Annu Rev Physiol. 2021;83:127–51.
Tsai VW, Zhang HP, Manandhar R, Lee-Ng KKM, Lebhar H, Marquis CP, et al. Treatment with the TGF-β superfamily cytokine MIC-1/GDF15 reduces the adiposity and corrects the metabolic dysfunction of mice with diet-induced obesity. Int J Obes. 2018;42:561–71.
Lockhart SM, Saudek V, O’Rahilly S. GDF15: a hormone conveying somatic distress to the brain. Endocr Rev. 2020;41:4.
Tsai VWW, Husaini Y, Sainsbury A, Brown DA, Breit SN. The MIC-1/GDF15–GFRAL pathway in energy homeostasis: implications for obesity, cachexia, and other associated diseases. Cell Metab. 2018;28:353–68.
Aguilar-Recarte D, Barroso E, Palomer X, Wahli W, Vazquez-Carrera M. Knocking on GDF15’s door for the treatment of type 2 diabetes mellitus. Trends Endocrinol Metab. 2022;33:741–54.
Kim-Muller JY, Song L, LaCarubba PB, Pashos E, Li X, Rinaldi A, et al. GDF15 neutralization restores muscle function and physical performance in a mouse model of cancer cachexia. Cell Rep. 2023;42:111947.
Tsui KH, Hsu SY, Chung LC, Lin YH, Feng TH, Lee TY, et al. Growth differentiation factor-15: a p53- and demethylation-upregulating gene represses cell proliferation, invasion, and tumorigenesis in bladder carcinoma cells. Sci Rep. 2015;5:12870.
Baek SJ, Kim JS, Nixon JB, DiAugustine RP, Eling TE. Expression of NAG-1, a transforming growth factor-β superfamily member, by troglitazone requires the early growth response gene EGR-1. J Biol Chem. 2004;279:6883–92.
Townsend LK, Weber AJ, Day EA, Shamshoum H, Shaw SJ, Perry CGR, et al. AMPK mediates energetic stress-induced liver GDF15. FASEB J. 2021;35:e21218.
Aguilar-Recarte D, Barroso E, Guma A, Pizarro-Delgado J, Pena L, Ruart M, et al. GDF15 mediates the metabolic effects of PPARβ/δ by activating AMPK. Cell Rep. 2021;36:109501.
Zhang Z, Xu X, Tian W, Jiang R, Lu Y, Sun Q, et al. ARRB1 inhibits non-alcoholic steatohepatitis progression by promoting GDF15 maturation. J Hepatol. 2020;72:976–89.
Baek SJ, Eling T. Growth differentiation factor 15 (GDF15): a survival protein with therapeutic potential in metabolic diseases. Pharmacol Ther. 2019;198:46–58.
Li JJ, Liu J, Lupino K, Liu X, Zhang L, Pei L. Growth differentiation factor 15 maturation requires proteolytic cleavage by PCSK3, -5, and -6. Mol Cell Biol. 2018;38:21.
Emmerson PJ, Wang F, Du Y, Liu Q, Pickard RT, Gonciarz MD, et al. The metabolic effects of GDF15 are mediated by the orphan receptor GFRAL. Nat Med. 2017;23:1215–9.
Mullican SE, Lin-Schmidt X, Chin CN, Chavez JA, Furman JL, Armstrong AA, et al. GFRAL is the receptor for GDF15 and the ligand promotes weight loss in mice and nonhuman primates. Nat Med. 2017;23:1150–7.
Hsu JY, Crawley S, Chen M, Ayupova DA, Lindhout DA, Higbee J, et al. Non-homeostatic body weight regulation through a brainstem-restricted receptor for GDF15. Nature. 2017;550:255–9.
Yang L, Chang CC, Sun Z, Madsen D, Zhu H, Padkjaer SB, et al. GFRAL is the receptor for GDF15 and is required for the anti-obesity effects of the ligand. Nat Med. 2017;23:1158–66.
Mazagova M, Buikema H, van Buiten A, Duin M, Goris M, Sandovici M, et al. Genetic deletion of growth differentiation factor 15 augments renal damage in both type 1 and type 2 models of diabetes. Am J Physiol Ren Physiol. 2013;305:F1249–56.
Gerstein HC, Pare G, Hess S, Ford RJ, Sjaarda J, Raman K, et al. Growth differentiation factor 15 as a novel biomarker for metformin. Diabetes Care. 2017;40:280–3.
Coll AP, Chen M, Taskar P, Rimmington D, Patel S, Tadross JA, et al. GDF15 mediates the effects of metformin on body weight and energy balance. Nature. 2020;578:444–8.
Day EA, Ford RJ, Smith BK, Mohammadi-Shemirani P, Morrow MR, Gutgesell RM, et al. Metformin-induced increases in GDF15 are important for suppressing appetite and promoting weight loss. Nat Metab. 2019;1:1202–8.
Klein AB, Nicolaisen TS, Johann K, Fritzen AM, Mathiesen CV, Gil C, et al. The GDF15–GFRAL pathway is dispensable for the effects of metformin on energy balance. Cell Rep. 2022;40:111258.
Aguilar-Recarte D, Barroso E, Zhang M, Rada P, Pizarro-Delgado J, Pena L, et al. A positive feedback loop between AMPK and GDF15 promotes metformin antidiabetic effects. Pharmacol Res. 2023;187:106578.
Kincaid JWR, Rimmington D, Tadross JA, Cimino I, Zvetkova I, Kaser A, et al. The gastrointestinal tract is a major source of the acute metformin-stimulated rise in GDF15. Sci Rep. 2024;14:1899.
Zhang SY, Bruce K, Danaei Z, Li RJW, Barros DR, Kuah R, et al. Metformin triggers a kidney GDF15-dependent area postrema axis to regulate food intake and body weight. Cell Metab. 2023;35:875–86.e875.
Lee CH, Olson P, Hevener A, Mehl I, Chong LW, Olefsky JM, et al. PPARδ regulates glucose metabolism and insulin sensitivity. Proc Natl Acad Sci USA. 2006;103:3444–9.
Saleem MA, O’Hare MJ, Reiser J, Coward RJ, Inward CD, Farren T, et al. A conditionally immortalized human podocyte cell line demonstrating nephrin and podocin expression. J Am Soc Nephrol. 2002;13:630–8.
Santamaria B, Marquez E, Lay A, Carew RM, Gonzalez-Rodriguez A, Welsh GI, et al. IRS2 and PTEN are key molecules in controlling insulin sensitivity in podocytes. Biochim Biophys Acta. 2015;1853:3224–33.
Jurado-Aguilar J, Barroso E, Bernard M, Zhang M, Peyman M, Rada P, et al. GDF15 activates AMPK and inhibits gluconeogenesis and fibrosis in the liver by attenuating the TGF-β1/SMAD3 pathway. Metabolism. 2024;152:155772.
Chung HK, Ryu D, Kim KS, Chang JY, Kim YK, Yi HS, et al. Growth differentiation factor 15 is a myomitokine governing systemic energy homeostasis. J Cell Biol. 2017;216:149–65.
Koh JH, Hancock CR, Terada S, Higashida K, Holloszy JO, Han DH. PPARβ is essential for maintaining normal levels of PGC-1α and mitochondria and for the increase in muscle mitochondria induced by exercise. Cell Metab. 2017;25:1176–85 e1175.
Cameron A, Appel J, Houghten RA, Lindberg I. Polyarginines are potent furin inhibitors. J Biol Chem. 2000;275:36741–9.
Chrysovergis K, Wang X, Kosak J, Lee SH, Kim JS, Foley JF, et al. NAG-1/GDF15 prevents obesity by increasing thermogenesis, lipolysis, and oxidative metabolism. Int J Obes. 2014;38:1555–64.
Kim HJ, Zhang XH, Park EY, Shin KH, Choi SH, Chun BG, et al. Metformin decreases meal size and number and increases c-Fos expression in the nucleus tractus solitarius of obese mice. Physiol Behav. 2013;110–11:213–20.
Souza-Mello V, Gregorio BM, Cardoso-de-Lemos FS, de Carvalho L, Aguila MB, Mandarim-de-Lacerda CA. Comparative effects of telmisartan, sitagliptin and metformin alone or in combination on obesity, insulin resistance, and liver and pancreas remodeling in C57BL/6 mice fed on a very high-fat diet. Clin Sci. 2010;119:239–50.
Kincaid JWR, Coll AP. Metformin and GDF15: where are we now?. Nat Rev Endocrinol. 2023;19:6–7.
Ost M, Igual Gil C, Coleman V, Keipert S, Efstathiou S, Vidic V, et al. Muscle-derived GDF15 drives diurnal anorexia and systemic metabolic remodeling during mitochondrial stress. EMBO Rep. 2020;21:e48804.
Cao J, Meng S, Chang E, Beckwith-Fickas K, Xiong L, Cole RN, et al. Low concentrations of metformin suppress glucose production in hepatocytes through AMP-activated protein kinase (AMPK). J Biol Chem. 2014;289:20435–46.
Ma T, Tian X, Zhang B, Li M, Wang Y, Yang C, et al. Low-dose metformin targets the lysosomal AMPK pathway through PEN2. Nature. 2022;603:159–65.
Meng RS, Pei ZH, Yin R, Zhang CX, Chen BL, Zhang Y, et al. AMP-activated protein kinase inhibits cardiac hypertrophy through reactivating peroxisome proliferator-activated receptor-α signaling pathway. Eur J Pharmacol. 2009;620:63–70.
Townsend LK, Medak K, Weber AJ, Dibe H, Shamshoum H, Wright DC. CHOP is dispensable for exercise-induced increases in GDF15. J Appl Physiol. 2022;132:413–22.
Yang M, Darwish T, Larraufie P, Rimmington D, Cimino I, Goldspink DA, et al. Inhibition of mitochondrial function by metformin increases glucose uptake, glycolysis and GDF15 release from intestinal cells. Sci Rep. 2021;11:2529.
Tobar N, Rocha GZ, Santos A, Guadagnini D, Assalin HB, Camargo JA, et al. Metformin acts in the gut and induces gut–liver crosstalk. Proc Natl Acad Sci USA. 2023;120:e2211933120.
Bailey CJ, Mynett KJ, Page T. Importance of the intestine as a site of metformin-stimulated glucose utilization. Br J Pharmacol. 1994;112:671–5.
Penicaud L, Hitier Y, Ferre P, Girard J. Hypoglycaemic effect of metformin in genetically obese (fa/fa) rats results from an increased utilization of blood glucose by intestine. Biochem J. 1989;262:881–5.
Wang HH, Lin SH, Hung SY, Chiou YY, Hsu WC, Chang CM, et al. Renal protective effect of metformin in type 2 diabetes patients. J Clin Endocrinol Metab. 2024. https://doi.org/10.1210/clinem/dgae123.
Borges CM, Fujihara CK, Malheiros D, de Avila VF, Formigari GP, Lopes de Faria JB. Metformin arrests the progression of established kidney disease in the subtotal nephrectomy model of chronic kidney disease. Am J Physiol Ren Physiol. 2020;318:F1229–36.
Rashid M, Shahzad M, Mahmood S, Khan K. Variability in the therapeutic response of metformin treatment in patients with type 2 diabetes mellitus. Pak J Med Sci. 2019;35:71–6.
Zhou K, Donnelly L, Yang J, Li M, Deshmukh H, Van Zuydam N, et al. Heritability of variation in glycaemic response to metformin: a genome-wide complex trait analysis. Lancet Diabetes Endocrinol. 2014;2:481–7.
Acknowledgements
We are indebted to the Biobank core facility of the Institut d’Investigacions Biomèdiques August Pi i Sunyer (IDIBAPS) for the technical help. We would like to thank the Language Services of the University of Barcelona for revising the manuscript. EB is a Serra Hunter fellow. This study was supported by the grants PID2024-155124OB-I00 and PID2021-122116OB-I00 (MVC and XP), PID2021-122766OB-I00 (AMV), and PRE2019-089839 funded by MICIU/AEI/10.13039/501100011033 and “ERDF, A Way of Making Europe”. CIBER de Diabetes y Enfermedades Metabólicas Asociadas (CIBERDEM) is a Carlos III Health Institute project. Support was also received from the CERCA Programme/Generalitat de Catalunya.
Author information
Authors and Affiliations
Contributions
JJA, EB, PR, MP, AR, and MVC performed the experiments and collected the data. JJA, JB, AMV, WW, XP, and MVC analyzed and interpreted the data. WW and XP drafted and revised the manuscript. MVC designed the experiments, interpreted the results, and wrote the manuscript. All authors have read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Jurado-Aguilar, J., Barroso, E., Rada, P. et al. PPARβ/δ contributes to the antidiabetic effect and the increase in GDF15 caused by metformin. Acta Pharmacol Sin (2026). https://doi.org/10.1038/s41401-025-01705-5
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41401-025-01705-5