Abstract
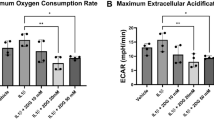
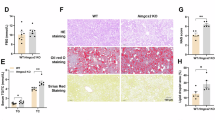
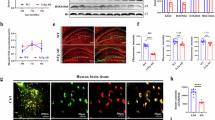
Cerebral edema is a severe complication following ischemic stroke. Recent studies have highlighted the crucial role of the glymphatic system (GS) in the clearance of water and macromolecules. GS dysfunction involving the disorders of AQP4 polarization may be crucial in the pathophysiology of cerebral edema. β-Hydroxybutyrate (BHB), the main component of the ketone body, has been shown to alleviate neurological deficits by restoring GS function in subarachnoid hemorrhage models and to reduce Aβ deposition in Alzheimer’s disease models. In this study we investigated the effects of BHB on cerebral edema following ischemic stroke and its mechanisms. The mice were fed a ketogenic diet (KD) or a normal diet for 4 weeks before transient middle cerebral artery occlusion (MCAO). Alternatively, the mice received BHB (5 g·kg−1·d−1) or vehicle post-MCAO. By using brain section analysis, transcranial macroimaging, two-photon in vivo imaging and MRI, we demonstrated that both KD and BHB treatment significantly enhanced GS function under normal and MCAO conditions. BHB reduced cerebral edema and infarct volume post-MCAO. Notably, delayed BHB treatment initiated 10 h post-MCAO still improved GS function, but did not influence infarct volume. Furthermore, we revealed that BHB increased α1-syntrophin expression and H3K27ac levels in α1-syntrophin (Snta1) enhancer, restoring AQP4 polarization. In addition, BHB also reduced HDAC3 expression and elevated p300 expression. These results suggest that a KD and BHB treatment enhance GS function in mice and that BHB also mitigates brain edema after MCAO. The potentiation of GS function by BHB is likely mediated by the inhibition of HDAC3 activity and the increase in p300 activity, which upregulate α1-syntrophin expression and restore AQP4 polarization.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout









Similar content being viewed by others
Data availability
The datasets used and/or analyzed during this study are available from the corresponding authors upon reasonable request.
References
GBD 2019 Stroke Collaborators. Global, regional, and national burden of stroke and its risk factors, 1990-2019: a systematic analysis for the Global Burden of Disease Study 2019. Lancet Neurol. 2021;20:795–820.
Ma Q, Li R, Wang L, Yin P, Wang Y, Yan C, et al. Temporal trend and attributable risk factors of stroke burden in China, 1990-2019: an analysis for the Global Burden of Disease Study 2019. Lancet Public Health. 2021;6:e897–906.
Rabinstein AA. Ischaemic brain oedema remains a major unmet need. Lancet Neurol. 2024;23:1171–73.
Battey TWK, Karki M, Singhal AB, Wu O, Sadaghiani S, Campbell BCV, et al. Brain edema predicts outcome after nonlacunar ischemic stroke. Stroke. 2014;45:3643–48.
Dower A, Mulcahy M, Maharaj M, Chen H, Lim CED, Li Y, et al. Surgical decompression for malignant cerebral oedema after ischaemic stroke. Cochrane Database Syst Rev. 2022;11:CD014989.
Mohney N, Alkhatib O, Koch S, O’Phelan K, Merenda A. What is the role of hyperosmolar therapy in hemispheric stroke patients?. Neurocrit Care. 2020;32:609–19.
Thaysen M, Westi E, Clarkson AN, Wellendorph P, Kristensen M. Rodent ischemic stroke models and their relevance in preclinical research. Neuroprotection. 2024;2:296–309.
Bakreen A, Jolkkonen J. The potential of a combined cell-based therapy and rehabilitation approach for stroke recovery. Neuroprotection. 2023;1:117–29.
Iliff JJ, Chen MJ, Plog BA, Zeppenfeld DM, Soltero M, Yang L, et al. Impairment of glymphatic pathway function promotes tau pathology after traumatic brain injury. J Neurosci. 2014;34:16180–93.
Iliff JJ, Wang M, Liao Y, Plogg BA, Peng W, Gundersen GA, et al. A paravascular pathway facilitates CSF flow through the brain parenchyma and the clearance of interstitial solutes, including amyloid beta. Sci Transl Med. 2012;4:147ra111.
Liu K, Zhu J, Chang Y, Lin Z, Shi Z, Li X, et al. Attenuation of cerebral edema facilitates recovery of glymphatic system function after status epilepticus. JCI Insight. 2021;6:e151835.
Zhu J, Mo J, Liu K, Chen Q, Li Z, He Y, et al. Glymphatic system impairment contributes to the formation of brain edema after ischemic stroke. Stroke. 2024;55:1393–404.
Hablitz LM, Nedergaard M. The glymphatic system. Curr Biol. 2021;31:R1371–75.
Harrison IF, Ismail O, Machhada A, Colgan N, Ohene Y, Nahavandi P, et al. Impaired glymphatic function and clearance of tau in an Alzheimer’s disease model. Brain. 2020;143:2576–93.
Kress BT, Iliff JJ, Xia M, Wang M, Wei HS, Zeppenfeld D, et al. Impairment of paravascular clearance pathways in the aging brain. Ann Neurol. 2014;76:845–61.
Zhou X, Li Y, Lenahan C, Ou Y, Wang M, He Y. Glymphatic system in the central nervous system, a novel therapeutic direction against brain edema after stroke. Front Aging Neurosci. 2021;13:698036.
Olson CA, Iniguez AJ, Yang GE, Fang P, Pronovost GN, Jameson KG, et al. Alterations in the gut microbiota contribute to cognitive impairment induced by the ketogenic diet and hypoxia. Cell Host Microbe. 2021;29:1378–92.
Van der Auwera I, Wera S, Van Leuven F, Henderson ST. A ketogenic diet reduces amyloid beta 40 and 42 in a mouse model of Alzheimer’s disease. Nutr Metab. 2005;2:28.
Hardy J. A hundred years of Alzheimer’s disease research. Neuron. 2006;52:3–13.
Tarasoff-Conway JM, Carare RO, Osorio RS, Glodzik L, Butler T, Fieremans E, et al. Clearance systems in the brain-implications for Alzheimer disease. Nat Rev Neurol. 2015;11:457–70.
Dedkova EN, Blatter LA. Role of beta-hydroxybutyrate, its polymer poly-beta-hydroxybutyrate and inorganic polyphosphate in mammalian health and disease. Front Physiol. 2014;5:260.
Tan X, Li X, Li R, Meng W, Xie Z, Li J, et al. beta-hydroxybutyrate alleviates neurological deficits by restoring glymphatic and inflammation after subarachnoid hemorrhage in mice. Exp Neurol. 2024;378:114819.
Wu Y, Gong Y, Luan Y, Li Y, Liu J, Yue Z, et al. BHBA treatment improves cognitive function by targeting pleiotropic mechanisms in transgenic mouse model of Alzheimer’s disease. FASEB J. 2020;34:1412–29.
Wang F, Xu C, Su C, Li J, Lin J. beta-Hydroxybutyrate attenuates painful diabetic neuropathy via restoration of the Aquaporin-4 polarity in the spinal glymphatic system. Front Neurosci. 2022;16:926128.
Nagelhus EA, Ottersen OP. Physiological roles of aquaporin-4 in brain. Physiol Rev. 2013;93:1543–62.
Amiry-Moghaddam M, Otsuka T, Hurn PD, Traystman RJ, Haug F, Froehner SC, et al. An alpha-syntrophin-dependent pool of AQP4 in astroglial end-feet confers bidirectional water flow between blood and brain. Proc Natl Acad Sci USA. 2003;100:2106–11.
Mestre H, Hablitz LM, Xavier AL, Feng W, Zou W, Pu T, et al. Aquaporin-4-dependent glymphatic solute transport in the rodent brain. Elife. 2018;7:e40070.
Landgrave-Gomez J, Mercado-Gomez OF, Vazquez-Garcia M, Rodriguez-Molina V, Cordova-Davalos L, Arriaga-Avila V, et al. Anticonvulsant effect of time-restricted feeding in a pilocarpine-induced seizure model: metabolic and epigenetic implications. Front Cell Neurosci. 2016;10:7.
Luo W, Yu Y, Wang H, Liu K, Wang Y, Huang M, et al. Up-regulation of MMP-2 by histone H3K9 beta-hydroxybutyrylation to antagonize glomerulosclerosis in diabetic rat. Acta Diabetol. 2020;57:1501–9.
Zhang J, Li X, Ren Y, Zhao Y, Xing A, Jiang C, et al. Intermittent fasting alleviates the increase of lipoprotein lipase expression in brain of a mouse model of Alzheimer’s disease: possibly mediated by beta-hydroxybutyrate. Front Cell Neurosci. 2018;12:1.
Zhu J, Li Z, Ji Z, Wu Y, He Y, Liu K, et al. Glycocalyx is critical for blood-brain barrier integrity by suppressing caveolin1-dependent endothelial transcytosis following ischemic stroke. Brain Pathol. 2022;32:e13006.
Chen W, Huang P, Zeng H, Lin J, Shi Z, Yao X. Cocaine-induced structural and functional impairments of the glymphatic pathway in mice. Brain Behav Immun. 2020;88:97–104.
Smith AJ, Yao X, Dix JA, Jin B, Verkman AS. Test of the ‘glymphatic’ hypothesis demonstrates diffusive and aquaporin-4-independent solute transport in rodent brain parenchym. Elife. 2017;6:e27679.
Ineichen BV, Schnell L, Gullo M, Kaiser J, Schneider MP, Mosberger AC, et al. Direct, long-term intrathecal application of therapeutics to the rodent CNS. Nat Protoc. 2017;12:104–31.
He X, Liu D, Zhang Q, Liang F, Dai G, Zeng J, et al. Voluntary exercise promotes glymphatic clearance of amyloid beta and reduces the activation of astrocytes and microglia in aged mice. Front Mol Neurosci. 2017;10:144.
Moore JE, Purcaro MJ, Pratt HE, Epstein CB, Shoresh N, Adrian J, et al. Expanded encyclopaedias of DNA elements in the human and mouse genomes. Nature. 2020;583:699–710.
Ulamek-Koziol M, Czuczwar SJ, Januszewski S, Pluta R. Ketogenic diet and epilepsy. Nutrients. 2019;11:2510.
Lin C, Wang S, Xie J, Zhu J, Xu J, Liu K, et al. Ketogenic diet and beta-Hydroxybutyrate alleviate ischemic brain injury in mice via an IRAKM-dependent pathway. Eur J Pharmacol. 2023;955:175933.
Lin C, Wang S, Wei X, Liu K, Peng Y, Yu M, et al. β-Hydroxybutyrate inhibits FOXO3a by histone H3K9 β-Hydroxybutyrylation to ameliorate stroke-related sarcopenia. J Funct Foods. 2024;120:106365.
Newman JC, Verdin E. beta-Hydroxybutyrate: a signaling metabolite. Annu Rev Nutr. 2017;37:51–76.
Shimazu T, Hirschey MD, Newman J, He W, Shirakawa K, Le Moan N, et al. Suppression of oxidative stress by beta-hydroxybutyrate, an endogenous histone deacetylase inhibitor. Science. 2013;339:211–14.
Falkenberg KJ, Johnstone RW. Histone deacetylases and their inhibitors in cancer, neurological diseases and immune disorders. Nat Rev Drug Discov. 2014;13:673–91.
Graff J, Rei D, Guan J, Wang W, Seo J, Hennig KM, et al. An epigenetic blockade of cognitive functions in the neurodegenerating brain. Nature. 2012;483:222–26.
Lu H, Ashiqueali R, Lin CI, Walchale A, Clendaniel V, Matheson R, et al. Histone deacetylase 3 inhibition decreases cerebral edema and protects the blood-brain barrier after stroke. Mol Neurobiol. 2023;60:235–46.
Creyghton MP, Cheng AW, Welstead GG, Kooistra T, Carey BW, Steine EJ, et al. Histone H3K27ac separates active from poised enhancers and predicts developmental state. Proc Natl Acad Sci USA. 2010;107:21931–36.
Wang M, Chen Z, Zhang Y. CBP/p300 and HDAC activities regulate H3K27 acetylation dynamics and zygotic genome activation in mouse preimplantation embryos. EMBO J. 2022;41:e112012.
Narita T, Ito S, Higashijima Y, Chu WK, Neumann K, Walter J, et al. Enhancers are activated by p300/CBP activity-dependent PIC assembly, RNAPII recruitment, and pause release. Mol Cell. 2021;81:2166–82.
Wang Z, Zang C, Rosenfeld JA, Schones DE, Barski A, Cuddapah S, et al. Combinatorial patterns of histone acetylations and methylations in the human genome. Nat Genet. 2008;40:897–903.
Ishida K, Yamada K, Nishiyama R, Hashimoto T, Nishida I, Abe Y, et al. Glymphatic system clears extracellular tau and protects from tau aggregation and neurodegeneration. J Exp Med. 2022;219:e20211275.
Zhang H, Wang J, Zhang S, Yan D, Dong Y, Zhang P, et al. Aquaporin 4 and its isoforms regulation ameliorate AQP4 Mis-localization-induced glymphatic dysfunction in ischemic stroke. J Adv Res. 2025. https://doi.org/10.1016/j.jare.2025.05.022. Online ahead of print.
Si X, Dai S, Fang Y, Tang J, Wang Z, Li Y, et al. Matrix metalloproteinase-9 inhibition prevents aquaporin-4 depolarization-mediated glymphatic dysfunction in Parkinson’s disease. J Adv Res. 2024;56:125–36.
Moelo C, Quillevere A, Le Roy L, Timsit S. (S)-roscovitine, a CDK inhibitor, decreases cerebral edema and modulates AQP4 and alpha1-syntrophin interaction on a pre-clinical model of acute ischemic stroke. Glia. 2024;72:322–37.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (82171345 & 82371467), the Guangdong Basic and Applied Basic Research Foundation (2024A1515012553 & 2023A1515110506), the China Postdoctoral Science Foundation (2024M751319), the Postdoctoral Fellowship Program of CPSF (GZC20231066), and the President Foundation of Nanfang Hospital, Southern Medical University (2023A005).
Author information
Authors and Affiliations
Contributions
MJY, RQX, JXX, and YCL performed the animal experiments and analysis. MJY, JWW, HPZ, and JXX performed the molecular biology experiments and analyzed the results. MJY prepared the manuscript with input from all the authors. KBH, SYP, YC, and GYY revised the manuscript. KBH and SYP conceived and designed the studies. KBH, SYP, and YC provided the funding. All the authors read and approved the final version of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yu, Mj., Xiong, Rq., Wu, Jw. et al. β-Hydroxybutyrate improves glymphatic system function and alleviates cerebral edema in mice after ischemic stroke. Acta Pharmacol Sin 47, 903–916 (2026). https://doi.org/10.1038/s41401-025-01706-4
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41401-025-01706-4