Abstract
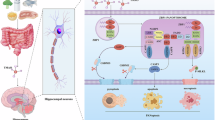
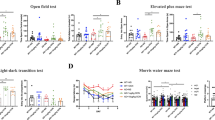
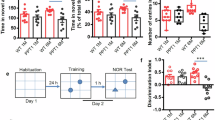
Alzheimer’s disease (AD), a prevalent neurodegenerative dementia, presents therapeutic challenges due to safety concerns about amyloid-targeting strategies. Traditional Chinese medicine (TCM) may offer alternative avenues for exploration. Ginsenoside Rg1, a key bioactive component of ginseng, has shown neuroprotective potential in okadaic acid (OKA)-induced rat model, its limited brain bioavailability suggests that its metabolite protopanaxatriol (Ppt) may exert these effects. In this study, we investigated the therapeutic effects of Ppt on OKA-induced mice model and the underlying mechanisms. Cultured hippocampal neurons were treated with OKA (0.5 nM) with or without Ppt co-treatment for 24 h. We showed that Ppt (1.25–40 nM) exerted dose-dependent neuroprotection against OKA-induced cytotoxicity, with the maximal protection observed at 10 nM. The suppressed tau aggregation by Ppt was confirmed using a Venus-tau bimolecular fluorescence complementation (BiFC) system. Molecular dynamics simulations and microscale thermophoresis (MST) revealed that Ppt bound to the catalytic domain of CDK5 at Cys83, destabilizing the CDK5/p25 complex. Co-immunoprecipitation (Co-IP) assays with CDK5 mutants (S159T, C83A, F80A and D86A) validated this interaction. In vivo mice were treated with Ppt (10 mg/kg, i.g.) for 25 days. On D8 and D9, the mice were bilaterally microinjected with OKA into the cerebral ventricles. We showed that Ppt administration improved spatial memory deficits in Novel Object Recognition and Barnes Maze tests; these effects were abolished in mice expressing a lentivirus-mediated CDK5[C83A] mutant. Hippocampal transcriptomic profiling in OKA-challenged mice following Ppt intervention revealed that Ppt modulated Drp1-mediated mitochondrial fission/fusion dynamics, mitigating OKA-induced mitochondrial homeostasis disruption. Collectively, these results demonstrate that Ppt attenuates tau pathology by selectively targeting CDK5 at Cys83, thereby reducing pathological kinase activity, rebalancing mitochondrial function, and improving cognitive outcomes in an OKA-induced mice neurodegeneration model. The study underscores the therapeutic potential of Ppt in AD treatment and supports CDK5 modulation as a strategic approach for addressing tau-related neurodegeneration.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Šimić G, Babić Leko M, Wray S, Harrington C, Delalle I, Jovanov-Milošević N, et al. Tau protein hyperphosphorylation and aggregation in Alzheimer’s disease and other tauopathies, and possible neuroprotective strategies. Biomolecules. 2016;6:6.
Murray ME, Lowe VJ, Graff-Radford NR, Liesinger AM, Cannon A, Przybelski SA, et al. Clinicopathologic and 11C-Pittsburgh compound B implications of Thal amyloid phase across the Alzheimer’s disease spectrum. Brain. 2015;138:1370–81.
Mahase E. Lecanemab and donanemab: NICE reconsiders controversial Alzheimer’s drugs. BMJ. 2025;388:r463.
Hernández-García D, Granado-Serrano AB, Martín-Gari M, Naudí A, Serrano JC. Efficacy of Panax ginseng supplementation on blood lipid profile. A meta-analysis and systematic review of clinical randomized trials. J Ethnopharmacol. 2019;243:112090.
Park HJ, Kim DH, Park SJ, Kim JM, Ryu JH. Ginseng in traditional herbal prescriptions. J Ginseng Res. 2012;36:225–41.
Zhang Y, Zhang Z, Wang H, Cai N, Zhou S, Zhao Y, et al. Neuroprotective effect of ginsenoside Rg1 prevents cognitive impairment induced by isoflurane anesthesia in aged rats via antioxidant, anti-inflammatory and anti-apoptotic effects mediated by the PI3K/AKT/GSK-3β pathway. Mol Med Rep. 2016;14:2778–84.
Song XY, Hu JF, Chu SF, Zhang Z, Xu S, Yuan YH, et al. Ginsenoside Rg1 attenuates okadaic acid induced spatial memory impairment by the GSK3β/tau signaling pathway and the Aβ formation prevention in rats. Eur J Pharmacol. 2013;710:29–38.
Feng L, Wang L, Hu C, Jiang X. Pharmacokinetics, tissue distribution, metabolism, and excretion of ginsenoside Rg1 in rats. Arch Pharm Res. 2010;33:1975–84.
Wang YZ, Chen J, Chu SF, Wang YS, Wang XY, Chen NH, et al. Improvement of memory in mice and increase of hippocampal excitability in rats by ginsenoside Rg1’s metabolites ginsenoside Rh1 and protopanaxatriol. J Pharm Sci. 2009;109:504–10.
Ihara Y, Morishima-Kawashima M, Nixon R. The ubiquitin-proteasome system and the autophagic-lysosomal system in Alzheimer disease. Cold Spring Harb Perspect Med. 2012;2:a006361.
Medina M, Avila J. New perspectives on the role of tau in Alzheimer’s disease. Implications for therapy. Biochem Pharmacol. 2014;88:540–7.
Voronkov M, Braithwaite SP, Stock JB. Phosphoprotein phosphatase 2A: a novel druggable target for Alzheimer’s disease. Future Med Chem. 2011;3:821–33.
Otero-Garcia M, Mahajani SU, Wakhloo D, Tang W, Xue YQ, Morabito S, et al. Molecular signatures underlying neurofibrillary tangle susceptibility in Alzheimer’s disease. Neuron. 2022;110:2929-2948.e8.
Bratic I, Trifunovic A. Mitochondrial energy metabolism and ageing. Biochim Biophys Acta. 2010;1797:961–7.
Macdonald R, Barnes K, Hastings C, Mortiboys H. Mitochondrial abnormalities in Parkinson’s disease and Alzheimer’s disease: can mitochondria be targeted therapeutically?. Biochem Soc Trans. 2018;46:891–909.
Petrozziello T, Bordt EA, Mills AN, Kim SE, Sapp E, Devlin BA, et al. Targeting Tau mitigates mitochondrial fragmentation and oxidative stress in amyotrophic lateral sclerosis. Mol Neurobiol. 2022;59:683–702.
Kandimalla R, Manczak M, Fry D, Suneetha Y, Sesaki H, Reddy PH. Reduced dynamin-related protein 1 protects against phosphorylated Tau-induced mitochondrial dysfunction and synaptic damage in Alzheimer’s disease. Hum Mol Genet. 2016;25:4881–97.
Patrick GN, Zukerberg L, Nikolic M, de la Monte S, Dikkes P, Tsai LH. Conversion of p35 to p25 deregulates Cdk5 activity and promotes neurodegeneration. Nature. 1999;402:615–22.
Pao PC, Tsai LH. Three decades of Cdk5. J Biomed Sci. 2021;28:79.
Wang Y, Zhao J, Guo FL, Gao X, Xie X, Liu S, et al. Metformin ameliorates synaptic defects in a mouse model of AD by inhibiting Cdk5 activity. Front Cell Neurosci. 2020;14:170.
Chou CH, Yang CR. Neuroprotective studies of evodiamine in an okadaic acid-induced neurotoxicity. Int J Mol Sci. 2021;22:5347.
Gao Y, Chu SF, Li JP, Zhang Z, Yan JQ, Wen ZL, et al. Protopanaxtriol protects against 3-nitropropionic acid-induced oxidative stress in a rat model of Huntington’s disease. Acta Pharmacol Sin. 2015;36:311–22.
Zhu J, Chu SF, Peng Y, Liu DD, Chen C, Jian WX, et al. Pyk2 inhibition attenuates hypoxic-ischemic brain injury in neonatal mice. Acta Pharmacol Sin. 2022;43:797–810.
Fernández MT, Zitko V, Gascón S, Novelli A. The marine toxin okadaic acid is a potent neurotoxin for cultured cerebellar neurons. Life Sci. 1991;49:Pl157–62.
Zhang P, Fu WY, Fu AK, Ip NY. S-nitrosylation-dependent proteasomal degradation restrains Cdk5 activity to regulate hippocampal synaptic strength. Nat Commun. 2015;6:8665.
Qu J, Nakamura T, Cao G, Holland EA, McKercher SR, Lipton SA. S-Nitrosylation activates Cdk5 and contributes to synaptic spine loss induced by beta-amyloid peptide. Proc Natl Acad Sci USA. 2011;108:14330–5.
Trott O, Olson AJ. AutoDock Vina: improving the speed and accuracy of docking with a new scoring function, efficient optimization, and multithreading. J Comput Chem. 2010;31:455–61.
Frisch M, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, et al, Gaussian 09, revision D. 01. Gaussian, Inc., Wallingford CT. 2009.
Wang J, Wang W, Kollman PA, Case DA. ANTECHAMBER: an accessory software package for molecular mechanical calculations. J Am Chem Soc. 2001;222 (U403).
Maier JA, Martinez C, Kasavajhala K, Wickstrom L, Hauser KE, Simmerling CJ, et al. ff14SB: improving the accuracy of protein side chain and backbone parameters from ff99SB. J Chem Theory Comput. 2015;11:3696–713.
Mark P, Nilsson LJ. Structure and dynamics of the TIP3P, SPC, and SPC/E water models at 298 K. J Phys Chem. 2001; 105: 9954-60.
Chen Y, Zheng Y, Fong P, Mao S, Wang QJ. The application of the MM/GBSA method in the binding pose prediction of FGFR inhibitors. Phys Chem Chem Phys. 2020; 22: 9656–63.
Genheden S, Ryde UJ. The MM/PBSA and MM/GBSA methods to estimate ligand-binding affinities. Expert Opin Drug Discov. 2015; 10: 449-61.
Weiser J, Shenkin PS, Still WC. Approximate atomic surfaces from linear combinations of pairwise overlaps (LCPO). J Comput Chem. 1999; 20: 217–30.
Hou T, Wang J, Li Y, Wang W. Assessing the performance of the MM/PBSA and MM/GBSA methods: I. The accuracy of binding free energy calculations based on molecular dynamics simulations. J Chem Inf Model. 2011;51:69–82.
Tak H, Haque MM, Kim MJ, Lee JH, Baik JH, Kim Y, et al. Bimolecular fluorescence complementation; lighting-up tau-tau interaction in living cells. PLoS One. 2013;8:e81682.
Peng Y, Ye JR, Wang SS, He WB, Feng ZP, Sun HS, et al. A small molecule 20C from Gastrodia elata inhibits α-synuclein aggregation and prevents progression of Parkinson’s disease. Cell Death Dis. 2023;14:594.
Kerppola TK. Visualization of molecular interactions using bimolecular fluorescence complementation analysis: characteristics of protein fragment complementation. Chem Soc Rev. 2009;38:2876–86.
Kim D, Lim S, Haque MM, Ryoo N, Hong HS, Rhim H, et al. Identification of disulfide cross-linked tau dimer responsible for tau propagation. Sci Rep. 2015;5:15231.
Takao D, Wang L, Boss A, Verhey KJ. Protein interaction analysis provides a map of the spatial and temporal organization of the ciliary gating zone. Curr Biol. 2017;27:2296–2306.e3.
Wang Y, Song X, Liu D, Lou YX, Luo P, Zhu T, et al. IMM-H004 reduced okadaic acid-induced neurotoxicity by inhibiting Tau pathology in vitro and in vivo. Neurotoxicology. 2019;75:221–32.
Qi Y, Guo L, Jiang Y, Shi Y, Sui H, Zhao L. Brain delivery of quercetin-loaded exosomes improved cognitive function in AD mice by inhibiting phosphorylated tau-mediated neurofibrillary tangles. Drug Deliv. 2020;27:745–55.
Taniguchi S, Fujita Y, Hayashi S, Kakita A, Takahashi H, Murayama S, et al. Calpain-mediated degradation of p35 to p25 in postmortem human and rat brains. FEBS Lett. 2001;489:46–50.
Sadleir KR, Vassar R. Cdk5 protein inhibition and Aβ42 increase BACE1 protein level in primary neurons by a post-transcriptional mechanism: implications of CDK5 as a therapeutic target for Alzheimer’s disease. J Biol Chem. 2012;287:7224–35.
Sheng Y, Zhang L, Su SC, Tsai LH, Julius Zhu J. Cdk5 is a new rapid synaptic homeostasis regulator capable of initiating the early Alzheimer-like pathology. Cereb Cortex. 2016;26:2937–51.
Mapelli M, Massimiliano L, Crovace C, Seeliger MA, Tsai LH, Meijer L, et al. Mechanism of CDK5/p25 binding by CDK inhibitors. J Med Chem. 2005;48:671–9.
Tarricone C, Dhavan R, Peng J, Areces LB, Tsai LH, Musacchio A. Structure and regulation of the CDK5-p25(nck5a) complex. Mol Cell. 2001;8:657–69.
Terni B, Boada J, Portero-Otin M, Pamplona R, Ferrer I. Mitochondrial ATP-synthase in the entorhinal cortex is a target of oxidative stress at stages I/II of Alzheimer’s disease pathology. Brain Pathol. 2010;20:222–33.
Baik SH, Kang S, Lee W, Choi H, Chung S, Kim JI, et al. A breakdown in metabolic reprogramming causes microglia dysfunction in Alzheimer’s disease. Cell Metab. 2019;30:493–507.e6.
Alldred MJ, Lee SH, Stutzmann GE, Ginsberg SD. Oxidative phosphorylation is dysregulated within the basocortical circuit in a 6-month old mouse model of Down Syndrome and Alzheimer’s disease. Front Aging Neurosci. 2021;13:707950.
Shafiei SS, Guerrero-Muñoz MJ, Castillo-Carranza DL. Tau oligomers: cytotoxicity, propagation, and mitochondrial damage. Front Aging Neurosci. 2017;9:83.
DuBoff B, Götz J, Feany MB. Tau promotes neurodegeneration via DRP1 mislocalization in vivo. Neuron. 2012;75:618–32.
Park J, Won J, Seo J, Yeo HG, Kim K, Kim YG, et al. Streptozotocin induces Alzheimer’s disease-like pathology in hippocampal neuronal cells via CDK5/Drp1-mediated mitochondrial fragmentation. Front Cell Neurosci. 2020;14:235.
Sun KH, de Pablo Y, Vincent F, Shah K. Deregulated Cdk5 promotes oxidative stress and mitochondrial dysfunction. J Neurochem. 2008;107:265–78.
Bossy-Wetzel E, Barsoum MJ, Godzik A, Schwarzenbacher R, Lipton SA. Mitochondrial fission in apoptosis, neurodegeneration and aging. Curr Opin Cell Biol. 2003;15:706–16.
Cherubini M, Puigdellívol M, Alberch J, Ginés S. Cdk5-mediated mitochondrial fission: a key player in dopaminergic toxicity in Huntington’s disease. Biochim Biophys Acta. 2015;1852:2145–60.
Meuer K, Suppanz IE, Lingor P, Planchamp V, Göricke B, Fichtner L, et al. Cyclin-dependent kinase 5 is an upstream regulator of mitochondrial fission during neuronal apoptosis. Cell Death Differ. 2007;14:651–61.
Tong, WF. The antitumor effects and mechanisms of protopanaxatriol 20S-PPT on nasopharyngeal carcinoma [D]. Jilin University, 2023. https://doi.org/10.27162/d.cnki.gjlin.2023.007757).
Acknowledgements
This work was supported by the CAMS Innovation Fund for Medical Sciences (CIFMS) (2021-I2M-1-020), the Opening Program of the Shanxi Key Laboratory of Chinese Medicine Encephalopathy (2022JD-KF-20), the Guangxi Key Laboratory of Traditional Chinese Medicine Quality Standards, Guangxi Institute of Chinese Medicine & Pharmaceutical Science (GZZK) (202401), the Provincial Natural Science Foundation of Hunan (2025JJ60645). We sincerely appreciate Prof. Yun Kyung Kim and Dr. Lim Sungsu at the Korea Institute of Science and Technology for their generous donation of pCMV6-htau40-VN173 and pCMV6-htau40-VC155 plasmids.
Author information
Authors and Affiliations
Contributions
NHC, ZZ, and YP designed the study. YP, SSW, KDL, JRY, and XY performed the experiments and drafted the manuscript; YP, SSW, ZZ, SFC, and NHC participated in data analysis; SFC, WBH, and KDL were involved in discussion of the experiments. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Peng, Y., Wang, Ss., Lai, Kd. et al. Protopanaxatriol restores cognitive function in okadaic acid-treated mice via direct inhibition of pathological CDK5 activity. Acta Pharmacol Sin (2026). https://doi.org/10.1038/s41401-025-01709-1
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41401-025-01709-1