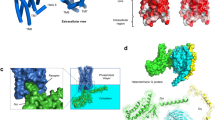
Abstract
Neuropsychiatry and neurodegenerative disorders (NPDs and NDDs) are often associated with various physiological factors. It is increasingly apparent that signal transducers like G-protein-coupled receptors (GPCRs) are critical in disease progression and constitute an important class of drug targets. Several members of class A, class B, and class C GPCR families, along with their cognate binding partners, are involved in neurotransmission and neuromodulation. The vast information about the different molecular states of these GPCRs with their signaling complexes unravels the understanding of residual specificity in these protein-protein interactions and the dynamicity of transmembrane helices. The atypical mode of interaction of GPCRs with binding partners provides valuable insights for GPCR-conformation-based drug designing. In this review, we highlight the structural features of the serotonin, dopamine, and corticotropin receptors, namely 5-HTR1A, 5-HTR1B, 5-HTR2A, 5-HTR2B, 5-HTR2C, 5-HTR4, 5-HTR6, 5-HTR7, D1-5R, and CRF1-2R when forming transient complexes with G-proteins and arrestins. The distinct structural characteristics of these receptors explain the underlying molecular mechanism for signaling specificity and related physiological processes. This review also summarizes the importance of these structural insights in developing newer and effective pharmacologically biased drugs that can mediate specific signaling pathways to GPCRs.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
The Lancet Neurology. Increasing awareness of the need for brain health. Lancet Neurol. 2024;23:325.
Feldman EL, Callaghan BC, Pop-Busui R, Zochodne DW, Wright DE, Bennet DL, et al. Diabetic neuropathy. Nat Rev Dis Prim [Internet]. 2019;5:42.
The Lancet Neurology. The evolving paradigm of Alzheimer’s disease diagnosis. Lancet Neurol. 2024;23:845.
Toft M. New gene involved in the pathogenesis of Parkinson’s disease. Lancet Neurol. 2024;23:550–2.
Ben-Joseph A, Marshall CR, Lees AJ, Noyce AJ. Ethnic variation in the manifestation of Parkinson’s disease: a narrative review. J Parkinsons Dis. 2020;10:31–45.
Cummings JL. The neuropsychiatric inventory: assessing psychopathology in dementia patients. Neurology. 1997;48:S10–6.
Laass MW, Schmitz R, Uhlig HH, Zimmer KP, Thamm M, Koletzko S. The prevalence of celiac disease in children and adolescents in Germany. Dtsch Arztebl Int. 2015;112:553–60.
Kahn RS, Sommer IE, Murray RM, Meyer-Lindenberg A, Weinberger DR, Cannon TD, et al. Schizophrenia. Nat Rev Dis Prim. 2015;1:15067.
Ripke S, Neale BM, Corvin A, Walters JTR, Farh KH, Holmans PA, et al. Biological insights from 108 schizophrenia-associated genetic loci. Nature. 2014;511:421–7.
Li P L, Snyder G E, Vanover K. Dopamine targeting drugs for the treatment of schizophrenia: past, present and future. Curr Top Med Chem. 2016;16:3385–403.
Sullivan PF, Neale MC, Kendler KS. Genetic epidemiology of major depression: review and meta-analysis. Am J Psychiatry. 2000;157:1552–62.
Fernandez-Pujals AM, Adams MJ, Thomson P, McKechanie AG, Blackwood DHR, Smith BH, et al. Epidemiology and heritability of major depressive disorder, stratified by age of onset, sex, and illness course in generation Scotland: Scottish Family Health Study (GS:SFHS). PLoS One. 2015;10:e0142197.
Luo Q, Chen J, Li Y, Wu Z, Lin X, Yao J, et al. Altered regional brain activity and functional connectivity patterns in major depressive disorder: a function of childhood trauma or diagnosis? J Psychiatr Res. 2022;147:237–47.
Roddy DW, Farrell C, Doolin K, Roman E, Tozzi L, Frodl T, et al. The hippocampus in depression: more than the sum of its parts? Advanced hippocampal substructure segmentation in depression. Biol Psychiatry. 2019;85:487–97.
Merikangas KR, Jin R, He JP, Kessler RC, Lee S, Sampson NA, et al. Prevalence and correlates of bipolar spectrum disorder in the World Mental Health Survey Initiative. Arch Gen Psychiatry. 2011;68:241–51.
Johansson V, Kuja-Halkola R, Cannon TD, Hultman CM, Hedman AM. A population-based heritability estimate of bipolar disorder - in a Swedish twin sample. Psychiatry Res. 2019;278:180–7.
Fountoulakis KN, Kelsoe JR, Akiskal H. Receptor targets for antidepressant therapy in bipolar disorder: an overview. J Affect Disord. 2012;138:222–38.
Fredriksson R, Lagerström MC, Lundin LG, Schiöth HB. The G-protein-coupled receptors in the human genome form five main families. Phylogenetic analysis, paralogon groups, and fingerprints. Mol Pharmacol. 2003;63:1256–72.
Yang D, Zhou Q, Labroska V, Qin S, Darbalaei S, Wu Y, et al. G protein-coupled receptors: structure- and function-based drug discovery. Signal Transduct Target Ther. 2021;6:7.
Kobilka BK. G protein coupled receptor structure and activation. Biochim Biophys Acta. 2007;1768:794–807.
Lee SM, Booe JM, Pioszak AA. Structural insights into ligand recognition and selectivity for class A, B, and C GPCRs. Eur J Pharmacol. 2015;763:196–205.
Wu H, Wang C, Gregory KJ, Han GW, Cho HP, Xia Y, et al. Structure of a class C GPCR metabotropic glutamate receptor 1 bound to an allosteric modulator. Science. 2014;344:58–64.
Komatsu H. Novel therapeutic GPCRs for psychiatric disorders. Int J Mol Sci. 2015;16:14109–21.
Furay AR, McDevitt RA, Miczek KA, Neumaier JF. 5-HT1B mRNA expression after chronic social stress. Behav Brain Res. 2011;224:350–7.
Mekli K, Payton A, Miyajima F, Platt H, Thomas E, Downey D, et al. The HTR1A and HTR1B receptor genes influence stress-related information processing. Eur Neuropsychopharmacol. 2011;21:129–39.
Hara MR, Sachs BD, Caron MG, Lefkowitz RJ. Pharmacological blockade of a β2AR-β-arrestin-1 signaling cascade prevents the accumulation of DNA damage in a behavioral stress model. Cell Cycle. 2013;12:219–24.
Albert PR, Lemonde S. 5-HT1A receptors, gene repression, and depression: guilt by association. Neuroscientist. 2004;10:575–93.
Lemonde S, Turecki G, Bakish D, Du L, Hrdina PD, Bown CD, et al. Impaired repression at a 5-hydroxytryptamine 1 A receptor gene polymorphism associated with major depression and suicide. J Neurosci. 2003;23:8788–99.
Parsey RV, Oquendo MA, Ogden RT, Olvet DM, Simpson N, Huang YY, et al. Altered serotonin 1 A binding in major depression: a [carbonyl-C-11]WAY100635 positron emission tomography study. Biol Psychiatry. 2006;59:106–13.
Richardson-Jones JW, Craige CP, Guiard BP, Stephen A, Metzger KL, Kung HF, et al. 5-HT1A autoreceptor levels determine vulnerability to stress and response to antidepressants. Neuron. 2010;65:40–52.
Richardson-Jones JW, Craige CP, Nguyen TH, Kung HF, Gardier AM, Dranovsky A, et al. Behavioral/systems/cognitive serotonin-1A autoreceptors are necessary and sufficient for the normal formation of circuits underlying innate anxiety. J Neurosci. 2011;31:6008–18.
Blier P, Piñeyro G, El Mansari M, Bergeron R, De Montigny C. Role of somatodendritic 5-HT autoreceptors in modulating 5-HT neurotransmissiona. Ann N Y Acad Sci. 1998;861:204–16.
Gross C, Zhuang X, Stark K, Ramboz S, Oosting R, Kirby L, et al. Serotonin1A receptor acts during development to establish normal anxiety-like behaviour in the adult. Nature. 2002;416:396–400.
Ramboz S, Oosting R, Amara DA, Kung HF, Blier P, Mendelsohn M, et al. Serotonin receptor 1 A knockout: an animal model of anxiety-related disorder. Proc Natl Acad Sci USA. 1998;95:14476–81.
Moret C, Briley M, Francè´ F. The possible role of 5-HT receptors in psychiatric disorders and their potential as a target for therapy. Eur J Pharmacol. 2000;404:1–12.
Tiger M, Varnäs K, Okubo Y, Lundberg J. The 5-HT 1B receptor - a potential target for antidepressant treatment. Psychopharmacology. 2018;235:1317–34.
Sauro KM, Becker WJ. The stress and migraine interaction. Headache. 2009;4:1378–86.
Minen MT, Begasse De Dhaem O, Kroon Van Diest A, Powers S, Schwedt TJ, Lipton R, et al. Migraine and its psychiatric comorbidities. J Neurol Neurosurg Psychiatry. 2016;87:741–9.
López-Figueroa AL, Norton CS, López-Figueroa MO, Armellini-Dodel D, Burke S, Akil H, et al. Serotonin 5-HT1A, 5-HT1B, and 5-HT2A receptor mRNA expression in subjects with major depression, bipolar disorder, and schizophrenia. Biol Psychiatry. 2004;55:225–33.
Tatarczyñska E, Kłodzińska A, Chojnacka-Wójcik E. Effects of combined administration of 5-HT1A and/or 5-HT1B receptor antagonists and paroxetine or fluoxetine in the forced swimming test in rats. Pol J Pharmacol. 2002;54:615–23.
Tatarczyńska E, Kłodzińska A, Stachowicz K, Chojnacka-Wójcik E. Effects of a selective 5-HT1B receptor agonist and antagonists in animal models of anxiety and depression. Behav Pharmacol. 2004;15:523–34.
Leonard BE. The HPA and immune axes in stress: the involvement of the serotonergic system. Eur Psychiatry. 2005;20:S302–6.
Bryson A, Carter O, Norman T, Kanaan R. 5-HT 2 A agonists: a novel therapy for functional neurological disorders?. Int J Neuropsychopharmacol. 2017;20:422–7.
Zhang G, Stackman RW. The role of serotonin 5-HT2A receptors in memory and cognition. Front Pharmacol. 2015;6:159509.
Fiorica-Howells E, Maroteaux L, Gershon MD. Serotonin and the 5-HT 2B receptor in the development of enteric neurons. J Neurosci. 2000;20:294–305.
Shyu KG. Serotonin 5-HT 2B receptor in cardiac fibroblast contributes to cardiac hypertrophy: a new therapeutic target for heart failure? Circ Res. 2009;104:1–3.
Bennet H, Mollet IG, Balhuizen A, Medina A, Nagorny C, Bagge A, et al. Serotonin (5-HT) receptor 2b activation augments glucose-stimulated insulin secretion in human and mouse islets of Langerhans. Diabetologia. 2016;59:744–54.
Yohn CN, Gergues MM, Samuels BA. The role of 5-HT receptors in depression. Mol Brain. 2017;10:28.
Berger M, Gray JA, Roth BL. The expanded biology of serotonin. Annu Rev Med. 2009;60:355–66.
Heisler LK, Zhou L, Bajwa P, Hsu J, Tecott LH. Serotonin 5-HT2C receptors regulate anxiety-like behavior. Genes Brain Behav. 2007;6:491–6.
Babić Leko M, Nikolac Perković M, Španić E, Švob Štrac D, Pleić N, Vogrinc Ž, et al. Serotonin receptor gene polymorphisms are associated with cerebrospinal fluid, genetic, and neuropsychological biomarkers of Alzheimer’s disease. Biomedicines. 2022;10:3118.
Rosel P, Arranz B, Urretavizcaya M, Oros M, San L, Navarro MA. Altered 5-HT2A and 5-HT4 postsynaptic receptors and their intracellular signalling systems IP3 and cAMP in brains from depressed violent suicide victims. Neuropsychobiology. 2004;49:189–95.
Compan V, Zhou M, Grailhe R, Gazzara RA, Martin R, Gingrich J, et al. Attenuated response to stress and novelty and hypersensitivity to seizures in 5-HT4 receptor knock-out mice. J Neurosci. 2004;24:412.
Conductier G, Dusticier N, Lucas G, Côté F, Debonnel G, Daszuta A, et al. Adaptive changes in serotonin neurons of the raphe nuclei in 5-HT4 receptor knock-out mouse. Eur J Neurosci. 2006;24:1053–62.
Borroto-Escuela DO, Narvaez M, Marcellino D, Parrado C, Narvaez JA, Tarakanov AO, et al. Galanin receptor-1 modulates 5-hydroxtryptamine-1A signaling via heterodimerization. Biochem Biophys Res Commun. 2010;393:767–72.
Tarakanov AO, Fuxe KG. Triplet puzzle: homologies of receptor heteromers. J Mol Neurosci. 2010;41:294–303.
Fuxe K, Borroto-Escuela DO, Romero-Fernandez W, Tarakanov AO, Calvo F, Garriga P, et al. On the existence and function of galanin receptor heteromers in the central nervous system. Front Endocrinol. 2012;3:127.
Renner U, Zeug A, Woehler A, Niebert M, Dityatev A, Dityateva G, et al. Heterodimerization of serotonin receptors 5-HT1A and 5-HT7 differentially regulates receptor signalling and trafficking. J Cell Sci. 2012;125:2486–99.
Molchan SE, Lawlor BA, Hill JL, Martinez RA, Davis CL, Mellow AM, et al. CSF monoamine metabolites and somatostatin in Alzheimer’s disease and major depression. Biol Psychiatry. 1991;29:1110–8.
Rocheville M, Lange DC, Kumar U, Patel SC, Patel RC, Patel YC. Receptors for dopamine and somatostatin: formation of hetero-oligomers with enhanced functional activity. Science. 2000;288:154–7.
Pei L, Li S, Wang M, Diwan M, Anisman H, Fletcher PJ, et al. Uncoupling the dopamine D1-D2 receptor complex exerts antidepressant-like effects. Nat Med. 2010;16:1393–5.
Dedic N, Chen A, Deussing JM. The CRF family of neuropeptides and their receptors-mediators of the central stress response. Curr Mol Pharmacol. 2018;11:4–31.
Keck ME. Corticotropin-releasing factor, vasopressin and receptor systems in depression and anxiety. Amino Acids. 2006;31:241–50.
Binder EB, Nemeroff CB. The CRF system, stress, depression and anxiety—insights from human genetic studies. Mol Psychiatry. 2010;15:574–88.
Bale TL, Vale WW. Increased depression-like behaviors in corticotropin-releasing factor receptor-2-deficient mice: sexually dichotomous responses. J Neurosci. 2003;23:5295–301.
Eggers AE. A serotonin hypothesis of schizophrenia. Med Hypotheses. 2013;80:791–4.
Szafran K, Faron-Górecka A, Kolasa M, Kuśmider M, Solich J, Żurawek D, et al. Potential role of G protein-coupled receptor (GPCR) heterodimerization in neuropsychiatric disorders: a focus on depression. Pharmacol Rep. 2013;65:1498–505.
Meltzer H Y, Massey BW, Horiguchi M. Serotonin receptors as targets for drugs useful to treat psychosis and cognitive impairment in schizophrenia. Curr Pharm Biotechnol. 2012;13:1572–86.
O’Donovan SM, Sullivan CR, McCullumsmith RE. The role of glutamate transporters in the pathophysiology of neuropsychiatric disorders. npj Schizophrenia. 2017;3:32.
Soares C, Da Ros LU, Machado LS, Rocha A, Lazzarotto G, Carello-Collar G, et al. The glutamatergic system in Alzheimer’s disease: a systematic review with meta-analysis. Mol Psychiatry. 2024;29:2261–73.
Javitt DC. Glutamate as a therapeutic target in psychiatric disorders. Mol Psychiatry. 2004;9:984–97.
Shah UH, González-Maeso J. Serotonin and glutamate interactions in preclinical schizophrenia models. ACS Chem Neurosci. 2019;10:3068–77.
Van Der Heijden FMMA, Tuinier S, Fekkes D, Sijben AES, Kahn RS, Verhoeven WMA. Atypical antipsychotics and the relevance of glutamate and serotonin. Eur Neuropsychopharmacol. 2004;14:259–65.
Ballanger B, Strafella AP, van Eimeren T, Zurowski M, Rusjan PM, Houle S, et al. Serotonin 2A receptors and visual hallucinations in Parkinson Disease. Arch Neurol. 2010;67:416–21.
Ballanger B, Klinger H, Eche J, Lerond J, Vallet A, Le Bars D, et al. Role of serotonergic 1A receptor dysfunction in depression associated with Parkinson’s disease. Mov Disord. 2012;27:84–9.
Gerfen CR, Engber TM, Mahan LC, Susel Z, Chase TN, Monsma FJ, et al. Dopamine receptor-regulated gene expression of striatonigral and striatopallidal neurons. Science. 1990;250:1429–32.
Obeso JA, Rodriguez-Oroz MC, Rodriguez M, Macias R, Alvarez L, Guridi J, et al. Pathophysiologic basis of surgery for Parkinson’s disease. Neurology. 2000;55:S7–12.
Thomas Broome S, Louangaphay K, Keay K, Leggio G, Musumeci G, Castorina A. Dopamine: an immune transmitter. Neural Regen Res. 2020;15:2173.
Lorke DE, Lu G, Cho E, Yew DT. Serotonin 5-HT2A and 5-HT6 receptors in the prefrontal cortex of Alzheimer and normal aging patients. BMC Neurosci. 2006;7:36.
Albasanz JL, Perez S, Barrachina M, Ferrer I, Martín M. Up-regulation of adenosine receptors in the frontal cortex in Alzheimer’s disease. Brain Pathol [Internet]. 2008;18:211.
Huang Y, Todd N, Thathiah A. The role of GPCRs in neurodegenerative diseases: avenues for therapeutic intervention. Curr Opin Pharmacol. 2017;32:96–110.
Pan X, Kaminga AC, Wen SW, Wu X, Acheampong K, Liu A. Dopamine and dopamine receptors in Alzheimer’s disease: a systematic review and network meta-analysis. Front Aging Neurosci. 2019;11:175.
Donthamsetti P, Gallo EF, Buck DC, Stahl EL, Zhu Y, Lane JR, et al. Arrestin recruitment to dopamine D2 receptor mediates locomotion but not incentive motivation. Mol Psychiatry. 2020;25:2086–100.
Koch G, Di Lorenzo F, Bonnì S, Giacobbe V, Bozzali M, Caltagirone C, et al. Dopaminergic modulation of cortical plasticity in Alzheimer’s disease patients. Neuropsychopharmacology. 2014;39:2654–61.
Krashia P, Nobili A, D’Amelio M. Unifying hypothesis of dopamine neuron loss in neurodegenerative diseases: focusing on Alzheimer’s disease. Front Mol Neurosci. 2019;12:123.
Nobili A, Latagliata EC, Viscomi MT, Cavallucci V, Cutuli D, Giacovazzo G, et al. Dopamine neuronal loss contributes to memory and reward dysfunction in a model of Alzheimer’s disease. Nat Commun. 2017;8:14727.
Campbell SN, Zhang C, Roe AD, Lee N, Lao KU, Monte L, et al. Impact of CRFR1 ablation on amyloid-β production and accumulation in a mouse model of Alzheimer’s disease. J Alzheimer’s Dis. 2015;45:1175–84.
Park H, Ran Y, Jung JI, Holmes O, Price AR, Smithson L, et al. The stress response neuropeptide CRF increases amyloid-β production by regulating γ-secretase activity. EMBO J. 2015;34:1674–86.
Huang S, Xu P, Shen DD, Simon IA, Mao C, Tan Y, et al. GPCRs steer Gi and Gs selectivity via TM5-TM6 switches as revealed by structures of serotonin receptors. Mol Cell. 2022;82:2681–95.e6.
Cao C, Barros-Álvarez X, Zhang S, Kim K, Dämgen MA, Panova O, et al. Signaling snapshots of a serotonin receptor activated by the prototypical psychedelic LSD. Neuron. 2022;110:3154–67.e7.
Choe HW, Kim YJ, Park JH, Morizumi T, Pai EF, Krau N, et al. Crystal structure of metarhodopsin II. Nature. 2011;471:651–5.
Rasmussen SGF, Devree BT, Zou Y, Kruse AC, Chung KY, Kobilka TS, et al. Crystal structure of the β2 adrenergic receptor-Gs protein complex. Nature. 2011;477:549–57.
Reiter E, Ahn S, Shukla AK, Lefkowitz RJ. Molecular mechanism of β-arrestin-biased agonism at seven-transmembrane receptors. Annu Rev Pharmacol Toxicol. 2012;52:179–97.
Desale SE, Chidambaram H, Chinnathambi S. G-protein coupled receptor, PI3K and Rho signaling pathways regulate the cascades of Tau and amyloid-β in Alzheimer’s disease. Mol Biomed. 2021;2:17.
Zamani A, Qu Z. Serotonin activates angiogenic phosphorylation signaling in human endothelial cells. FEBS Lett. 2012;586:2360–5.
Nakano N, Matsuda S, Ichimura M, Minami A, Ogino M, Murai T, et al. PI3K/AKT signaling mediated by G protein-coupled receptors is involved in neurodegenerative Parkinson’s disease (Review). Int J Mol Med. 2017;39:253–60.
Zempel H, Thies E, Mandelkow E, Mandelkow EM. Aβ oligomers cause localized Ca 2+ elevation, missorting of endogenous tau into dendrites, tau phosphorylation, and destruction of microtubules and spines. J Neurosci. 2010;30:11938–50.
Kitagishi Y, Nakanishi A, Ogura Y, Matsuda S. Dietary regulation of PI3K/AKT/GSK-3β pathway in Alzheimer’s disease. Alzheimers Res Ther. 2014;6:1–7.
McCorvy JD, Roth BL. Structure and function of serotonin G protein-coupled receptors. Pharmacol Ther. 2015;150:129–42.
Rangel-Barajas C, Israel Coronel I, Florán B. Dopamine receptors and neurodegeneration. Aging Dis. 2015;6:349.
Dautzenberg FM, Hauger RL. The CRF peptide family and their receptors: yet more partners discovered. Trends Pharmacol Sci. 2002;23:71–7.
Trzaskowski B, Latek D, Yuan S, Ghoshdastider U, Debinski A, Filipek S. Action of molecular switches in GPCRs - theoretical and experimental studies. Curr Med Chem. 2012;19:1090–109.
García-Nafría J, Nehmé R, Edwards PC, Tate CG. Cryo-EM structure of the serotonin 5-HT1B receptor coupled to heterotrimeric Go. Nature. 2018;558:7711.
Kim K, Chung KY. Many faces of the GPCR-arrestin interaction. Arch Pharm Res. 2020;43:890–9.
Teng X, Chen S, Wang Q, Chen Z, Wang X, Huang N, et al. Structural insights into G protein activation by D1 dopamine receptor. Sci Adv. 2022;8:eabo4158.
Ma S, Shen Q, Zhao LH, Mao C, Zhou XE, Shen DD, et al. Molecular basis for hormone recognition and activation of corticotropin-releasing factor receptors. Mol Cell. 2020;77:669–80.e4.
Ballesteros JA, Weinstein H. Integrated methods for the construction of three-dimensional models and computational probing of structure-function relations in G protein-coupled receptors. Methods Neurosci. 1995;25:366–428.
Palczewski K, Kumasaka T, Hori T, Behnke CA, Motoshima H, Fox BA, et al. Crystal structure of rhodopsin: a G protein-coupled receptor. Science. 2000;289:739–45.
Rosenbaum DM, Rasmussen SGF, Kobilka BK. The structure and function of G-protein-coupled receptors. Nature. 2009;459:356.
Audet M, Bouvier M. Restructuring G-protein-coupled receptor activation. Cell. 2012;151:14–23.
Rovati GE, Capra V, Neubig RR. The highly conserved DRY motif of class A G protein-coupled receptors: beyond the ground state. Mol Pharmacol. 2007;71:959–64.
Edward Zhou X, Melcher K, Eric Xu H. Structural biology of G protein-coupled receptor signaling complexes. Protein Sci. 2019;28:487–501.
Gurevich VV, Gurevich EV. GPCR signaling regulation: the role of GRKs and arrestins. Front Pharmacol. 2019;10:125.
Gurevich EV, Gurevich VV. Arrestins: ubiquitous regulators of cellular signaling pathways. Genome Biol. 2006;7:236.
Peterson YK, Luttrell LM. The diverse roles of arrestin scaffolds in G protein-coupled receptor signaling. Pharmacol Rev. 2017;69:256–97.
Kang Y, Zhou XE, Gao X, He Y, Liu W, Ishchenko A, et al. Crystal structure of rhodopsin bound to arrestin by femtosecond X-ray laser. Nature. 2015;523:561–7.
Yin W, Li Z, Jin M, Yin YL, de Waal PW, Pal K, et al. A complex structure of arrestin-2 bound to a G protein-coupled receptor. Cell Res. 2019;29:971–83.
Jaakola VP, Prilusky J, Sussman JL, Goldman A. G protein-coupled receptors show unusual patterns of intrinsic unfolding. Protein Eng Des Sel. 2005;18:103–10.
Gurevich VV, Gurevich EV. The molecular acrobatics of arrestin activation. Trends Pharmacol Sci. 2004;25:105–11.
Shukla AK, Manglik A, Kruse AC, Xiao K, Reis RI, Tseng WC, et al. Structure of active β-arrestin-1 bound to a G-protein-coupled receptor phosphopeptide. Nature. 2013;497:137–41.
Shukla AK, Westfield GH, Xiao K, Reis RI, Huang LY, Tripathi-Shukla P, et al. Visualization of arrestin recruitment by a G-protein-coupled receptor. Nature. 2014;512:218–22.
Gurevich VV, Gurevich EV. The structural basis of arrestin-mediated regulation of G-protein-coupled receptors. Pharmacol Ther. 2006;110:465–502.
Azevedo AW, Doan T, Moaven H, Sokal I, Baameur F, Vishnivetskiy SA, et al. C-terminal threonines and serines play distinct roles in the desensitization of rhodopsin, a G protein-coupled receptor. eLife. 2015;4:e05981.
Mendez A, Burns ME, Roca A, Lem J, Wu LW, Simon MI, et al. Rapid and reproducible deactivation of rhodopsin requires multiple phosphorylation sites. Neuron. 2000;28:153–64.
McDowell JH, Robinson PR, Miller RL, Brannock MT, Arendt A, Smith WC, et al. Activation of arrestin: requirement of phosphorylation as the negative charge on residues in synthetic peptides from the carboxyl-terminal region of rhodopsin. Invest Ophthalmol Vis Sci. 2001;42:1439–43.
Whistler JL, Tsao P, Von Zastrow M. A phosphorylation-regulated brake mechanism controls the initial endocytosis of opioid receptors but is not required for post-endocytic sorting to lysosomes. J Biol Chem. 2001;276:34331–8.
Kim OJ, Gardner BR, Williams DB, Marinec PS, Cabrera DM, Peters JD, et al. The role of phosphorylation in D1 dopamine receptor desensitization: evidence for a novel mechanism of arrestin association. J Biol Chem. 2004;279:7999–8010.
Kishi H, Krishnamurthy H, Galet C, Bhaskaran RS, Ascoli M. Identification of a short linear sequence present in the C-terminal tail of the rat follitropin receptor that modulates arrestin-3 binding in a phosphorylation-independent fashion. J Biol Chem. 2002;277:21939–46.
Kim KM, Valenzano KJ, Robinson SR, Yao WD, Barak LS, Caron MG. Differential regulation of the dopamine D2and D3 receptors by G protein-coupled receptor kinases and β-arrestins. J Biol Chem. 2001;276:37409–14.
Lee KB, Ptasienski JA, Pals-Rylaarsdam R, Gurevich VV, Hosey MM. Arrestin binding to the M2 muscarinic acetylcholine receptor is precluded by an inhibitory element in the third intracellular loop of the receptor. J Biol Chem. 2000;275:9284–9.
Degraff JL, Gurevich VV, Benovic JL. The third intracellular loop of alpha 2-adrenergic receptors determines subtype specificity of arrestin interaction. J Biol Chem. 2002;277:43247–52.
Bhaskaran RS, Min L, Krishnamurthy H, Ascoli M. Studies with chimeras of the gonadotropin receptors reveal the importance of third intracellular loop threonines on the formation of the receptor/nonvisual arrestin complex. Biochemistry. 2003;42:13950–9.
Gurevich VV, Dion SB, Onorato JJ, Ptasienski J, Kim CM, Sterne-Marr R, et al. Arrestin interactions with G protein-coupled receptors. J Biol Chem. 1995;270:720–31.
Hüttenrauch F, Nitzki A, Lin FT, Höning S, Oppermann M. Beta-arrestin binding to CC chemokine receptor 5 requires multiple C-terminal receptor phosphorylation sites and involves a conserved Asp-Arg-Tyr sequence motif. J Biol Chem. 2002;277:30769–77.
Kim K, Che T, Panova O, DiBerto JF, Lyu J, Krumm BE, et al. Structure of a hallucinogen-activated Gq-coupled 5-HT2A serotonin receptor. Cell. 2020;182:1574–88.e19.
Wang D, Liu D, Yuchi J, He F, Jiang Y, Cai S, et al. MusiteDeep: a deep-learning based webserver for protein post-translational modification site prediction and visualization. Nucleic Acids Res. 2020;48:W140–6.
Chen M, Zhang W, Gou Y, Xu D, Wei Y, Liu D, et al. GPS 6.0: an updated server for prediction of kinase-specific phosphorylation sites in proteins. Nucleic Acids Res. 2023;51:W243–50.
Waterhouse A, Bertoni M, Bienert S, Studer G, Tauriello G, Gumienny R, et al. SWISS-MODEL: homology modelling of protein structures and complexes. Nucleic Acids Res. 2018;46:W296–303.
Delano WL. PyMOL: an open-source molecular graphics tool. 2002. CCP4 Newsletter 40, contribution 11.
Meng EC, Goddard TD, Pettersen EF, Couch GS, Pearson ZJ, Morris JH, et al. UCSF ChimeraX: tools for structure building and analysis. Protein Sci. 2023;32:e4792.
Weller M, Virmaux N, Mandel P. Light-stimulated phosphorylation of rhodopsin in the retina: the presence of a protein kinase that is specific for photobleached rhodopsin. Proc Natl Acad Sci USA. 1975;72:381–5.
Benovic JL, Strasser RH, Caron MG, Lefkowitz RJ. Beta-adrenergic receptor kinase: identification of a novel protein kinase that phosphorylates the agonist-occupied form of the receptor. Proc Natl Acad Sci USA. 1986;83:2797–801.
Benovic JL, Onorato JJ, Arriza JL, Stone WC, Lohse M, Jenkins NA, et al. Cloning, expression, and chromosomal localization of beta-adrenergic receptor kinase 2. A new member of the receptor kinase family. J Biol Chem. 1991;266:14939–46.
Ambrose C, James M, Barnes G, Lin C, Bates G, Altherr M, et al. A novel G protein-coupled receptor kinase gene cloned from 4p16.3. Hum Mol Genet. 1992;1:697–703.
Kunapuli P, Benovic JL. Cloning and expression of GRK5: a member of the G protein-coupled receptor kinase family. Proc Natl Acad Sci USA. 1993;90:5588–92.
Benovic JL, Gomez J. Molecular cloning and expression of GRK6. A new member of the G protein-coupled receptor kinase family. J Biol Chem. 1993;268:19521–7.
Hisatomi O, Matsuda S, Satoh T, Kotaka S, Imanishi Y, Tokunaga F. A novel subtype of G-protein-coupled receptor kinase, GRK7, in teleost cone photoreceptors. FEBS Lett. 1998;424:159–64.
Krasel C, Lohse MJ. G Protein Coupled Receptor Kinases. xPharm: The Comprehensive Pharmacology Reference. Elsevier; 2007. page 1–19.
Pitcher JA, Freedman NJ, Lefkowitz RJ. G protein-coupled receptor kinases. Annu Rev Biochem. 1998;67:653–92.
Reiter E, Lefkowitz RJ. GRKs and β-arrestins: roles in receptor silencing, trafficking and signaling. Trends Endocrinol Metab. 2006;17:159–65.
Inglesef J, Freedman NJ, Koch WJ, Lefkowitz RJ. Structure and mechanism of the G protein-coupled receptor kinases. J Biol Chem. 1993;268:23735–8.
Rankin ML, Marinec PS, Cabrera DM, Wang Z, Jose PA, Sibley DR. The D1 dopamine receptor is constitutively phosphorylated by G protein-coupled receptor kinase 4. Mol Pharmacol. 2006;69:759–69.
Pronin AN, Morris AJ, Surguchov A, Benovic JL. Synucleins are a novel class of substrates for G protein-coupled receptor kinases. J Biol Chem. 2000;275:26515–22.
He L, Zhao Q, Qi J, Wang Y, Han W, Chen Z, et al. Structural insights into constitutive activity of 5-HT 6 receptor. Proc Natl Acad Sci USA. 2023;120:e2209917120.
Yin J, Chen KYM, Clark MJ, Hijazi M, Kumari P, Bai XC, et al. Structure of a D2 dopamine receptor–G-protein complex in a lipid membrane. Nature. 2020;584:125–9.
Barthet G, Gaven F, Framery B, Shinjo K, Nakamura T, Claeysen S, et al. Uncoupling and endocytosis of 5-hydroxytryptamine 4 receptors. J Biol Chem. 2005;280:27924–34.
Nebigil CG, Garnovskaya MN, Casanas SJ, Mulheron JG, Parker EM, Gettys TW, et al. Agonist-induced desensitization and phosphorylation of human 5-HT1A receptor expressed in Sf9 insect cells. Biochemistry. 1995;34:11954–62.
Chen Q, Plasencia M, Li Z, Mukherjee S, Patra D, Chen CL, et al. Structures of rhodopsin in complex with G-protein-coupled receptor kinase 1. Nature. 2021;595:600–5.
Duan J, Liu H, Zhao F, Yuan Q, Ji Y, Cai X, et al. GPCR activation and GRK2 assembly by a biased intracellular agonist. Nature. 2023;620:676–81.
Bychkov ER, Gurevich VV, Joyce JN, Benovic JL, Gurevich EV. Arrestins and two receptor kinases are upregulated in Parkinson’s disease with dementia. Neurobiol Aging. 2008;29:379–96.
Frieder A, Fersh M, Hainline R, Deligiannidis KM. Pharmacotherapy of postpartum depression: current approaches and novel drug development. CNS Drugs. 2019;33:265–82.
Ferguson JM. SSRI antidepressant medications: adverse effects and tolerability. Prim Care Companion J Clin Psychiatry. 2001;3:22–7.
Lenzer J. FDA warns that antidepressants may increase suicidality in adults. BMJ. 2005;331:70.
CHMP. Questions and answers on the withdrawal of the marketing application for pristiqs. 2008. Available from: http://www.emea.europa.eu
Clemow DB, Johnson KW, Hochstetler HM, Ossipov MH, Hake AM, Blumenfeld AM. Lasmiditan mechanism of action – review of a selective 5-HT1F agonist. J Headache Pain. 2020;21:71.
Martí-Solano M, Sanz F, Pastor M, Selent J. A dynamic view of molecular switch behavior at serotonin receptors: implications for functional selectivity. PLoS One. 2014;9:e109312.
Kling RC, Tschammer N, Lanig H, Clark T, Gmeiner P. Active-state model of a dopamine D2 receptor - Gαi complex stabilized by aripiprazole-type partial agonists. PLoS One. 2014;9:e100069.
Ferraiolo M, Hermans E. The complex molecular pharmacology of the dopamine D2 receptor: Implications for pramipexole, ropinirole, and rotigotine. Pharmacol Ther. 2023;245:108392.
Yegla B, Rubin J, Grall M, Formella A, Jenner P. Apomorphine differentially engages the cAMP and β-arrestin signaling pathways relative to dopamine at human dopamine receptors. J Neural Transm. 2025;132:1751–60.
Pogorelov VM, Rodriguiz RM, Roth BL, Wetsel WC. The G protein biased serotonin 5-HT2A receptor agonist lisuride exerts anti-depressant drug-like activities in mice. Front Mol Biosci. 2023;10:1233743.
Casiraghi M, Wang H, Brennan PC, Habrian C, Hübner H, Schmidt MF, et al. Structure and dynamics determine G protein coupling specificity at a class A GPCR. Sci Adv. 2025;11:eadq3971.
Wang C, Jiang Y, Ma J, Wu H, Wacker D, Katritch V, et al. Structural basis for molecular recognition at serotonin receptors. Science. 2013;340:610–4.
Xu P, Huang S, Zhang H, Mao C, Zhou XE, Cheng X, et al. Structural insights into the lipid and ligand regulation of serotonin receptors. Nature. 2021;592:469–73.
Huang S, Xu P, Tan Y, You C, Zhang Y, Jiang Y, et al. Structural basis for recognition of anti-migraine drug lasmiditan by the serotonin receptor 5-HT1F–G protein complex. Cell Res. 2021;31:1036–8.
Zhang S, Chen H, Zhang C, Yang Y, Popov P, Liu J, et al. Inactive and active state structures template selective tools for the human 5-HT5A receptor. Nat Struct Mol Biol. 2022;29:677–87.
Xu P, Huang S, Krumm BE, Zhuang Y, Mao C, Zhang Y, et al. Structural genomics of the human dopamine receptor system. Cell Res. 2023;33:604–16.
Zhuang Y, Xu P, Mao C, Wang L, Krumm B, Zhou XE, et al. Structural insights into the human D1 and D2 dopamine receptor signaling complexes. Cell. 2021;184:931–42.e18.
Kimura KT, Asada H, Inoue A, Kadji FMN, Im D, Mori C, et al. Structures of the 5-HT2A receptor in complex with the antipsychotics risperidone and zotepine. Nat Struct Mol Biol. 2019;26:121–8.
Fan L, Tan L, Chen Z, Qi J, Nie F, Luo Z, et al. Haloperidol bound D2 dopamine receptor structure inspired the discovery of subtype selective ligands. Nat Commun. 2020;11:1074.
Xu P, Huang S, Mao C, Krumm BE, Zhou XE, Tan Y, et al. Structures of the human dopamine D3 receptor-Gi complexes. Mol Cell. 2021;81:1147–59.e4.
Xu Z, Guo L, Yu J, Shen S, Wu C, Zhang W, et al. Ligand recognition and G-protein coupling of trace amine receptor TAAR1. Nature. 2023;624:672–81.
Liu H, Zheng Y, Wang Y, Wang Y, He X, Xu P, et al. Recognition of methamphetamine and other amines by trace amine receptor TAAR1. Nature. 2023;624:663–71.
Ullrich A, Schneider J, Braz JM, Neu E, Staffen N, Stanek M, et al. Discovery of a functionally selective serotonin 5-HT1A receptor agonist for the treatment of pain. bioRxiv [Preprint]. 2023 [cited 2025 Apr 20];2023.09.11.557127. Available from: https://doi.org/10.1101/2023.09.11.557127
Chen Z, Yu J, Wang H, Xu P, Fan L, Sun F, et al. Flexible scaffold-based cheminformatics approach for polypharmacological drug design. Cell. 2024;187:2194–208.e22.
Warren AL, Lankri D, Cunningham MJ, Serrano IC, Parise LF, Kruegel AC, et al. Structural pharmacology and therapeutic potential of 5-methoxytryptamines. Nature. 2024;630:237–46.
Yin W, Zhou XE, Yang D, de Waal PW, Wang M, Dai A, et al. Crystal structure of the human 5-HT1B serotonin receptor bound to an inverse agonist. Cell Discov. 2018;4:12.
Miyagi H, Asada H, Suzuki M, Takahashi Y, Yasunaga M, Suno C, et al. The discovery of a new antibody for BRIL-fused GPCR structure determination. Sci Rep. 2020;10:11669.
Kaplan AL, Confair DN, Kim K, Barros-Álvarez X, Rodriguiz RM, Yang Y, et al. Bespoke library docking for 5-HT2A receptor agonists with antidepressant activity. Nature. 2022;610:582–91.
Chen Z, Fan L, Wang H, Yu J, Lu D, Qi J, et al. Structure-based design of a novel third-generation antipsychotic drug lead with potential antidepressant properties. Nat Neurosci. 2022;25:39–49.
Cao D, Yu J, Wang H, Luo Z, Liu X, He L, et al. Structure-based discovery of nonhallucinogenic psychedelic analogs. Science. 2022;375:403–11.
Lyu J, Kapolka N, Gumpper R, Alon A, Wang L, Jain MK, et al. AlphaFold2 structures guide prospective ligand discovery. Science. 2024;384:eadn6354.
Oguma T, Jino K, Nakahara K, Asada H, Fuchino K, Nagatani K, et al. Dual 5-HT2A and 5-HT2C receptor inverse agonist that affords in vivo antipsychotic efficacy with minimal hERG inhibition for the treatment of dementia-related psychosis. J Med Chem. 2024;67:14478–92.
Gumpper RH, Jain MK, Kim K, Sun R, Sun N, Xu Z, et al. The structural diversity of psychedelic drug actions revealed. Nat Commun. 2025;16:2734.
Wacker D, Wang C, Katritch V, Han GW, Huang XP, Vardy E, et al. Structural features for functional selectivity at serotonin receptors. Science. 2013;340:615–9.
Liu W, Wacker D, Gati C, Han GW, James D, Wang D, et al. Serial femtosecond crystallography of G protein–coupled receptors. Science. 2013;342:1521–4.
Wacker D, Wang S, McCorvy JD, Betz RM, Venkatakrishnan AJ, Levit A, et al. Crystal structure of an LSD-bound human serotonin receptor. Cell. 2017;168:377–389.e12.
Ishchenko A, Wacker D, Kapoor M, Zhang A, Han GW, Basu S, et al. Structural insights into the extracellular recognition of the human serotonin 2B receptor by an antibody. Proc Natl Acad Sci USA. 2017;114:8223–8.
McCorvy JD, Wacker D, Wang S, Agegnehu B, Liu J, Lansu K, et al. Structural determinants of 5-HT2B receptor activation and biased agonism. Nat Struct Mol Biol. 2018;25:787–96.
Peng Y, McCorvy JD, Harpsøe K, Lansu K, Yuan S, Popov P, et al. 5-HT2C receptor structures reveal the structural basis of GPCR polypharmacology. Cell. 2018;172:719–730.e14.
Gumpper RH, Fay JF, Roth BL. Molecular insights into the regulation of constitutive activity by RNA editing of 5HT2C serotonin receptors. Cell Rep. 2022;40:111211.
Teng X, Chen S, Nie Y, Xiao P, Yu X, Shao Z, et al. Ligand recognition and biased agonism of the D1 dopamine receptor. Nat Commun. 2022;13:3186.
Xiao P, Yan W, Gou L, Zhong YN, Kong L, Wu C, et al. Ligand recognition and allosteric regulation of DRD1-Gs signaling complexes. Cell. 2021;184:943–56.e18.
Sun B, Feng D, Chu MLH, Fish I, Lovera S, Sands ZA, et al. Crystal structure of dopamine D1 receptor in complex with G protein and a non-catechol agonist. Nat Commun. 2021;12:3305.
Liang YL, Belousoff MJ, Zhao P, Koole C, Fletcher MM, Truong TT, et al. Toward a structural understanding of class B GPCR peptide binding and activation. Mol Cell. 2020;77:656–68.e5.
Zhao LH, Lin J, Ji SY, Zhou XE, Mao C, Shen DD, et al. Structure insights into selective coupling of G protein subtypes by a class B G protein-coupled receptor. Nat Commun. 2022;13:6670.
Peng L, Gu L, Li B, Hertz L. Fluoxetine and all other SSRIs are 5-HT2B agonists - importance for their therapeutic effects. Curr Neuropharmacol. 2014;12:365–79.
Richelson E, Nelson A. Antagonism by antidepressants of neurotransmitter receptors of normal human brain in vitro. J Pharmacol Exp Ther. 1984;230:94–102.
Dunner DL, Goldstein DJ, Mallinckrodt C, Lu Y, Detke MJ. Duloxetine in treatment of anxiety symptoms associated with depression. Depress Anxiety. 2003;18:53–61.
Berendsen HHG, Broekkamp CLE. Indirect in vivo 5-HT 1A -agonistic effects of the new antidepressant mirtazapine. Psychopharmacology. 1997;133:275–82.
Cruz MP. Vilazodone HCl (Viibryd): a serotonin partial agonist and reuptake inhibitor for the treatment of major depressive disorder. Pharmacol Ther. 2012;37:28.
Ikemoto F, Toru T, Aijima H, Natsumeda Y. [Rizatriptan (Maxalt), a new entity of triptan for migraine: pharmacology and therapeutic relevance]. Nihon Yakurigaku Zasshi. 2004;123:295–302.
Jacob H, Braekow P, Schwarz R, Höhm C, Kirchhefer U, Hofmann B, et al. Ergotamine stimulates human 5-HT4-serotonin receptors and human H2-histamine receptors in the heart. Int J Mol Sci. 2023;24:4749.
Tuplin EW, Holahan MR. Aripiprazole, a drug that displays partial agonism and functional selectivity. Curr Neuropharmacol. 2017;15:1192–207.
Ghanbari R, El Mansari M, Shahid M, Blier P. Electrophysiological characterization of the effects of asenapine at 5-HT(1A), 5-HT(2A), alpha(2)-adrenergic and D(2) receptors in the rat brain. Eur Neuropsychopharmacol. 2009;19:177–87.
Fakra E, Azorin JM. Clozapine for the treatment of schizophrenia. Expert Opin Pharmacother. 2012;13:1923–35.
Schmid CL, Streicher JM, Meltzer HY, Bohn LM. Clozapine acts as an agonist at serotonin 2A receptors to counter MK-801-induced behaviors through a βarrestin2-independent activation of Akt. Neuropsychopharmacology. 2014;39:1902–13.
Bymaster FP, Nelson DL, Delapp NW, Falcone JF, Eckols K, Truex LL, et al. Antagonism by olanzapine of dopamine D1, serotonin2, muscarinic, histamine H1 and α1-adrenergic receptors in vitro. Schizophr Res. 1999;37:107–22.
Janssen PA, Niemegeers CJ, Awouters F, Schellekens KH, Megens AA, Meert TF. Pharmacology of risperidone (R 64 766), a new antipsychotic with serotonin-S2 and dopamine-D2 antagonistic properties. J Pharmacol Exp Ther. 1988;244:685–93.
Citrome L. Cariprazine for the treatment of schizophrenia: a review of this dopamine D3-preferring D3/D2 receptor partial agonist. Clin Schizophr Relat Psychoses. 2016;10:109–19.
Haloperidol - PubMed [Internet]. [cited 2025 Mar 16]. Available from: https://pubmed.ncbi.nlm.nih.gov/32809727/
Viaro R, Longo F, Vincenzi F, Varani K, Morari M. l-DOPA promotes striatal dopamine release through D1 receptors and reversal of dopamine transporter. Brain Res. 2021;1768:147583.
Wood M, Dubois V, Scheller D, Gillard M. Rotigotine is a potent agonist at dopamine D1 receptors as well as at dopamine D2 and D3 receptors. Br J Pharmacol. 2015;172:1124–35.
Acknowledgements
We would also like to express our gratitude towards all the web servers and free open-access software used in this study. All illustrations have been created/edited using https://www.biorender.com/.
Funding
The authors declare that no funds, grants, or other support were received during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
KP – conceptualization, editing, and review of manuscript; ODG – writing and editing of original manuscript, formal analysis, preparation of images; IC – writing and editing of original manuscript, preparation of images. All authors contributed equally to the preparation of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Dutta Gupta, O., Chakraborty, I. & Pal, K. A structural overview of G-protein-coupled receptors in neurological disorders. Acta Pharmacol Sin (2026). https://doi.org/10.1038/s41401-025-01712-6
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41401-025-01712-6