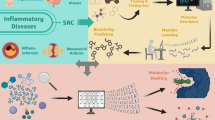
Abstract
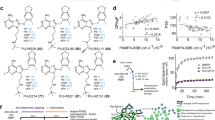
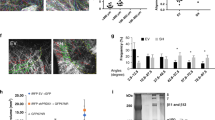
Peroxiredoxin 1 (PRDX1) is a pivotal antioxidant enzyme maintaining intracellular reactive oxygen species (ROS) balance. Deficiency of PRDX1 aggravates oxidative stress-related pathologies, whereas enhanced PRDX1 activity confers cytoprotection. Small-molecule agonists boosting PRDX1 peroxidase activity hold therapeutic promise, yet to date only two such agonists-rosmarinic acid (RA) and salvianolic acid B (SAB)-have been reported, both by our laboratory. These polyphenolic compounds are chemically rigid and recalcitrant to modification. Here, we resolved the crystal structure of PRDX1 in complex with salvianolic acid C (SAC), revealing a conserved danshensu substructure shared by SAC, RA, and SAB. Guided by this pharmacophore, we designed a scaffold hopping core structure and generated 160 derivatives via in situ click reaction. Among them, LC-PDA-01, a non-polyphenolic scaffold, exhibited the highest PRDX1 activation (EC50 = 111.8 nM). This work discloses the first structurally tractable PRDX1 agonist and highlights combinatorial click chemistry’s utility in transforming natural product motifs into drug-like molecules.

This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout









Similar content being viewed by others
References
Barnham KJ, Masters CL, Bush AI. Neurodegenerative diseases and oxidative stress. Nat Rev Drug Discov. 2004;3:205–14.
Sachidanandam K, Fagan SC, Ergul A. Oxidative stress and cardiovascular disease: antioxidants and unresolved issues. Cardiovasc Drug Rev. 2005;23:115–32.
Lawless MW, O’Byrne KJ, Gray SG. Targeting oxidative stress in cancer. Expert Opin Ther Targets. 2010;14:1225–45.
Giacco F, Brownlee M. Oxidative stress and diabetic complications. Circ Res. 2010;107:1058–70.
Forrester SJ, Kikuchi DS, Hernandes MS, Xu Q, Griendling KK. Reactive oxygen species in metabolic and inflammatory signaling. Circ Res. 2018;122:877–902.
Ju S, Singh MK, Han S, Ranbhise J, Ha J, Choe W, et al. Oxidative stress and cancer therapy: controlling cancer cells using reactive oxygen species. Int J Mol Sci. 2024;25:12387.
Afzal S, Abdul Manap AS, Attiq A, Albokhadaim I, Kandeel M, Alhojaily SM. From imbalance to impairment: the central role of reactive oxygen species in oxidative stress-induced disorders and therapeutic exploration. Front Pharmacol. 2023;14:1269581.
Canakci CF, Cicek Y, Canakci V. Reactive oxygen species and human inflammatory periodontal diseases. Biochemistry. 2005;70:619–28.
Tang SP, Mao XL, Chen YH, Yan LL, Ye LP, Li SW. Reactive oxygen species induce fatty liver and ischemia-reperfusion injury by promoting inflammation and cell death. Front Immunol. 2022;13:870239.
Nesci S, Spagnoletta A, Oppedisano F. Inflammation, mitochondria and natural compounds together in the circle of trust. Int J Mol Sci. 2023;24:6106.
Zhang C, Li CM, Jiang Q. Role of oxidative stress in intervertebral disc degeneration: mechanisms, pathogenesis, and therapeutic strategies. Mol Biol Rep. 2025;52:444.
Wood ZA, Poole LB, Karplus PA. Peroxiredoxin evolution and the regulation of hydrogen peroxide signaling. Science. 2003;300:650–3.
Perkins A, Nelson KJ, Parsonage D, Poole LB, Karplus PA. Peroxiredoxins: guardians against oxidative stress and modulators of peroxide signaling. Trends Biochem Sci. 2015;40:435–45.
Troussicot L, Burmann BM, Molin M. Structural determinants of multimerization and dissociation in 2-Cys peroxiredoxin chaperone function. Structure. 2021;29:640–54.
Song Y, Wang X, Sun Y, Yu N, Tian Y, Han J, et al. PRDX1 inhibits ferroptosis by binding to Cullin-3 as a molecular chaperone in colorectal cancer. Int J Biol Sci. 2024;20:5070–86.
Zhao Q, Ding Y, Deng Z, Lee OY, Gao P, Chen P, et al. Natural products triptolide, celastrol, and withaferin A inhibit the chaperone activity of peroxiredoxin I. Chem Sci. 2015;6:4124–30.
Neumann CA, Krause DS, Carman CV, Das S, Dubey DP, Abraham JL, et al. Essential role for the peroxiredoxin Prdx1 in erythrocyte antioxidant defence and tumour suppression. Nature. 2003;424:561–5.
Rhee SG, Chae HZ, Kim K. Peroxiredoxins: a historical overview and speculative preview of novel mechanisms and emerging concepts in cell signaling. Free Radic Biol Med. 2005;38:1543–52.
Rhee SG, Kil IS. Multiple functions and regulation of mammalian peroxiredoxins. Annu Rev Biochem. 2017;86:749–75.
Wood ZA, Schroder E, Robin Harris J, Poole LB. Structure, mechanism and regulation of peroxiredoxins. Trends Biochem Sci. 2003;28:32–40.
Cox AG, Winterbourn CC, Hampton MB. Mitochondrial peroxiredoxin involvement in antioxidant defence and redox signalling. Biochem J. 2009;425:313–25.
Ismail T, Kim Y, Lee H, Lee DS, Lee HS. Interplay between mitochondrial peroxiredoxins and ROS in cancer development and progression. Int J Mol Sci. 2019;20:4407.
Kim S, Lee W, Jo H, Sonn SK, Jeong SJ, Seo S, et al. The antioxidant enzyme Peroxiredoxin-1 controls stroke-associated microglia against acute ischemic stroke. Redox Biol. 2022;54:102347.
Almasi N, Torok S, Valkusz Z, Tajti M, Csonka A, Murlasits Z, et al. Sigma-1 receptor engages an anti-inflammatory and antioxidant feedback loop mediated by peroxiredoxin in experimental colitis. Antioxidants. 2020;9:1081.
Torok S, Almasi N, Veszelka M, Borzsei D, Szabo R, Varga C. Protective effects of H2S donor treatment in experimental colitis: a focus on antioxidants. Antioxidants. 2023;12:1025.
Martinez-Pinna R, Ramos-Mozo P, Madrigal-Matute J, Blanco-Colio LM, Lopez JA, Calvo E, et al. Identification of peroxiredoxin-1 as a novel biomarker of abdominal aortic aneurysm. Arterioscler Thromb Vasc Biol. 2011;31:935–43.
Tang C, Yin G, Huang C, Wang H, Gao J, Luo J, et al. Peroxiredoxin-1 ameliorates pressure overload-induced cardiac hypertrophy and fibrosis. Biomed Pharmacother. 2020;129:110357.
Mei W, Peng Z, Lu M, Liu C, Deng Z, Xiao Y, et al. Peroxiredoxin 1 inhibits the oxidative stress induced apoptosis in renal tubulointerstitial fibrosis. Nephrology. 2015;20:832–42.
Watanabe A, Yoneda M, Ikeda F, Sugai A, Sato H, Kai C. Peroxiredoxin 1 is required for efficient transcription and replication of measles virus. J Virol. 2011;85:2247–53.
Yin W, Xu H, Bai Z, Wu Y, Zhang Y, Liu R, et al. Inhibited peroxidase activity of peroxiredoxin 1 by palmitic acid exacerbates nonalcoholic steatohepatitis in male mice. Nat Commun. 2025;16:598.
Xu H, Zhao H, Ding C, Jiang D, Zhao Z, Li Y, et al. Celastrol suppresses colorectal cancer via covalent targeting peroxiredoxin 1. Signal Transduct Target Ther. 2023;8:51.
Li Y, Zhu Y, Shang F, Xu L, Jiang D, Sun B, et al. Discovery of urea derivatives of celastrol as selective peroxiredoxin 1 inhibitors against colorectal cancer cells. J Med Chem. 2024;67:7176–96.
Chen S, Wang Z, Gao J, Wang Y, Liang J, Zhu Y, et al. Rapid discovery of celastrol derivatives as potent and selective PRDX1 inhibitors via microplate-based parallel compound library and in situ screening. J Med Chem. 2025;68:13609–27.
Dang W, Wang X, Li H, Xu Y, Li X, Huang S, et al. Augmentation of PRDX1–DOK3 interaction alleviates rheumatoid arthritis progression by suppressing plasma cell differentiation. Acta Pharm Sin B. 2025;15:3997–4013.
Yin ZY, Zheng T, Ho CT, Huang QR, Wu QL, Zhang M. Improving the stability and bioavailability of tea polyphenols by encapsulations: a review. Food Sci Hum Wellness. 2022;11:537–56.
Ciupei D, Colisar A, Leopold L, Stanila A, Diaconeasa ZM. Polyphenols: from classification to therapeutic potential and bioavailability. Foods. 2024;13:4131.
Bishnoi P, Saroha B, Kumar S, Kumar G, Bhardwaj A, Kumari M, et al. Click chemistry: an overview and recent updates in the medicinal attributes of click-derived heterocycles. Mol Divers. 2025;29:5219–81.
Hou W, Zhang Y, Huang F, Chen W, Gu Y, Wang Y, et al. Bioinspired selenium-nitrogen exchange (SeNEx) click chemistry suitable for nanomole-scale medicinal chemistry and bioconjugation. Angew Chem Int Ed. 2024;63:e202318534.
Ghosal K, Bhattacharyya SK, Mishra V, Zuilhof H. Click chemistry for biofunctional polymers: from observing to steering cell behavior. Chem Rev. 2024;124:13216–13300.
Thirumurugan P, Matosiuk D, Jozwiak K. Click chemistry for drug development and diverse chemical-biology applications. Chem Rev. 2013;113:4905–79.
Jiang X, Hao X, Jing L, Wu G, Kang D, Liu X, et al. Recent applications of click chemistry in drug discovery. Expert Opin Drug Discov. 2019;14:779–89.
Khandelwal R, Vasava M, Abhirami RB, Karsharma M. Recent advances in triazole synthesis via click chemistry and their pharmacological applications: a review. Bioorg Med Chem Lett. 2024;112:129927.
Agrahari AK, Bose P, Jaiswal MK, Rajkhowa S, Singh AS, Hotha S, et al. Cu(I)-catalyzed click chemistry in glycoscience and their diverse applications. Chem Rev. 2021;121:7638–956.
Wang X, Huang B, Liu X, Zhan P. Discovery of bioactive molecules from CuAAC click-chemistry-based combinatorial libraries. Drug Discov Today. 2016;21:118–32.
Liang L, Astruc. D. The copper(I)-catalyzed alkyne-azide cycloaddition (CuAAC) “click” reaction and its applications: An overview. Coord Chem Rev. 2011;255:2933–45.
Jing L, Wu G, Zhao F, Jiang X, Liu N, Feng D, et al. Discovery of potent HIV-1 NNRTIs by CuAAC click-chemistry-based miniaturized synthesis, rapid screening and structure optimization. Eur J Med Chem. 2024;277:116772.
Itoh Y, Zhan P, Tojo T, Jaikhan P, Ota Y, Suzuki M, et al. Discovery of selective histone deacetylase 1 and 2 inhibitors: screening of a focused library constructed by click chemistry, kinetic binding analysis, and biological evaluation. J Med Chem. 2023;66:15171–88.
Yang M, Lee MK, Gao S, Song L, Jang HY, Jo I, et al. Miniaturized modular click chemistry-enabled rapid discovery of unique SARS-CoV-2 M(pro) inhibitors with robust potency and drug-like profile. Adv Sci. 2024;11:e2404884.
Meng G, Guo T, Ma T, Zhang J, Shen Y, Sharpless KB, et al. Modular click chemistry libraries for functional screens using a diazotizing reagent. Nature. 2019;574:86–89.
Muller J, Feifel SC, Schmiederer T, Zocher R, Sussmuth RD. In vitro synthesis of new cyclodepsipeptides of the PF1022-type: probing the alpha-D-hydroxy acid tolerance of PF1022 synthetase. ChemBioChem. 2009;10:323–8.
Wang R, Chen X, Li H, Chen X, Sun D, Yu D, et al. Danshensu inhibits SARS-CoV-2 by targeting its main protease as a specific covalent inhibitor and discovery of bifunctional compounds eliciting antiviral and anti-inflammatory activity. Int J Biol Macromol. 2024;257:128623.
Acknowledgements
This work was partially supported by the National Key Research and Development Program of China (2023YFD1800102 to ZHL) and supported by the National Natural Science Foundation of China (81903538 to HZ, 82104064 to Heng Xu, 82304301 to Huan Xiong), the Strategic Priority Research Program of the Chinese Academy of Sciences (XDB0830301 to CL), the Science and Technology Commission of Shanghai Municipality (22ZR1474200 to HZ, 25ZR1402556 to Heng Xu), Science and Technology Department of Guizhou Province ([2024]015), Applied Basic Research Foundation of Yunnan Province (202501BC070005), Zhongshan Municipal Bureau of Science and Technology (CXTD2023010 to CL, 2023B2030 to Huan Xiong). We are grateful to National Centre for Protein Science Shanghai (Protein Expression and Purification system) for their instrument support and technical assistance. We thank the staff from BL19U1 beamline of National Facility for Protein Science in Shanghai (NFPS) at Shanghai Synchrotron Radiation Facility, for assistance during data collection.
Author information
Authors and Affiliations
Contributions
CL, Huan Xiong, and HZ conceived the original hypothesis and supervised the study. YYZ, SC, SNZ and YSZ performed the study. YYZ, ZYW, XMW, ZHL and Heng Xu performed the protein purification and other biochemical experiments. SC, SNZ, YSZ, JYL, JJL, PFL and WLX synthesized the PRDX1 agonists. YYZ, SC, HengXu, HuanXiong and HZ wrote the paper. All authors read and contributed to the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhu, Yy., Chen, S., Zhang, Sn. et al. Design and evaluation of peroxiredoxin 1 agonist based on scaffold hopping of salvianolic acids and combinatorial click chemistry. Acta Pharmacol Sin (2026). https://doi.org/10.1038/s41401-026-01765-1
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41401-026-01765-1