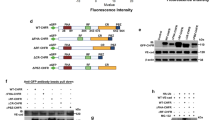
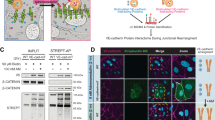
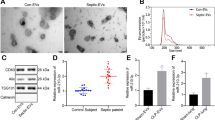
Abstract
Endothelial barrier dysfunction and consequent vascular injury are central contributors to acute lung injury (ALI) during sepsis. However, the underlying mechanisms remain incompletely understood, and effective therapeutic strategies targeting endothelial repair are still lacking. Here, we identify that intracellular leucine-rich α2-glycoprotein 1 (LRG1) in endothelial cells (EC) is significantly upregulated and directly promotes the degradation of vascular endothelial cadherin (VE-cadherin), a core adherens junction protein essential for maintaining vascular barrier integrity in septic ALI. Mechanistically, LRG1 recruits the E3 ubiquitin ligase membrane-associated ring-CH-type finger 2 (MARCH2) to catalyze K48-linked polyubiquitination of VE-cadherin at lysine 633, leading to its proteasomal degradation and subsequent endothelial barrier disruption. Genetic deletion of Lrg1 or pharmacological intervention with a proteolysis targeting chimera (PROTAC)-based degradation strategy significantly reduced VE-cadherin loss, alleviated endothelial hyperpermeability, and mitigated ALI in septic mice. Collectively, our study elucidates a previously unrecognized role of endothelial LRG1 in disrupting EC adherens junctions, providing novel insights into the pathogenesis of sepsis-associated injury and proposing a potential therapeutic strategy for sepsis-induced ALI and acute respiratory distress syndrome (ARDS).

This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Bersten AD, Edibam C, Hunt T, Moran J. Incidence and mortality of acute lung injury and the acute respiratory distress syndrome in three Australian States. Am J Respir Crit Care Med. 2002;165:443–8.
Liu Q, Du JC, Yu XZ, Xu J, Huang FM, Li XY, et al. miRNA-200c-3p is crucial in acute respiratory distress syndrome. Cell Discov. 2017;3:17021.
Xia Y, Xu QC, Guo ZY, Zhang HJ, Cao YY, Qi YP, et al. A modified screening protocol for ARDS in patients with respiratory support based on SpO2 and FiO2: a single-center prospective, observational study. J Intensive Med. 2025;5:51–7.
Zhang HL, Xu YD, Huang X, Yang SY, Li RT, Wu YR. Extracorporeal membrane oxygenation in adult patients with sepsis and septic shock: Why, how, when, and for whom. J Intensive Med. 2024;4:62–72.
Zou SF, Bai J, Liang CX, Yang JF, Xiao J. Plasmacytoid dendritic cells in sepsis-associated acute lung injury: a potential therapeutic target. Anesthesiology and perioperative. Science. 2025;3:36.
Meyer NJ, Gattinoni L, Calfee CS. Acute respiratory distress syndrome. Lancet. 2021;398:622–37.
Auriemma CL, Zhuo HJ, Delucchi K, Deiss T, Liu T, Jauregui A, et al. Acute respiratory distress syndrome-attributable mortality in critically ill patients with sepsis. Intensive Care Med. 2020;46:1222–31.
Wick KD, Ware LB, Matthay MA. Acute respiratory distress syndrome. BMJ. 2024;387:e076612.
Matthay MA, Arabi Y, Arroliga AC, Bernard G, Bersten AD, Brochard LJ, et al. A new global definition of acute respiratory distress syndrome. Am J Respir Crit Care Med. 2024;209:37–47.
Yang K, Fan M, Wang XH, Xu JJ, Wang YN, Gill PS, et al. Lactate induces vascular permeability via disruption of VE-cadherin in endothelial cells during sepsis. Sci Adv. 2022;8:eabm8965.
Giannotta M, Trani M, Dejana E. VE-cadherin and endothelial adherens junctions: active guardians of vascular integrity. Dev Cell. 2013;26:441–54.
Tiruppathi C, Wang DM, Ansari MO, Bano S, Tsukasaki Y, Mukhopadhyay A, et al. Ubiquitin ligase CHFR mediated degradation of VE-cadherin through ubiquitylation disrupts endothelial adherens junctions. Nat Commun. 2023;14:6582.
Dong WJ, He BF, Qian H, Liu Q, Wang D, Li J, et al. RAB26-dependent autophagy protects adherens junctional integrity in acute lung injury. Autophagy. 2018;14:1677–92.
Yang Y, Dong XL, Zheng SN, Sun JB, Ye J, Chen J, et al. GSTpi regulates VE-cadherin stabilization through promoting S-glutathionylation of Src. Redox Biol. 2020;30:101416.
Gong HX, Rehman J, Tang HY, Wary K, Mittal M, Chaturvedi P, et al. HIF2α signaling inhibits adherens junctional disruption in acute lung injury. J Clin Invest. 2015;125:652–64.
Dritsoula A, Camilli C, Moss SE, Greenwood J. The disruptive role of LRG1 on the vasculature and perivascular microenvironment. Front Cardiovasc Med. 2024;11:1386177.
Camilli C, Hoeh AE, De Rossi GD, Moss SE, Greenwood J. LRG1: an emerging player in disease pathogenesis. J Biomed Sci. 2022;29:6.
Shirai R, Hirano F, Ohkura N, Ikeda K, Inoue S. Up-regulation of the expression of leucine-rich alpha(2)-glycoprotein in hepatocytes by the mediators of acute-phase response. Biochem Biophys Res Commun. 2009;382:776–9.
Pang KT, Ghim M, Liu CH, Tay HM, Fhu CW, Chia RN, et al. Leucine-rich α-2-glycoprotein 1 suppresses endothelial cell activation through ADAM10-mediated shedding of TNF-α receptor. Front Cell Dev Biol. 2021;9:706143.
Wang J, Wang J, Zhong JC, Liu HB, Li WM, Chen ML, et al. LRG1 promotes atherosclerosis by inducing macrophage M1-like polarization. Proc Natl Acad Sci USA. 2024;121:e2405845121.
O’Connor MN, Kallenberg DM, Camilli C, Pilotti C, Dritsoula A, Jackstadt R, et al. LRG1 destabilizes tumor vessels and restricts immunotherapeutic potency. Med. 2021;2:1231–52.
Hisata S, Racanelli AC, Kermani P, Schreiner R, Houghton S, Palikuqi B, et al. Reversal of emphysema by restoration of pulmonary endothelial cells. J Exp Med. 2021;218:e20200938.
Li N, Wang W, Jiang WY, Xiong R, Geng Q. Cytosolic DNA-STING-NLRP3 axis is involved in murine acute lung injury induced by lipopolysaccharide. Clin Transl Med. 2020;10:e228.
Kumar S, Xu JY, Perkins C, Guo FK, Snapper S, Finkelman FD, et al. Cdc42 regulates neutrophil migration via crosstalk between WASp, CD11b, and microtubules. Blood. 2012;120:3563–74.
Xu JQ, Gao CG, He YJ, Fang XZ, Sun DY, Peng ZK, et al. NLRC3 expression in macrophage impairs glycolysis and host immune defense by modulating the NF-κB-NFAT5 complex during septic immunosuppression. Mol Ther. 2023;31:154–73.
Bi YL, Chen XY, Wei BJ, Wang LC, Gong LY, Li HM, et al. DEPTOR stabilizes ErbB2 to promote the proliferation and survival of ErbB2-positive breast cancer cells. Theranostics. 2021;11:6355–69.
Macosko EZ, Basu A, Satija R, Nemesh J, Shekhar K, Goldman M, et al. Highly parallel genome-wide expression profiling of individual cells using nanoliter droplets. Cell. 2015;161:1202–14.
Becht E, McInnes L, Healy J, Dutertre CA, Kwok IWH, Ng LG, et al. Dimensionality reduction for visualizing single-cell data using UMAP. Nat Biotechnol. 2018;37:38–44.
Hu CX, Li TY, Xu YQ, Zhang XX, Li F, Bai J, et al. CellMarker 2.0: an updated database of manually curated cell markers in human/mouse and web tools based on scRNA-seq data. Nucleic Acids Res. 2023;51:d870–6.
Jiang S, Qian QH, Zhu TT, Zong WT, Shang YF, Jin T, et al. Cell Taxonomy: a curated repository of cell types with multifaceted characterization. Nucleic Acids Res. 2023;51:d853–60.
Zhang ZR, Tang Z, Ma XW, Sun K, Fan LP, Fang J, et al. TAOK1 negatively regulates IL-17-mediated signaling and inflammation. Cell Mol Immunol. 2018;15:794–802.
Kim SS, Harford JB, Moghe M, Rait A, Pirollo KF, Chang EH. Targeted nanocomplex carrying siRNA against MALAT1 sensitizes glioblastoma to temozolomide. Nucleic Acids Res. 2018;46:1424–40.
Wang XM, Abraham S, McKenzie JAG, Jeffs N, Swire M, Tripathi VB, et al. LRG1 promotes angiogenesis by modulating endothelial TGF-β signalling. Nature. 2013;499:306–11.
Shu T, Ning WS, Wu D, Xu JQ, Han QQ, Huang MH, et al. Plasma proteomics identify biomarkers and pathogenesis of COVID-19. Immunity. 2020;53:1108–22.e5.
Dejana E, Vestweber D. The role of VE-cadherin in vascular morphogenesis and permeability control. Prog Mol Biol Transl Sci. 2013;116:119–44.
Ibrahim AFM, Shen LN, Tatham MH, Dickerson D, Prescott AR, Abidi N, et al. Antibody RING-mediated destruction of endogenous proteins. Mol Cell. 2020;79:155–66.
Yamada T, Yang Y, Bonni A. Spatial organization of ubiquitin ligase pathways orchestrates neuronal connectivity. Trends Neurosci. 2013;36:218–26.
Leclair HM, André-Grégoire G, Treps L, Azzi S, Bidère N, Gavard J. The E3 ubiquitin ligase MARCH3 controls the endothelial barrier. FEBS Lett. 2016;590:3660–8.
Seo T, Lowery AM, Xu HF, Giang W, Troyanovsky SM, Vincent PA, et al. MARCH family E3 ubiquitin ligases selectively target and degrade cadherin family proteins. PLoS One. 2024;19:e0290485.
Xu XQ, Xu TR, Ji WT, Wang C, Ren YJ, Xiong XB, et al. Herpes simplex virus 1-induced ferroptosis contributes to viral encephalitis. mBio. 2023;14:e0237022.
Li HL, Zhang YL, Rao GB, Zhang CT, Guan ZQ, Huang ZY, et al. Rift Valley fever virus coordinates the assembly of a programmable E3 ligase to promote viral replication. Cell. 2024;187:6896–913.e15.
Fukuda H, Nakamura N, Hirose S. MARCH-III is a novel component of endosomes with properties similar to those of MARCH-II. J Biochem. 2006;139:137–45.
Zeng LW, Feng L, Liu R, Lin H, Shu HB, Li S. The membrane-associated ubiquitin ligases MARCH2 and MARCH3 target IL-5 receptor alpha to negatively regulate eosinophilic airway inflammation. Cell Mol Immunol. 2022;19:1117–29.
Chathuranga K, Kim TH, Lee H, Park JS, Kim JH, Chathuranga WAG, et al. Negative regulation of NEMO signaling by the ubiquitin E3 ligase MARCH2. Embo J. 2020;39:e105139.
Schupp JC, Adams TS, Cosme C Jr, Raredon MSB, Yuan Y, Omote N, et al. Integrated single-cell atlas of endothelial cells of the human lung. Circulation. 2021;144:286–302.
Liu SL, Bi YG, Han TT, Li YE, Wang QH, Wu NN, et al. The E3 ubiquitin ligase MARCH2 protects against myocardial ischemia-reperfusion injury through inhibiting pyroptosis via negative regulation of PGAM5/MAVS/NLRP3 axis. Cell Discov. 2024;10:24.
Nakamura N. The role of the transmembrane ring finger proteins in cellular and organelle function. Membranes (Basel). 2011;1:354–93.
Xia D, Ji WL, Xu CT, Lin X, Wang XK, Xia Y, et al. Knockout of MARCH2 inhibits the growth of HCT116 colon cancer cells by inducing endoplasmic reticulum stress. Cell Death Dis. 2017;8:e2957.
Burslem GM, Crews CM. Proteolysis-targeting chimeras as therapeutics and tools for biological discovery. Cell. 2020;181:102–14.
Fan LY, Qi YQ, Yang X, Xu YR, Zhang YN, Wang LD, et al. Targeted degradation of LRG1 to attenuate renal fibrosis. Asian J Pharm Sci. 2024;19:100941.
Zhang JJ, Qi YJ, Li YZ, Zhu FR, Geng YZ, Li Y, et al. PROTAC based targeted degradation of LRG1 for mitigating corneal neovascularization. J Control Release. 2025;381:113567.
Dritsoula A, Dowsett L, Pilotti C, O’Connor MN, Moss SE, Greenwood J. Angiopathic activity of LRG1 is induced by the IL-6/STAT3 pathway. Sci Rep. 2022;12:4867.
Fu J, Wei CG, Zhang WJ, Schlondorff D, Wu JS, Cai MC, et al. Gene expression profiles of glomerular endothelial cells support their role in the glomerulopathy of diabetic mice. Kidney Int. 2018;94:326–45.
Hong Q, Cai H, Zhang L, Li ZZ, Zhong F, Ni ZH, et al. Modulation of transforming growth factor-β-induced kidney fibrosis by leucine-rich α-2 glycoprotein-1. Kidney Int. 2022;101:299–314.
Li YE, Liu SL, Wang LT, Du YX, Wu L, Chen HR, et al. March2 alleviates aortic aneurysm/dissection by regulating PKM2 polymerization. Circ Res. 2025;136:e73–93.
Hoeh AE, Chang JH, Mueller RS, Basche M, Fantin A, Sepetis A, et al. LRG1 alters pericyte phenotype and compromises vascular maturation. Cells. 2025;14:593.
Acknowledgements
This study was supported by grants from the Hubei Provincial Key Research and Development Program of China (2023BCB091, 2025BCB004), the National Science and Technology Maior Project (20232D0506504), and the National Natural Science Foundation of China (82372176, 82272217, and 82002026).
Author information
Authors and Affiliations
Contributions
XWF performed majority of the experiments. XWF and JJF conducted the in vitro experiment and helped with cell culture. YRZ and YZ performed the morphology analysis. XWF and JQX drafted the manuscript. YQ, XZ and DYZ provided technical advice. JQX and YS were responsible for concept, data collection and supervision, and reviewed and revised the draft. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Fang, Xw., Fu, Jj., Zhang, Yr. et al. Intracellular LRG1 recruits MARCH2 to ubiquitinate and degrade endothelial VE-cadherin in septic lung injury. Acta Pharmacol Sin (2026). https://doi.org/10.1038/s41401-026-01804-x
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41401-026-01804-x