Abstract
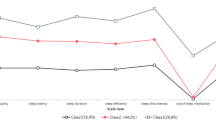
Sleep disruption has received little attention in hematopoietic cell transplantation (HCT). The goal of this study was to describe severity, course, and predictors of sleep disruption following HCT. A secondary data analysis was conducted of the Blood and Marrow Transplantation Clinical Trials Network (BMT CTN) 0902 study. Participants completed a modified version of the Pittsburgh Sleep Quality Index prior to transplant and 100 and 180 days posttransplant. Growth mixture models were used to characterize subgroups of patients based on baseline sleep disruption and change over time. A total of 570 patients (mean age 55 years, 42% female) were included in the current analyses. Patients could be grouped into four distinct classes based on sleep disruption: (1) clinically significant sleep disruption at baseline that did not improve over time (20%); (2) clinically significant sleep disruption at baseline that improved over time (22%); (3) sleep disruption that did not reach clinical significance at baseline and did not improve over time (45%); and (4) no sleep disruption at baseline or over time (13%). These data provide a more comprehensive understanding of sleep disruption that can be used to develop interventions to improve sleep in HCT recipients.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
American Psychiatric Association. Diagnostic and statistical manual of mental disorders. 5th ed. Washington D.C.: American Psychiatric Association; 2013.
Bevans MF, Mitchell SA, Marden S. The symptom experience in the first 100 days following allogeneic hematopoietic stem cell transplantation (HSCT). Support Care Cancer. 2008;16:1243–54.
Miller MF, Karten C, Longacre M, LeBlanc TW. Symptom burden and palliative care for patients with multiple myeloma: Cancer Experience Registry findings. J Clin Oncol. 2015;33:81. 29_suppl
Lee SJ, Joffe S, Kim HT, Socie G, Gilman AL, Wingard JR, et al. Physicians’ attitudes about quality-of-life issues in hematopoietic stem cell transplantation. Blood. 2004;104:2194–200.
Jim HS, Evans B, Jeong JM, Gonzalez BD, Johnston L, Nelson AM, et al. Sleep disruption in hematopoietic cell transplantation recipients: prevalence, severity, and clinical management. Biol Blood Marrow Transplant. 2014;20:1465–84.
McQuellon RP, Russell GB, Rambo TD, Craven BL, Radford J, Perry JJ, et al. Quality of life and psychological distress of bone marrow transplant recipients: the ‘time trajectory’ to recovery over the first year. Bone Marrow Transplant. 1998;21:477–86.
Aaronson NK, Ahmedzai S, Bergman B, Bullinger M, Cull A, Duez NJ, et al. The European Organization for Research and Treatment of Cancer QLQ-C30: a quality-of-life instrument for use in international clinical trials in oncology. J Natl Cancer Inst. 1993;85:365–76.
Bieri S, Roosnek E, Helg C, Verholen F, Robert D, Chapuis B, et al. Quality of life and social integration after allogeneic hematopoietic SCT. Bone Marrow Transplant. 2008;42:819–27.
Grulke N, Albani C, Bailer H. Quality of life in patients before and after haematopoietic stem cell transplantation measured with the European Organization for Research and Treatment of Cancer (EORTC) Quality of Life Core Questionnaire QLQ-C30. Bone Marrow Transplant. 2012;47:473–82.
Faulhaber GA, Furlanetto TW, Astigarraga CC, Moser Filho HL, Paludo AP, Silla LM. Association of busulfan and cyclophosphamide conditioning with sleep disorders after hematopoietic stem cell transplantation. Acta Haematol. 2010;124:125–8.
Boonstra L, Harden K, Jarvis S, Palmer S, Kavanaugh-Carveth P, Barnett J, et al. Sleep disturbance in hospitalized recipients of stem cell transplantation. Clin J Oncol Nurs. 2011;15:271–6.
Rischer J, Scherwath A, Zander AR, Koch U, Schulz-Kindermann F. Sleep disturbances and emotional distress in the acute course of hematopoietic stem cell transplantation. Bone Marrow Transplant. 2009;44:121–8.
Gallicchio L, Kalesan B. Sleep duration and mortality: a systematic review and meta-analysis. J Sleep Res. 2009;18:148–58.
Cappuccio FP, D’Elia L, Strazzullo P, Miller MA. Sleep duration and all-cause mortality: a systematic review and meta-analysis of prospective studies. Sleep. 2010;33:585–92.
Guariniello LD, Vicari P, Lee KS, de Oliveira AC, Tufik S. Bone marrow and peripheral white blood cells number is affected by sleep deprivation in a murine experimental model. J Cell Physiol. 2012;227:361–6.
Liu L, Mills PJ, Rissling M, Fiorentino L, Natarajan L, Dimsdale JE, et al. Fatigue and sleep quality are associated with changes in inflammatory markers in breast cancer patients undergoing chemotherapy. Brain Behav Immun. 2012;26:706–13.
Irwin MR, Olmstead R, Carroll JE. Sleep disturbance, sleep duration, and inflammation: a systematic review and meta-analysis of cohort studies and experimental sleep deprivation. Biol Psychiatry. 2016;80:40–52.
Nelson AM, Coe CL, Juckett MB, Rumble ME, Rathouz PJ, Hematti P, et al. Sleep quality following hematopoietic stem cell transplantation: longitudinal trajectories and biobehavioral correlates. Bone Marrow Transplant. 2014;49:1405–11.
Muthén B. Latent variable analysis: growth mixture modeling and related techniques for longitudinal data. In: Kaplan D, editor. The Sage handbook of quantitative methodology for the social sciences. Thousand Oaks CA: Sage; 2004. p. 345–68.
Ram N, Grimm KJ. Growth mixture modeling: a method for identifying differences in longitudinal change among unobserved groups. Int J Behav Dev. 2009;33:565–76.
Jacobsen PB, Le-Rademacher J, Jim H, Syrjala K, Wingard JR, Logan B, et al. Exercise and stress management training prior to hematopoietic cell transplantation: Blood and Marrow Transplant Clinical Trials Network (BMT CTN) 0902. Biol Blood Marrow Transplant. 2014;20:1530–6.
Sorror ML, Maris MB, Storb R, Baron F, Sandmaier BM, Maloney DG, et al. Hematopoietic cell transplantation (HCT)-specific comorbidity index: a new tool for risk assessment before allogeneic HCT. Blood. 2005;106:2912–9.
Buysse DJ, Reynolds CF 3rd, Monk TH, Berman SR, Kupfer DJ. The Pittsburgh Sleep Quality Index: a new instrument for psychiatric practice and research. Psychiatry Res. 1989;28:193–213.
Phillips KM, Jim HS, Donovan KA, Pinder-Schenck MC, Jacobsen PB. Characteristics and correlates of sleep disturbances in cancer patients. Support Care Cancer. 2012;20:357–65.
Syrjala KL, Chapko ME. Evidence for a biopsychosocial model of cancer treatment-related pain. Pain. 1995;61:69–79.
Syrjala KL, Chapko MK, Vitaliano PP, Cummings C, Sullivan KM. Recovery after allogeneic marrow transplantation: prospective study of predictors of long-term physical and psychosocial functioning. Bone Marrow Transplant. 1993;11:319–27.
Ware JE, Snow KK, Kosinski M, Gandek B. SF-36 health survey: manual and interpretation guide. Boston: The Health Institute; 1993.
Jacobsen PB, Phillips KM, Jim HS, Small BJ, Faul LA, Meade CD, et al. Effects of self-directed stress management training and home-based exercise on quality of life in cancer patients receiving chemotherapy: a randomized controlled trial. Psychooncology. 2013;22:1229–35.
Krischer MM, Xu P, Meade CD, Jacobsen PB. Self-administered stress management training in patients undergoing radiotherapy. J Clin Oncol. 2007;25:4657–62.
Muthén LK, Muthén B. Mplus User’s Guide. 6th ed. Los Angeles, CA: Muthén & Muthén; 2007.
Altmaier EM, Ewell M, McQuellon R, Geller N, Carter SL, Henslee-Downey J, et al. The effect of unrelated donor marrow transplantation on health-related quality of life: a report of the unrelated donor marrow transplantation trial (T-cell depletion trial). Biol Blood Marrow Transplant. 2006;12:648–55.
Palesh OG, Roscoe JA, Mustian KM, Roth T, Savard J, Ancoli-Israel S, et al. Prevalence, demographics, and psychological associations of sleep disruption in patients with cancer: University of Rochester Cancer Center-Community Clinical Oncology Program. J Clin Oncol. 2010;28:292–8.
Savard J, Villa J, Ivers H, Simard S, Morin CM. Prevalence, natural course, and risk factors of insomnia comorbid with cancer over a 2-month period. J Clin Oncol. 2009;27:5233–9.
Hjermstad MJ, Knobel H, Brinch L, Fayers PM, Loge JH, Holte H, et al. A prospective study of health-related quality of life, fatigue, anxiety and depression 3-5 years after stem cell transplantation. Bone Marrow Transplant. 2004;34:257–66.
Cohen MZ, Rozmus CL, Mendoza TR, Padhye NS, Neumann J, Gning I, et al. Symptoms and quality of life in diverse patients undergoing hematopoietic stem cell transplantation. J Pain Symptom Manag. 2012;44:168–80.
Syrjala KL, Sutton SK, Jim HS, Knight JM, Wood WA, Lee SJ, et al. Cancer and treatment distress psychometric evaluation over time: a BMT CTN 0902 secondary analysis. Cancer. 2017;123:1416–23.
Jim HS, Sutton SK, Small BJ, Jacobsen PB, Wood WA, Knight JM, et al. Trajectories of quality of life after hematopoietic cell transplantation: Secondary analysis of Blood and Marrow Transplant Clinical Trials Network 0902 data. Biol Blood Marrow Transplant. 2016;22:2077–83.
Denlinger CS, Sanft T, Baker KS, Baxi S, Broderick G, Demark-Wahnefried W. et al. Survivorship, version 2.2017, NCCN Clinical Practice Guidelines in Oncology. J Natl Compr Cancer Netw. 2017;15:1140–63.
Acknowledgements
We are grateful for the investigators and patients who participated in this trial.
Funding
Support for this study was provided by grant U10HL069294 to the Blood and Marrow Transplant Clinical Trials Network from the National Heart, Lung, and Blood Institute (NHLBI) and the National Cancer Institute (NCI). Additional support was provided by the National Center for Advancing Translational Sciences [National Institutes of Health (NIH)] through grant numbers UL1TR001436 and KL2TR001438. KLS is supported in part by NCI R01 CA215134. The content is solely the responsibility of the authors and does not necessarily represent the official views of the above mentioned parties. This work was performed while PBJ was at the Moffitt Cancer Center and does not represent the views of NIH.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
HSLJ is a consultant for RedHill Biopharma. The other authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Jim, H.S.L., Sutton, S., Majhail, N. et al. Severity, course, and predictors of sleep disruption following hematopoietic cell transplantation: a secondary data analysis from the BMT CTN 0902 trial. Bone Marrow Transplant 53, 1038–1043 (2018). https://doi.org/10.1038/s41409-018-0138-0
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41409-018-0138-0
This article is cited by
-
The mutual impacts of stem cells and sleep: opportunities for improved stem cell therapy
Stem Cell Research & Therapy (2025)
-
The mediating role of health anxiety, resilience, and body image in the relationship between pain and sleep in hematopoietic stem cell transplantation patients
BMC Psychology (2025)
-
Life 2.0: a comprehensive cross-sectional profiling of long-term allogeneic hematopoietic cell transplantation survivors compared to a matched general population cohort
Bone Marrow Transplantation (2025)
-
The level of fatigue, insomnia, depression, anxiety, stress, and the relationship between these symptoms following allogeneic hematopoietic stem cell transplantation: a cross-sectional study
Supportive Care in Cancer (2023)