Abstract
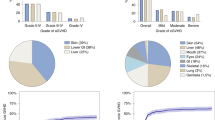
Glucocorticoids for primary therapy of acute GVHD have limited responses. A phase I/II trial tested 4 weeks of deacetylase inhibitor panobinostat started within 48 h of glucocorticoids (1 mg/kg/day prednisone or equivalent) as primary treatment for patients with either classic acute GVHD (n = 16) or acute GVHD overlapping with chronic (n = 6). Four patients received 2.5 mg/m2 IV three times a week (TIW). Subsequent to discontinuation of IV panobinostat, patients received oral doses (PO). Two patients treated with 10 mg TIW (PO level 1) had progressive GVHD, after which patients were treated with 5 mg TIW (PO level −1; n = 16); 31/41 adverse events were possibly related, including thrombocytopenia (n = 13), leukopenia (n = 7), hypercholesterolemia (n = 3), hypertriglyceridemia (n = 5), anemia (n = 1), fatigue (n = 1), and hepatobiliary disorder (n = 1). GVHD responses were complete (n = 12) or partial (n = 3), with 1 progression at PO level −1. T-regulatory cells increased at day 8, CD4/CD8 and monocytes exhibited enhanced H3 acetylation, and CD4 or CD8 numbers remained unchanged with a decreased interleukin 12p40 plasma level. Panobinostat in combination with prednisone is safe and warrants further testing in GVHD.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Cutler CS, Koreth J, Ritz J. Mechanistic approaches for the prevention and treatment of chronic GVHD. Blood. 2017;129:22–29. https://doi.org/10.1182/blood-2016-08-686659.
Nassereddine S, Rafei H, Elbahesh E, Tabbara I. Acute graft versus host disease: a comprehensive review. Anticancer Res. 2017;37:1547–55.
Martin PJ, Rizzo JD, Wingard JR, Ballen K, Curtin PT, Cutler C, et al. First and second-line systemic treatment of acute graft-versus-host disease: recommendations of the American Society of Blood and Marrow Transplantation. Biol Blood Marrow Transplant. 2012;18:1150–63. https://doi.org/10.1016/j.bbmt.2012.04.005.
Chen Y-B, Perales M-A, Li S, Kempner M, Reynolds C, Brown J, et al. Phase I multicenter trial of brentuximab vedotin for steroid refractory acute graft-vs.-host disease (GVHD). Blood 2017. https://doi.org/10.1182/blood-2017-03-772210
Miklos D, Cutler CS, Arora M, Waller EK, Jagasia M, Pusic I, et al. Multicenter open-label phase 2 study of ibrutinib in chronic graft versus host disease (cgvhd) after failure of corticosteroids. Blood 2016;128:LBA-3.
Zeiser R, Burchert A, Lengerke C, Verbeek M, Maas-Bauer K, Metzelder SK, et al. Ruxolitinib in corticosteroid-refractory graft-versus-host disease after allogeneic stem cell transplantation: a multi-center survey. Leukemia. 2015;29:2062–8. https://doi.org/10.1038/leu.2015.212
Koreth J, Kim HT, Jones KT, Lange PB, Reynolds CG, Chammas MJ, et al. Efficacy, durability, and response predictors of low-dose interleukin-2 therapy for chronic graft-versus-host disease. Blood. 2016;128:130–7. https://doi.org/10.1182/blood-2016-02-702852.
Cutler C, Miklos D, Kim HT, Treister N, Woo S-B, Bienfang D, et al. Rituximab for steroid-refractory chronic graft-versus-host disease. Blood. 2006;108:756–62. https://doi.org/10.1182/blood-2006-01-0233.
Chen R, Chen B, Dreger P, Schmitt M, Schmitt A. Extracorporeal photopheresis for steroid-refractory chronic graft-versus-host disease after allogeneic hematopoietic stemcell transplantation: a systematic review and meta-analysis. Cancer Transl Med. 2015;1:201–8. https://doi.org/10.4103/2395-3977.172859.
Alousi AM, Weisdorf DJ, Logan BR, Bolaños-Meade J, Carter S, DiFronzo N, et al. Etanercept, mycophenolate, denileukin, or pentostatin plus corticosteroids for acute graft-versus-host disease: a randomized phase 2 trial from the Blood and Marrow Transplant Clinical Trials Network. Blood. 2009;114:511–7. https://doi.org/10.1182/blood-2009-03-212290.
Leoni F, Zaliani A, Bertolini G, Porro G, Pagani P, Pozzi P, et al. The antitumor histone deacetylase inhibitor suberoylanilide hydroxamic acid exhibits antiinflammatory properties via suppression of cytokines. Proc Natl Acad Sci USA. 2002;99:2995–3000.
Mishra N, Reilly CM, Brown DR, Ruiz P, Gilkeson GS. Histone deacetylase inhibitors modulate renal disease in the MRL-lpr/lpr mouse. J Clin Invest. 2003;111:539–52.
Tao R, de Zoeten EF, Ozkaynak E, Chen C, Wang L, Porrett PM, et al. Deacetylase inhibition promotes the generation and function of regulatory T cells. Nat Med. 2007;13:1299–307.
Nencioni A, Beck J, Werth D, Grunebach F, Patrone F, Ballestrero A, et al. Histone deacetylase inhibitors affect dendritic cell differentiation and immunogenicity. Clin Cancer Res. 2007;13:3933–41. https://doi.org/10.1158/1078-0432.ccr-06-2903.
Minucci S, Pelicci PG. Histone deacetylase inhibitors and the promise of epigenetic (and more) treatments for cancer. Nat Rev Cancer. 2006;6:38–51.
Marks PA. Discovery and development of SAHA as an anticancer agent. Oncogene. 2007;26:1351–6.
Reddy P, Maeda Y, Hotary K, Liu C, Reznikov LL, Dinarello CA, et al. Histone deacetylase inhibitor suberoylanilide hydroxamic acid reduces acute graft-versus-host disease and preserves graft-versus-leukemia effect. Proc Natl Acad Sci USA. 2004;101:3921–6.
Leng C, Gries M, Ziegler J, Lokshin A, Mascagni P, Lentzsch S, et al. Reduction of graft-versus-host disease by histone deacetylase inhibitor suberonylanilide hydroxamic acid is associated with modulation of inflammatory cytokine milieu and involves inhibition of STAT1. Exp Hematol. 2006;34:776–87.
Choi SW, Braun T, Chang L, Ferrara JLM, Pawarode A, Magenau JM, et al. Vorinostat plus tacrolimus and mycophenolate to prevent graft-versus-host disease after related-donor reduced-intensity conditioning allogeneic haemopoietic stem-cell transplantation: a phase 1/2 trial. Lancet Oncol. 2014;15:87–95.
Choi SW, Gatza E, Hou G, Sun Y, Whitfield J, Song Y, et al. Histone deacetylase inhibition regulates inflammation and enhances Tregs after allogeneic hematopoietic cell transplantation in humans. Blood. 2015;125:815–9. https://doi.org/10.1182/blood-2014-10-605238.
Choi SW, Braun T, Henig I, Gatza E, Magenau J, Parkin B, et al. Vorinostat plus tacrolimus/methotrexate to prevent GVHD following myeloablative conditioning unrelated donor HCT. Blood 2017. https://doi.org/10.1182/blood-2017-06-790469
San-Miguel JF, Hungria VTM, Yoon S-S, Beksac M, Dimopoulos MA, Elghandour A, et al. Panobinostat plus bortezomib and dexamethasone versus placebo plus bortezomib and dexamethasone in patients with relapsed or relapsed and refractory multiple myeloma: a multicentre, randomised, double-blind phase 3 trial. Lancet Oncol. 2014;15:1195–206. https://doi.org/10.1016/S1470-2045(14)70440-1.
Richardson PG, Laubach JP, Lonial S, Moreau P, Yoon S-S, Hungria VTM, et al. Panobinostat: a novel pan-deacetylase inhibitor for the treatment of relapsed or relapsed and refractory multiple myeloma. Expert Rev Anticancer Ther. 2015;15:737–48. https://doi.org/10.1586/14737140.2015.1047770.
Wang D, Iclozan C, Liu C, Xia C, Anasetti C, Yu X-Z. LBH589 enhances T-cell activation in vivo and accelerates graft-versus-host disease in mice. Biol Blood Marrow Transplant. 2012;18:1182. 10.1016/j.bbmt.2012.06.002.e1181.
Bug G, Burchert A, Wagner E-M, Kroeger N, Jedlickova Z, Gueller S. et al. Phase I/II study of the deacetylase inhibitor panobinostat as maintenance therapy after an allogeneic stem cell transplantation in patients with high-risk MDS or AML: the Panobest-Trial. Blood. 2015;126:4344.
Cornelissen JJ, van Norden Y, van Gelder M, Breems DA, Maertens J, Jongen-Lavrencic M. et al. Early post-transplant epigenetic therapy by panobinostat and decitabine followed by donor lymphocyte infusion (DLI): interim results of the HOVON-116 Phase I/II Feasibility Study in poor-risk AML recipients of allogeneic stem cell transplantation (alloHSCT). Blood. 2016;128:832.
Tawara I, Koyama M, Liu C, Toubai T, Thomas D, Evers R, et al. Interleukin-6 modulates graft-versus-host responses after experimental allogeneic bone marrow transplantation. Clin Cancer Res. 2011;17:77–88. https://doi.org/10.1158/1078-0432.CCR-10-1198.
Song W, Tai YT, Tian Z, Hideshima T, Chauhan D, Nanjappa P, et al. HDAC inhibition by LBH589 affects the phenotype and function of human myeloid dendritic cells. Leukemia. 2011;25:161–8. http://www.nature.com/leu/journal/v25/n1/suppinfo/leu2010244s1.html.
Przepiorka D, Weisdorf D, Martin P, Klingemann HG, Beatty P, Hows J. et al. 1994 Consensus Conference on Acute GVHD Grading. Bone Marrow Transplant. 1995;15:825–8.
Filipovich AH, Weisdorf D, Pavletic S, Socie G, Wingard JR, Lee SJ, et al. National Institutes of Health Consensus Development Project on Criteria for Clinical Trials in Chronic Graft-versus-Host Disease: I. Diagnosis and Staging Working Group Report. Biol Blood Marrow Transplant. 2005;11:945–56. https://doi.org/10.1016/j.bbmt.2005.09.004
Martin P, Schoch G, Fisher L, Byers V, Anasetti C, Appelbaum F, et al. A retrospective analysis of therapy for acute graft-versus-host disease: initial treatment. Blood. 1990;76:1464–72.
Bacigalupo A, Ballen K, Rizzo D, Giralt S, Lazarus H, Ho V, et al. Defining the Intensity of Conditioning Regimens: Working Definitions. Biol Blood Marrow Transplant. 2009;15:1628–33. https://doi.org/10.1016/j.bbmt.2009.07.004.
DeAngelo DJ, Spencer A, Bhalla KN, Prince HM, Fischer T, Kindler T. et al. Phase Ia/II, two-arm, open-label, dose-escalation study of oral panobinostat administered via two dosing schedules in patients with advanced hematologic malignancies. Leukemia. 2013;27:1628–36. https://doi.org/10.1038/leu.2013.38.
Khot A, Dickinson M, Prince HM. Panobinostat in lymphoid and myeloid malignancies. Expert Opin Investig Drugs. 2013;22:1211–23. https://doi.org/10.1517/13543784.2013.815165.
Levine JE, Logan B, Wu J, Alousi AM, Ho V, Bolaños-Meade J, et al. Graft-versus-host disease treatment: predictors of survival. Biol Blood Marrow Transplant. 2010;16:1693–9. https://doi.org/10.1016/j.bbmt.2010.05.019.
MacMillan ML, Weisdorf DJ, Wagner JE, DeFor TE, Burns LJ, Ramsay NK, et al. Response of 443 patients to steroids as primary therapy for acute graft-versus-host disease: comparison of grading systems. Biol Blood Marrow Transplant. 2002;8:387–94.
Martin PJ, Bachier CR, Klingemann H-G, McCarthy PL, Szabolcs P, Uberti JP, et al. Endpoints for clinical trials testing treatment of acute graft-versus-host disease: a joint statement. Biol Blood Marrow Transplant. 2009;15:777–84. https://doi.org/10.1016/j.bbmt.2009.03.012.
Welniak LA, Blazar BR, Wiltrout RH, Anver MR, Murphy WJ. Role of interleukin-12 in acute graft-versus-host disease(1). Transplant Proc. 2001;33:1752–3.
Zhao Z, Xie F, Zhang X, Yang J, Wang R, Yang R, et al. Update on the association between interleukin-12 p40 gene polymorphism and risk of psoriasis: a meta-analysis. Dermatol Sin. 2016;34:126–30. https://doi.org/10.1016/j.dsi.2016.01.001.
Acknowledgements
We thank Erika Elmer, Michele Burton, Jermaine Castro, and Carmen Patton for trial coordination/data acquisitions. We thank Sonya Smyk (Moffitt Cancer Center) for editorial assistance. Novartis Pharmaceuticals Corporation supported this trial and provided PANO.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Perez, L., Fernandez, H., Horna, P. et al. Phase I trial of histone deacetylase inhibitor panobinostat in addition to glucocorticoids for primary therapy of acute graft-versus-host disease. Bone Marrow Transplant 53, 1434–1444 (2018). https://doi.org/10.1038/s41409-018-0163-z
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41409-018-0163-z