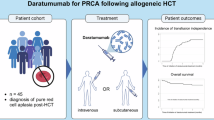
Abstract
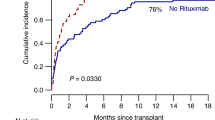
Pure red cell aplasia (PRCA) and autoimmune hemolytic anemia (AIHA) post-hematopoietic stem cell transplantation (HSCT) are an unmet medical need with no established standard of care, significantly affecting the patient quality of life and posing a challenge for clinicians. The anti-CD38 IgG-kappa Daratumumab appears to be a safe and efficace treatment compared to prior drugs. Our study is a prospective monocentric investigation assessing the use of daratumumab in these complications following allo-HSCT. Here we describe our experience on six patients with a median age of 65 years. All treated patients, except one, who died because of sepsis during GVHD exacerbation, reached transfusion independence with erythropoietin suspension. Poor graft function remains a management challenge for clinicians and has a significant impact on the patient’s quality of life. Currently, therapeutic options for PRCA and for the least common AIHA, appear ineffective, making it difficult to address the diverse needs of post-transplant patients. Although our data and those previously reported in the literature are preliminary, daratumumab prompts further reflection on its use in this setting of patients.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Marco-Ayala J, Gómez-Seguí I, Sanz G, Solves P. Pure red cell aplasia after major or bidirectional ABO-incompatible hematopoietic stem cell transplantation: to treat or not to treat, that is the question. Bone Marrow Transpl. 2021;56:769–78.
Bolan CD, Leitman SF, Griffith LM, Wesley RA, Procter JL, Stroncek DF, et al. Delayed donor red cell chimerism and pure red cell aplasia following major ABO-incompatible nonmyeloablative hematopoietic stem cell transplantation. Blood. 2001;98:1687–94.
González-Vicent M, Sanz J, Fuster JL, Cid J, de Heredia CD, Morillo D, et al. Autoimmune hemolytic anemia (AIHA) following allogeneic hematopoietic stem cell transplantation (HSCT): A retrospective analysis and a proposal of treatment on behalf of the Grupo Español De Trasplante de Medula Osea en Niños (GETMON) and the Grupo Español de Trasplante Hematopoyetico (GETH). Transfus Med Rev. 2018;32:179–85.
Salas MQ, Alahmari A, Lipton JH. Successful treatment of refractory red cell aplasia after allogeneic hematopoietic cell transplantation with daratumumab. Eur J Haematol 2020;104:145–7.
DeZern AE, Franklin C, Tsai HL, Hollingsworth Imus P, Cooke KR, Varadhan R, et al. Relationship of donor age and relationship to outcomes of haploidentical transplantation with posttransplant cyclophosphamide. Blood Adv. 2021;5:1360–8.
Zhu P, Wu Y, Cui D, Shi J, Yu J, Zhao Y, et al. Prevalence of pure red cell aplasia following major ABO-incompatible hematopoietic stem cell transplantation. Front Immunol. 2022;13:829670.
Means RT Jr. Pure red cell aplasia: the second hundred years. Am J Med Sci. 2023;366:160–6.
Ozkurt ZN, Yegin ZA, Yenicesu I, Aki SZ, Yagci M, Sucak GT. Impact of ABO-incompatible donor on early and late outcome of hematopoietic stem cell transplantation. Transpl Proc. 2009;41:3851–8.
Tekgündüz SA, Özbek N. ABO blood group mismatched hematopoietic stem cell transplantation. Transfus Apher Sci. 2016;54:24–9.
Pang Y, Kalsi SS, Childs R, Arai S. Post-hematopoietic stem cell transplantation immune-mediated anemia: a literature review and novel therapeutics. Blood Adv. 2022;6:2707–21.
Watz E, Remberger M, Ringden O, Lundahl J, Ljungman P, Mattsson J, et al. Analysis of donor and recipient ABO incompatibility and antibody-associated complications after allogeneic stem cell transplantation with reduced-intensity conditioning. Biol Blood Marrow Transpl. 2014;20:264–71.
Liu M, Zhang T, Dong X, Wang H. Acquired pure red cell aplasia: unraveling the immune pathogenesis. J Bio-X Res. 2023;6:138–48.
Migdady Y, Pang Y, Kalsi SS, Childs R, Arai S. Post-hematopoietic stem cell transplantation immune-mediated anemia: a literature review and novel therapeutics. Blood Adv. 2022;6:2707–21.
Zhang L, Chen N, Xu Z, Liang Q, Pan H, Zhao J, et al. Good treatment-free survival of monoclonal gammopathy of undetermined significance associated pure red cell aplasia after bortezomib plus dexamethasone. Blood Cells Mol Dis. 2021;89:102573.
Sackett K, Cohn CS, Fahey-Ahrndt K, Smith AR, Johnson AD. Successful treatment of pure red cell aplasia because of ABO major mismatched stem cell transplant. J Clin Apher. 2018;33:108–12.
Jeyaraman P, Borah P, Rajput P, Dayal N, Pathak S, Naithani R. Daratumumab for pure red cell aplasia post ABO incompatible allogeneic hematopoietic stem cell transplant for aplastic anemia. Blood Cells Mol Dis. 2021;88:102464.
Chang TY, Jaing TH, Wen YC, Huang IA, Chen SH, Tsay PK. Risk factor analysis of autoimmune hemolytic anemia after allogeneic hematopoietic stem cell transplantation in children. Med (Baltim). 2016;95:e5396.
Li Z, Rubinstein SM, Thota R, Savani M, Brissot E, Shaw BE, et al. Immune-mediated complications after hematopoietic stem cell transplantation. Biol Blood Marrow Transpl. 2016;22:1368–75.
Gabelli M, Ademokun C, Cooper N, Amrolia PI. Pathogenesis, risk factors and therapeutic options for autoimmune haemolytic anaemia in the post-transplant setting. Br J Haematol. 2022;196:45–62.
Robillard N, Wuillème S, Moreau P, Béné MC. Immunophenotype of normal and myelomatous plasma-cell subsets. Front Immunol. 2014;5:137.
Martino R, García-Cadenas I, Esquirol A. Daratumumab may be the most effective treatment for post-engraftment pure red cell aplasia due to persistent anti-donor isohemagglutinins after major ABO-mismatched allogeneic transplantation. Bone Marrow Transpl. 2022;57:282–5.
Chapuy CI, Kaufman RM, Alyea EP, Connors JM. Daratumumab for delayed red-cell engraftment after allogeneic transplantation. N. Engl J Med. 2018;379:1846–50.
Gangat N, Bleeker J, Lynch D, Olteanu H, Letendre L, Tefferi A. Daratumumab for treatment-refractory acquired idiopathic pure red cell aplasia. Haematologica. 2022;107:2523–6.
Weverling FG, Roeven MWH, Nijssen KK, Broers AEC, Dovern E, Van Rhenen A, et al. Efficacy and safety of daratumumab in pure red cell aplasia after allogeneic transplantation: Dutch real-world data. Blood Adv. 2024;8:1683–6.
Schuetz C, Hoenig M, Moshous D, Weinstock C, Castelle M, Bendavid M, et al. Daratumumab in life-threatening autoimmune hemolytic anemia following hematopoietic stem cell transplantation. Blood Adv. 2018;2:2550.
Yang EH, Muhsen IN, Samarkandi H, El Fakih R, Aljurf M, Hanbali A. Role of anti-CD38 monoclonal antibodies in the treatment of adult immune hematological diseases. Hematol Oncol Stem Cell Ther. 2023;17:4–12.
Acknowledgements
A thank to JANSSEN for providing drugs for the treatment of these patients as compassionate use.
Author information
Authors and Affiliations
Contributions
SG, MAL and LDM collected and revised data and wrote the paper, EM was involved in the management of patients, LT is responsible of the cell manipulation center, SS and PC revised the manuscript
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Giammarco, S., Limongiello, M.A., Di Marino, L. et al. The role of daratumumab in complications post-allogeneic hematopoietic stem cell transplantation: a single-center prospective study on PRCA and AIHA. Bone Marrow Transplant 60, 305–309 (2025). https://doi.org/10.1038/s41409-024-02479-w
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41409-024-02479-w