Abstract
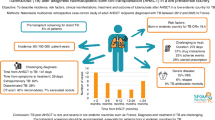
Tuberculosis (TB) is rare following hematopoietic cell transplantation (HCT). In this multinational retrospective study, we report the frequency, characteristics, and outcome of TB following HCT performed during 2000–2019. Fifty-two patients (35 (67%) males, 15 (29%) children) from 24 centers developed TB following allogeneic (n = 47) or autologous (n = 5) HCT; with the relative frequency of 0.21% and 0.025%, respectively. Forty (77%) were bacteriologically, 12 (23%) clinically confirmed. The median time from HCT to TB was 135 (range, 16–3225) days. Eighteen (35%) patients with extrapulmonary TB (mainly involving lymph nodes and liver/spleen) were significantly younger, developed TB shorter after HCT, more often had inherited underlying disease, and received immunosuppressive therapy at TB diagnosis as compared to pulmonary TB. Five (22%) of 23 patients with drug-susceptibility testing performed, were resistant to at least one anti-TB drug. Treatment success was achieved in 38/50 (76%) of treated patients. One-year overall survival reached 75.7% and the 1-year cumulative incidence of TB-associated death was 18.1%. Concluding, TB is a rare, albeit severe complication, which can develop any time after HCT, frequently involves extrapulmonary sites, and results in high mortality rates. High proportion of drug-resistant TB warrants routine susceptibility testing.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Behr MA, Kaufmann E, Duffin J, Edelstein PH, Ramakrishnan L. Latent tuberculosis: two centuries of confusion. Am J Respir Crit Care Med 2021;204:142–8.
Bergeron A, Mikulska M, De Greef J, Bondeelle L, Franquet T, Herrmann J-L, et al. Mycobacterial infections in adults with haematological malignancies and haematopoietic stem cell transplants: guidelines from the 8th European Conference on Infections in Leukaemia. Lancet Infect Dis. 2022;22:e359-e369.
Hyun J, Lee M, Jung I, Kim E, Hahn SM, Kim YR, et al. Changes in tuberculosis risk after transplantation in the setting of decreased community tuberculosis incidence: a national population-based study, 2008-2020. Ann Clin Microbiol Antimicrob 2024;23:1.
Cordonnier C, Martino R, Trabasso P, Held TK, Akan H, Ward MS, et al. Mycobacterial infection: a difficult and late diagnosis in stem cell transplant recipients. Clin Infect Dis 2004;38:1229–36.
de la Cámara R, Martino R, Granados E, Rodriguez-Salvanés FJ, Rovira M, Cabrera R, et al. Tuberculosis after hematopoietic stem cell transplantation: incidence, clinical characteristics and outcome. Spanish Group on Infectious Complications in Hematopoietic Transplantation. Bone Marrow Transplant 2000;26:291–8.
Lee HJ, Lee DG, Choi SM, Park SH, Cho SY, Choi JK, et al. The demanding attention of tuberculosis in allogeneic hematopoietic stem cell transplantation recipients: High incidence compared with general population. PLoS One 2017;12:e0173250.
Zeng QZ, Zhang YY, Wu YJ, Zhang ZY, Zhang JN, Fu HX, et al. Frequency, risk factors, and outcome of active tuberculosis following allogeneic hematopoietic stem cell transplantation. Biol Blood Marrow Transplant 2020;26:1203–9.
de Oliveira Rodrigues M, de Almeida Testa LH, Dos Santos ACF, Zanetti LP, da Silva Ruiz L, de Souza MP, et al. Latent and active tuberculosis infection in allogeneic hematopoietic stem cell transplant recipients: a prospective cohort study. Bone Marrow Transplant 2021;56:2241–7.
Kapoor J, Mirgh SP, Khushoo V, Mehta P, Ahmed R, Bansal N, et al. Study of clinical characteristics, risk factors and outcomes for tuberculosis post allogeneic stem cell transplant: never count it out. Ther Adv Infect Dis 2021;8:20499361211008674.
Ip MS, Yuen KY, Woo PC, Luk WK, Tsang KW, Lam WK, et al. Risk factors for pulmonary tuberculosis in bone marrow transplant recipients. Am J Respir Crit Care Med 1998;158:1173–7.
Yoo JW, Jo KW, Kim SH, Lee SO, Kim JJ, Park SK, et al. Incidence, characteristics, and treatment outcomes of mycobacterial diseases in transplant recipients. Transpl Int 2016;29:549–58.
Roy V, Weisdorf D. Mycobacterial infections following bone marrow transplantation: a 20 year retrospective review. Bone Marrow Transplant 1997;19:467–70.
Ku SC, Tang JL, Hsueh PR, Luh KT, Yu CJ, Yang PC. Pulmonary tuberculosis in allogeneic hematopoietic stem cell transplantation. Bone Marrow Transplant 2001;27:1293–7.
Erdstein AA, Daas P, Bradstock KF, Robinson T, Hertzberg MS. Tuberculosis in allogeneic stem cell transplant recipients: still a problem in the 21st century. Transpl Infect Dis 2004;6:142–6.
Aljurf M, Gyger M, Alrajhi A, Sahovic E, Chaudhri N, Musa M, et al. Mycobacterium tuberculosis infection in allogeneic bone marrow transplantation patients. Bone Marrow Transplant 1999;24:551–4.
Agrawal N, Aggarwal M, Kapoor J, Ahmed R, Shrestha A, Kaushik M, et al. Incidence and clinical profile of tuberculosis after allogeneic stem cell transplantation. Transpl Infect Dis. 2018;20.
Lee J, Lee MH, Kim WS, Kim K, Park SH, Lee SH, et al. Tuberculosis in hematopoietic stem cell transplant recipients in Korea. Int J Hematol 2004;79:185–8.
Definitions and reporting framework for tuberculosis—2013 revision (updated December 2014 and January 2020): World Health Organization; 2020 [Available from: https://apps.who.int/iris/bitstream/handle/10665/79199/?sequence=1.
Nahid P, Dorman SE, Alipanah N, Barry PM, Brozek JL, Cattamanchi A, et al. Official American Thoracic Society/Centers for disease Control and Prevention/Infectious Diseases Society of America Clinical Practice Guidelines: treatment of drug-susceptible tuberculosis. Clin Infect Dis 2016;63:e147–e95.
Companion handbook to the WHO guidelines for the programmatic management of drug-resistant tuberculosis 2014 [Available from: https://apps.who.int/iris/bitstream/handle/10665/130918/9789241548809_eng.pdf.
Sun H-Y, Singh N. Opportunistic infection-associated immune reconstitution syndrome in transplant recipients. Clinical Infectious Diseases 2011;53:168–76.
Global tuberculosis report 2023. Geneva: World Health Organization; 2023. Licence: CC BY-NC-SA 3.0 IGO. Geneva;2024.
Roy Chowdhury R, Vallania F, Yang Q, Lopez Angel CJ, Darboe F, Penn-Nicholson A, et al. A multi-cohort study of the immune factors associated with M. tuberculosis infection outcomes. Nature 2018;560:644–8.
Elkington P, Polak ME, Reichmann MT, Leslie A. Understanding the tuberculosis granuloma: the matrix revolutions. Trends Mol Med 2022;28:143–54.
Fan WC, Liu CJ, Hong YC, Feng JY, Su WJ, Chien SH, et al. Long-term risk of tuberculosis in haematopoietic stem cell transplant recipients: a 10-year nationwide study. Int J Tuberc Lung Dis 2015;19:58–64.
Orfao NH, Andrade RLP, Ruffino-Netto A, da Silva LWF, Villa TCS, Seifert ML, et al. Influence of COVID-19 on the notification of drug-resistant pulmonary tuberculosis cases. BMC Infect Dis 2023;23:497.
8th European Conference on Infection in Leukemia. Tuberculosis (TB). 2019 [cited 2022 7 March 2022]. Available from: https://www.ebmt.org/sites/default/files/2019-12/ECIL%208-Tuberculosis%20and%20atypical%20mycobacterial%20infections%20-%20Final%20Slide%20Set.pdf.
Global Tuberculosis Report 2021. Geneva: World Health Organization; 2021. Licence: CC BY-NC-SA 3.0 IGO. Geneva;2021.
Organization WH. WHO consolidated guidelines on tuberculosis. Module 4: treatment-drug-susceptible tuberculosis treatment: World Health Organization; 2022.
Shaw ES, Stoker NG, Potter JL, Claassen H, Leslie A, Tweed CD, et al. Bedaquiline: what might the future hold? Lancet Microbe. 2024:100909.
Yamashita F, Sasa Y, Yoshida S, Hisaka A, Asai Y, Kitano H, et al. Modeling of rifampicin-induced CYP3A4 activation dynamics for the prediction of clinical drug-drug interactions from in vitro data. PLoS One 2013;8:e70330.
Tuloup V, France M, Garreau R, Bleyzac N, Bourguignon L, Tod M, et al. Model-based comparative analysis of rifampicin and rifabutin drug-drug interaction profile. Antimicrob Agents Chemother 2021;65:e0104321.
Zelunka EJ. Intravenous cyclosporine-rifampin interaction in a pediatric bone marrow transplant recipient. Pharmacotherapy 2002;22:387–90.
Almaghrabi RS, Nizami I, Alameer R, Alshehri N, Almohaizeie A, Alrajhi AA, et al. Successful use of rifamycin-sparing regimens for the treatment of active tuberculosis in lung transplant recipients. Exp Clin Transplant 2021;19:359–66.
Suzuki H, Matsuda Y, Noda M, Oishi H, Watanabe T, Sado T, et al. Management of De Novo mycobacterial infection after lung transplantation without rifampicin: case series of a single institution. Transplant Proc 2018;50:2764–7.
Sun HY, Munoz P, Torre-Cisneros J, Aguado JM, Lattes R, Montejo M, et al. Mycobacterium tuberculosis-associated immune reconstitution syndrome in solid-organ transplant recipients. Transplantation 2013;95:1173–81.
Luetkemeyer AF, Kendall MA, Nyirenda M, Wu X, Ive P, Benson CA, et al. Tuberculosis immune reconstitution inflammatory syndrome in A5221 STRIDE: timing, severity, and implications for HIV-TB programs. J Acquir Immune Defic Syndr 2014;65:423–8.
Acknowledgements
We are indebted to all centres participating to the EBMT database, and especially the ones who contributed to this study.
Author information
Authors and Affiliations
Contributions
JDS, JS, MM, AWG designed the study. JDS, DA, IV wrote the manuscript. IV, LW, NK managed data, GT did the statistics. DA, JS, and RC supervised the study. MK, NBA, AC, MA (Mahmoud Aljurf), NK, GO, JP, MG, MP, LLC, AT, AP, MA (Mohsen Al Zahrani), MCA, GB, ACEB, KC, AC, MF, PJ, GK, SDL, SM, JN, FP, KP, JAS, IY, MZ, AWG, RC, LG, MM, JS critically revised the paper and approved the final version.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
This retrospective study was approved by the Infectious Diseases Working Party (IDWP) of the EBMT and was performed in accordance with the Declaration of Helsinki.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Drozd-Sokołowska, J., Tridello, G., Verheggen, I. et al. Tuberculosis after hematopoietic cell transplantation: retrospective study on behalf of the Infectious Diseases Working Party of the EBMT. Bone Marrow Transplant 60, 603–616 (2025). https://doi.org/10.1038/s41409-025-02530-4
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41409-025-02530-4