Abstract
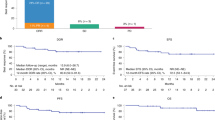
Older patients with lymphoma are typically underrepresented in clinical trials with chimeric antigen receptor T cell (CAR T) therapy. In this multicenter, observational study we aimed to assess the safety and efficacy of standard CD19 CAR T in patients 80 years of age or older. At total of 88 patients, median age 82 (range, 80-89) years, were included. Diffuse large B cell lymphoma (DLBCL) (N = 60, 68.2%) represented the most common histology. Patients were treated mostly with axicabtagene ciloleucel (N = 41, 46.6%) followed by lisocabtagene maraleucel (N = 25, 28.4%). Cytokine release syndrome (CRS) (any grade) was seen in 68 (77.3%) and 51 (58%) developed immune effector cell-associated neurotoxicity syndrome (ICANS). Incidence of grade 3-4 CRS and ICANS were 7.4% and 31.4%, respectively. For patients with DLBCL/tFL, the 1-year NRM, relapse, PFS, and OS were 11.6%, 40.8%, 47.6%, and 61.2%, respectively. We conclude that CAR T is feasible and effective in patients 80 years or older with B cell lymphomas. These patients must be provided the opportunity to be evaluated for this curative approach.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The datasets generated for this study are available from the corresponding author upon reasonable request and approval by all authors
References
Neelapu SS, Locke FL, Bartlett NL, Lekakis LJ, Miklos DB, Jacobson CA, et al. Axicabtagene ciloleucel CAR T-cell therapy in refractory large B-cell lymphoma. N Engl J Med. 2017;377:2531–44.
Schuster SJ, Bishop MR, Tam CS, Waller EK, Borchmann P, McGuirk JP, et al. Tisagenlecleucel in adult relapsed or refractory diffuse large B-cell lymphoma. N Engl J Med. 2019;380:45–56.
Abramson, Palomba JS, Gordon LI ML, Lunning MA, Wang M, Arnason J, et al. Lisocabtagene maraleucel for patients with relapsed or refractory large B-cell lymphomas (TRANSCEND NHL 001): a multicentre seamless design study. Lancet. 2020;396:839–52.
Jacobson CA, Chavez JC, Sehgal AR, William BM, Munoz J, Salles G, et al. Axicabtagene ciloleucel in relapsed or refractory indolent non-Hodgkin lymphoma (ZUMA-5): a single-arm, multicentre, phase 2 trial. Lancet Oncol. 2022;23:91–103.
Fowler NH, Dickinson M, Dreyling M, Martinez-Lopez J, Kolstad A, Butler J, et al. Tisagenlecleucel in adult relapsed or refractory follicular lymphoma: the phase 2 ELARA trial. Nat Med. 2022;28:325–32.
Wang M, Munoz J, Goy A, Locke FL, Jacobson CA, Hill BT, et al. KTE-X19 CAR T-cell therapy in relapsed or refractory mantle-cell lymphoma. N Engl J Med. 2020;382:1331–42.
Locke FL, Miklos DB, Jacobson CA, Perales MA, Kersten MJ, Oluwole OO, et al. Axicabtagene ciloleucel as second-line therapy for large B-cell lymphoma. N Engl J Med. 2022;386:640–54.
Kamdar M, Solomon SR, Arnason J, Johnston PB, Glass B, Bachanova V, et al. Lisocabtagene maraleucel versus standard of care with salvage chemotherapy followed by autologous stem cell transplantation as second-line treatment in patients with relapsed or refractory large B-cell lymphoma (TRANSFORM): results from an interim analysis of an open-label, randomised, phase 3 trial. Lancet. 2022;399:2294–308.
Westin JR, Oluwole OO, Kersten MJ, Miklos DB, Perales MA, Ghobadi A, et al. Survival with axicabtagene ciloleucel in large B-cell lymphoma. N Engl J Med. 2023;389:148–57.
Maus MV, Alexander S, Bishop MR, Brudno JN, Callahan C, Davila ML, et al. Society for Immunotherapy of Cancer (SITC) clinical practice guideline on immune effector cell-related adverse events. J Immunother Cancer. 2020;8:e001511.
Lee DW, Santomasso BD, Locke FL, Ghobadi A, Turtle CJ, Brudno JN, et al. ASTCT consensus grading for cytokine release syndrome and neurologic toxicity associated with immune effector cells. Biol Blood Marrow Transplant. 2019;25:625–38.
Yakoub-Agha I, Chabannon C, Bader P, Basak GW, Bonig H, Ciceri F, et al. Management of adults and children undergoing chimeric antigen receptor T-cell therapy: best practice recommendations of the European Society for Blood and Marrow Transplantation (EBMT) and the Joint Accreditation Committee of ISCT and EBMT (JACIE). Haematologica. 2020;105:297–316.
Jain T, Olson TS, Locke FL. How I treat cytopenias after CAR T-cell therapy. Blood. 2023;141:2460–9.
Gurney M, Baranwal A, Rosenthal A, Kharfan-Dabaja MA, Kenderian SS, Lin Y, et al. Features and factors associated with myeloid neoplasms after chimeric antigen receptor T-cell therapy. JAMA Oncol. 2024;10:532–5.
Abu Sabaa A, Morth C, Hasselblom S, Hedstrom G, Flogegard M, Stern M, et al. Age is the most important predictor of survival in diffuse large B-cell lymphoma patients achieving event-free survival at 24 months: a Swedish population-based study. Br J Haematol. 2021;193:906–14.
Junlen HR, Peterson S, Kimby E, Lockmer S, Linden O, Nilsson-Ehle H, et al. Follicular lymphoma in Sweden: nationwide improved survival in the rituximab era, particularly in elderly women: a Swedish Lymphoma Registry study. Leukemia. 2015;29:668–76.
Chihara D, Liao L, Tkacz J, Franco A, Lewing B, Kilgore KM, et al. Real-world experience of CAR T-cell therapy in older patients with relapsed/refractory diffuse large B-cell lymphoma. Blood. 2023;142:1047–55.
Sharma A, Singh V, Deol A. Epidemiology and predictors of 30-day readmission in CAR-T cell therapy recipients. Transplant Cell Ther. 2023;29:108.e1–e7.
Jacobson CA, Locke FL, Ma L, Asubonteng J, Hu ZH, Siddiqi T, et al. Real-world evidence of axicabtagene ciloleucel for the treatment of large B cell lymphoma in the United States. Transplant Cell Ther. 2022;28:581.e1–e8.
Yassine F, Iqbal M, Murthy H, Kharfan-Dabaja MA, Chavez JC. Real world experience of approved chimeric antigen receptor T-cell therapies outside of clinical trials. Curr Res Transl Med. 2020;68:159–70.
Maloney DG, Kuruvilla J, Liu FF, Kostic A, Kim Y, Bonner A, et al. Matching-adjusted indirect treatment comparison of liso-cel versus axi-cel in relapsed or refractory large B cell lymphoma. J Hematol Oncol. 2021;14:140.
Bachy E, Le Gouill S, Di Blasi R, Sesques P, Manson G, Cartron G, et al. A real-world comparison of tisagenlecleucel and axicabtagene ciloleucel CAR T cells in relapsed or refractory diffuse large B cell lymphoma. Nat Med. 2022;28:2145–54.
Berning P, Shumilov E, Maulhardt M, Boyadzhiev H, Kerkhoff A, Call S, et al. Chimeric antigen receptor-T cell therapy shows similar efficacy and toxicity in patients with diffuse large B-cell lymphoma aged 70 and older compared to younger patients: A multicenter cohort study. Hemasphere. 2024;8:e54.
Acknowledgements
The authors thank Ms. Zhuo Li, Mayo Clinic, Jacksonville, FL for the statistical support. This work was submitted in part, as an abstract, to the 2024 Annual Meeting of the European Hematology Association in Madrid, Spain.
Author information
Authors and Affiliations
Contributions
MAK-D: Designed the study, provided and analyzed the data and prepared the manuscript, RM: Designed the study, provided and analyzed the data and reviewed the manuscript, NE (Easwar): Provided and analyzed the data and reviewed the manuscript, PJ: Provided and analyzed the data and reviewed the manuscript, JY: Provided and analyzed the data and reviewed the manuscript MI: Designed the study, provided and analyzed the data and reviewed the manuscript, NE (Epperla): Provided and analyzed the data and reviewed the manuscript, NA: Provided and analyzed the data and reviewed the manuscript, MH: Provided and analyzed the data and reviewed the manuscript, AB: Provided and analyzed the data and reviewed the manuscript, MN: Provided and analyzed the data and reviewed the manuscript, DJ: Provided and analyzed the data and reviewed the manuscript, PRG: Provided and analyzed the data and reviewed the manuscript, BD: Provided and analyzed the data and reviewed the manuscript, UG: Provided and analyzed the data and reviewed the manuscript, JM: Provided and analyzed the data and reviewed the manuscript, JS-S: Provided and analyzed the data and reviewed the manuscript, FLL: Provided and analyzed the data and reviewed the manuscript, JF: Provided and analyzed the data and reviewed the manuscript, AK: Provided and analyzed the data and reviewed the manuscript, EA: Provided and analyzed the data and reviewed the manuscript, KA: Provided and analyzed the data and reviewed the manuscript, APR: Provided and analyzed the data and reviewed the manuscript, FL: Provided and analyzed the data and reviewed the manuscript, OSA: Provided and analyzed the data and reviewed the manuscript, LL: Provided and analyzed the data and reviewed the manuscript, AM: Provided and analyzed the data and reviewed the manuscript, OOO: Provided and analyzed the data and reviewed the manuscript, JL: Provided and analyzed the data and reviewed the manuscript, MDJ: Provided and analyzed the data and reviewed the manuscript, TS: Provided and analyzed the data and reviewed the manuscript, UD: Provided and analyzed the data and reviewed the manuscript, MAM: Provided and analyzed the data and reviewed the manuscript, JMc: Provided and analyzed the data and reviewed the manuscript, YL: Provided and analyzed the data and reviewed the manuscript, SY: Provided and analyzed the data and reviewed the manuscript, JCC : Designed the study, provided and analyzed the data and prepared the manuscript.
Corresponding author
Ethics declarations
Competing interests
MAK-D reports grant/research from Bristol Myers Squibb, Novartis and Pharmacyclics, and lecture/honorarium from Kite Pharma; NE (Epperla) reports research funding from Beigene and Eli Lilly. Speakers Bureau for Beigene, Incyte, and Novartis. Ad board for ADC Therapeutics. NA reports Ad Board participation for Bristol Myers Squibb. Consultancy fees for Kite/ Gilead. MH reports research support/Funding: ADC Therapeutics, Spectrum Pharmaceuticals, Astellas Pharma. Consultancy: ADC Therapeutics, Omeros, CRISPR, BMS, Kite, AbbVie, Caribou, Genmab, Autolus. Speaker’s Bureau: ADC Therapeutics, AstraZeneca, Bei Gene, Kite. DMC: Inc, Genentech, Myeloid Therapeutics, CRISPR. PRG reports consultancy services to Kite/Gilead Pharma, Bristol Myers Squibb (BMS) and served on the advisory boards of Pharmacyclics LLC, ADC Therapeutics, Cellectar Biosciences, Ono Pharma, CRISPR therapeutics, IPSEN Biopharma, and Regeneron Pharmaceuticals. AB reports honoraria from Autolus and Kite Pharma. BD reports stocks in Syndax and Iovance Biotherapeutics. Honoraria: MJH Healthcare Holdings, LLC, Curio Science, Gilead Sciences. Consulting/Advisory: Janssen Oncology, Boxer Capital, ADC Therapeutics, Ellipsis, Acrotech Biopharma. Speakers’ Bureau: Gilead Sciences. Research: Pfizer, Poseida therapeutics, MEI Pharma, Angiocrine Bioscience, Takeda, Janssen Oncology, WUGEN, Inc., Orca Bio, Gilead Sciences, AlloVir, Molecular Templates, Atara Biotherapeutics. Travel/accomodations: Poseida. UG reports research/grant support from Bristol Myers Sqibb, Kite Pharma and Novartis. Consultancy/speaker bureau from Kite Pharma. JM reports consulting for Pharmacyclics/Abbvie, Bayer, Gilead/Kite, Beigene, Pfizer, Janssen, Celgene/BMS, Kyowa, Alexion, Fosunkite, Seattle Genetics, Karyopharm, Aurobindo, Verastem, Genmab, Genzyme, Genentech/Roche, ADC Therapeutics, Epizyme, Beigene, Novartis, Morphosys/Incyte, MEI, TG Therapeutics, AstraZeneca, Eli Lilly. Research funding from Bayer, Gilead/Kite, Celgene, Merck, Portola, Incyte, Genentech, Pharmacyclics, Seattle Genetics, Janssen, Millennium, Novartis, Beigene. Honoraria from Targeted Oncology, OncView, Curio, and Physicians’ Education Resource. JS-S reports Speaking for Pfizer (SeaGen) and Ad Board for SeaGen, MassiveBio, ADC therapeutics, Genmab, BeiGene, AbbVie, and Bristol Myers Squibb. FLL reports consulting/advisory role for Allogene, Amgen, bluebird bio, Bristol Myers Squibb, Celgene, Calibr, Cellular Biomedicine Group, Cowen, ecoR1, Emerging Therapy Solutions Gerson Lehman Group, GammaDelta Therapeutics, Iovance, Janssen, Kite, a Gilead Company, Legend Biotech, Novartis, Umoja Biopharma, and Wugen; research funding from Allogene, Kite, and Novartis; and patents, royalties, other intellectual property from several patents held by the institution in my name (unlicensed) in the field of cellular immunotherapy. FL reports advisory board for ADC Therapeutics. OOO reports consultancy and advisory board for Pfizer, Kite, Gilead, AbbVie, Janssen, TGR therapeutics, ADC, Novartis, Epizyme, Curio science, Nektar, Cargo, Caribou. Institution funding from Kite, Pfizer, Daichi Sankyo, Allogene. Honoraria from Pfizer, Gilead. MDJ reports consultancy/advisory for Kite/Gilead, Novartis. Research funding from Kite/Gilead, Incyte, and Loxo@Lilly. TS reports consultancy for BioLine Rx. MAM reports consultancy for AbbVie. JMc reports consulting for Envision, Novartis, Caribou Bio, Sana Technologies, Legend Biotech. Ad Board participation for Autolus, Consulting/Ad Board for Kite/Scimentum, AlloVIr, Bristol Myers Squibb, CRISPR, Nektar. YL reports consulting/advisory board for Janssen, Sanofi, Bristol Myers Squibb, Regeneron, Genentech. Research from Janssen, Bristol Myers Squibb. Scientific Advisory Boards for NexImmune, Caribou. DSMB/EDMC for Pfizer. SY reports consulting for Bristol Myers Squibb and Advisory board for Kite Pharma. JCC Reports advisory/consulting role for Genmab, ADC Therapeutics, Kite/Gilead, Novartis, Astrazeneca, Cellectis, Allogene, BMS, Janssen, Abbvie, Beigene, Pfizer. Research support from Genmab, Merck, Janssen, RM, NE (Easwar), PJ, MI, MN, JF, EA, KA, APR, OSA, LL, JL declare no relevant conflicts of interest to disclose.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kharfan-Dabaja, M.A., Mohty, R., Easwar, N. et al. Chimeric antigen receptor T cell therapy in octogenarians with B cell lymphoma: a real-world US multicenter collaborative study. Bone Marrow Transplant 60, 632–639 (2025). https://doi.org/10.1038/s41409-025-02541-1
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41409-025-02541-1
This article is cited by
-
Therapeutic landscape of primary refractory and relapsed diffuse large B-cell lymphoma: Recent advances and emerging therapies
Journal of Hematology & Oncology (2025)