Abstract
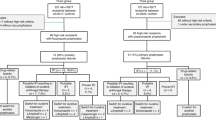
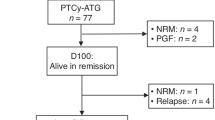
Incidence of breakthrough proven-probable invasive fungal infections (b-PP-IFIs) in allogeneic haematopoietic cell transplant recipients (allo-HCT-r) receiving mould-active prophylaxis (MAP) and post-transplant cyclophosphamide (PT-Cy) is largely unknown. Retrospective study on allo-HCT-r, classified at high-risk for IFIs whether ≥1 of the following conditions was met: 1] active disease; 2] cord-blood; 3] previous transplant; 4] acute graft-versus-host-disease (a-GVHD) grade≥3; 5] mismatched-related or unrelated donor with neutropenia before transplant or grade-2 a-GVHD or Cytomegalovirus infection. Objectives were to estimate cumulative incidence function (CIF) of b-PP-IFIs, evaluate infection-related mortality (IRM) and predictive factors of b-PP-IFIs. Overall, 473 allo-HCT-r (n = 286 posaconazole, n = 187 voriconazole) were analysed: 64.7% were at high-risk, 81.6% received PT-Cy. Fifteen b-PP-IFIs occurred: 14/306 in high-risk, 1/167 in non-high-risk group. CIF of b-PP-IFIs in high-risk group was 2.0% (95%CI = 0.8–4.1%) at 30-day and 5.1% (95%CI = 2.9–8.2%) at 1-year post-transplant. The 1-year CIF of IRM was higher in allo-HCT-r with b-PP-IFIs compared to those without [46.7% (95%CI = 19.6–70%) vs. 8.2% (95%CI = 5.3–9.2%), Gray’s test: p < 0.001]. In allo-HCT-r receiving PT-Cy, neutropenia before transplant [sHR 7.54 (95%CI = 1.81–31.43)] and chronic myeloproliferative disorders versus AML/MDS [sHR 7.72 (95%CI = 1.68–35.42)] increased risk of b-PP-IFIs, while donor type did not. MAP effectively prevented IFIs. PT-Cy conferred a comparable risk of b-PP-IFIs in matched compared to mismatched-transplants.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
Data availability
Materials described in the manuscript, including all relevant raw data, will be available upon demanding to the corresponding author.
References
Ullmann AJ, Lipton JH, Vesole DH, Chandrasekar P, Langston A, Tarantolo SR, et al. Posaconazole or fluconazole for prophylaxis in severe graft-versus-host disease. N. Engl J Med. 2007;356:335–47.
Maertens JA, Girmenia C, Bruggemann RJ, Duarte RF, Kibbler CC, Ljungman P, et al. European guidelines for primary antifungal prophylaxis in adult haematology patients: summary of the updated recommendations from the European Conference on Infections in Leukaemia. J Antimicrob Chemother. 2018;73:3221–30.
Stemler J, Mellinghoff SC, Khodamoradi Y, Sprute R, Classen AY, Zapke SE, et al. Primary prophylaxis of invasive fungal diseases in patients with haematological malignancies: 2022 update of the recommendations of the Infectious Diseases Working Party (AGIHO) of the German Society for Haematology and Medical Oncology (DGHO). J Antimicrob Chemother. 2023;78:1813–26.
Teh BW, Yeoh DK, Haeusler GM, Yannakou CK, Fleming S, Lindsay J, et al. Consensus guidelines for antifungal prophylaxis in haematological malignancy and haemopoietic stem cell transplantation, 2021. Intern Med J. 2021;51:67–88.
Girmenia C, Barosi G, Piciocchi A, Arcese W, Aversa F, Bacigalupo A, et al. Primary prophylaxis of invasive fungal diseases in allogeneic stem cell transplantation: revised recommendations from a consensus process by gruppo italiano trapianto midollo osseo (GITMO). Biol Blood Marrow Transpl. 2014;20:1080–8.
Winston DJ, Maziarz RT, Chandrasekar PH, Lazarus HM, Goldman M, Blumer JM, et al. Intravenous and oral itraconazole versus intravenous and oral fluconazole for long-term antifungal prophylaxis in allogeneic hematopoietic stem-cell transplant recipients. A multicenter, randomized trial. Ann Intern Med. 2003;138:705–13.
Marr KA, Crippa F, Leisenring W, Hoyle M, Boeckh M, Balajee SA, et al. Itraconazole versus fluconazole for prevention of fungal infections in patients receiving allogeneic stem cell transplants. Blood. 2004;103:1527–33.
Wingard JR, Carter SL, Walsh TJ, Kurtzberg J, Small TN, Baden LR, et al. Randomized, double-blind trial of fluconazole versus voriconazole for prevention of invasive fungal infection after allogeneic hematopoietic cell transplantation. Blood. 2010;116:5111–8.
Marks DI, Pagliuca A, Kibbler CC, Glasmacher A, Heussel CP, Kantecki M, et al. Voriconazole versus itraconazole for antifungal prophylaxis following allogeneic haematopoietic stem-cell transplantation. Br J Haematol. 2011;155:318–27.
Girmenia C, Raiola AM, Piciocchi A, Algarotti A, Stanzani M, Cudillo L, et al. Incidence and outcome of invasive fungal diseases after allogeneic stem cell transplantation: a prospective study of the gruppo italiano trapianto midollo osseo (GITMO). Biol Blood Marrow Transpl. 2014;20:872–80.
Bogler Y, Stern A, Su Y, Lee YJ, Seo SK, Shaffer B, et al. Efficacy and safety of isavuconazole compared with voriconazole as primary antifungal prophylaxis in allogeneic hematopoietic cell transplant recipients. Med Mycol. 2021;59:970–9.
Kraljevic M, Khanna N, Medinger M, Passweg J, Masouridi-Levrat S, Chalandon Y, et al. Clinical considerations on posaconazole administration and therapeutic drug monitoring in allogeneic hematopoietic cell transplant recipients. Med Mycol. 2021;59:701–11.
Stern A, Su Y, Lee YJ, Seo S, Shaffer B, Tamari R, et al. A single center, open-label trial of isavuconazole prophylaxis against invasive fungal infection in patients undergoing allogeneic hematopoietic cell transplant (HCT). Biol Blood Marrow Transpl. 2020;26:1195–202.
Little JS, Shapiro RM, Aleissa MM, Kim A, Bai Park Chang J, Kubiak DW, et al. Invasive yeast infection after haploidentical donor hematopoietic cell transplantation associated with cytokine release syndrome. Transpl Cell Ther. 2022;28:508.e501–8.
Fayard A, Daguenet E, Blaise D, Chevallier P, Labussiere H, Berceanu A, et al. Evaluation of infectious complications after haploidentical hematopoietic stem cell transplantation with post-transplant cyclophosphamide following reduced intensity and myeloablative conditioning: a study on behalf of the francophone Society of Stem Cell Transplantation and Cellular Therapy (SFGM-TC). Bone Marrow Transpl. 2019;54:1586–94.
Esquirol A, Pascual MJ, Kwon M, Perez A, Parody R, Ferra C, et al. Severe infections and infection-related mortality in a large series of haploidentical hematopoietic stem cell transplantation with post-transplant cyclophosphamide. Bone Marrow Transpl. 2021;56:2432–44.
Mohty R, Brissot E, Battipaglia G, Ruggeri A, Dulery R, Bonnin A, et al. Infectious complications after post-transplantation cyclophosphamide and anti-thymocyte globulin-based haploidentical stem cell transplantation. Br J Haematol. 2019;187:e64–8.
Papanicolaou GA, Chen M, He N, Martens MJ, Kim S, Batista MV, et al. Incidence and Impact of Fungal Infections in PostTransplantation Cyclophosphamide-Based Graft-versus-Host Disease Prophylaxis and Haploidentical Hematopoietic Cell Transplantation: A Center for International Blood and Marrow Transplant Research Analysis. Transpl Cell Ther. 2024;30:114.e1–114.e16.
Sorror LM, Maris MB, Storb R, Baron F, Sandmaier BM, Maloney DG, et al. Hematopoietic cell transplantation (HCT)-specific comorbidity index: a new tool for risk assessment before allogeneic HCT. Blood. 2005;106:2912–9.
Donnelly PJ, Chen SC, Kauffman CA, Steinbach WJ, Baddley JW, Verweij PE, et al. Revision and update of the consensus definitions of invasive fungal disease from the European Organization for Research and Treatment of Cancer and the Mycoses Study Group Education and Research Consortium. Clin Infect Dis. 2020;71:1367–76.
Cornely OA, Hoenig M, Lass-Flör C, Chen S, Kontoyiannis DP, Morrissey CO, et al. Defining breakthrough invasive fungal infection–position paper of the mycoses study group education and research consortium (MSG-ERC) and the European Confederation of Medical Mycology (ECMM). Mycoses. 2019;62:716–29.
Harris AC, Young R, Devine S, Hogan WJ, Ayuk F, Bunworasate U, et al. International, multicenter standardization of acute graft-versus-host disease clinical data collection: a report from the Mount Sinai Acute GVHD International Consortium. Biol Blood Marrow Transpl. 2016;22:4–10.
Jagasia MH, Greinix HT, Arora M, Williams KM, Wolff D, Cowen EW, et al. National institutes of health consensus development project on criteria for clinical trials in chronic graft versus-host disease: the 2014 diagnosis and staging working group report. Biol Blood Marrow Transpl. 2015;21:389–401.
Marty FM, Ljungman P, Chemaly RF, Maertens J, Dadwal SS, Duarte RF, et al. Letermovir prophylaxis for cytomegalovirus in hematopoietic-cell transplantation. N. Engl J Med. 2017;377:2433–44.
Ljungman P, Boeckh M, Hirsch HH, Josephson F, Lundgren J, Nichols G, et al. Definitions of cytomegalovirus infection and disease in transplant patients for use in clinical trials. Clin Infect Dis. 2017;64:87–91.
Ullmann AJ, Aguado JM, Arikan-Akdagli S, Denning DW, Groll AH, Lagrou K, et al. Diagnosis and management of Aspergillus diseases: executive summary of the 2017 ESCMID-ECMM-ERS guideline. Clin Microbiol Infect. 2018;24:e1–e38.
Greco R, Lorentino F, Albanese S, Lupo Stanghellini MT, Giglio F, Piemontese S, et al. Post-transplantation cyclophosphamide- and sirolimus-based graft-versus-host-disease prophylaxis in allogeneic stem cell transplant. Transpl Cell Ther. 2021;27:776.e1–776.e13.
Casper J, Knauf W, Kiefer T, Wolff D, Steiner B, Hammer U, et al. Treosulfan and fludarabine: a new toxicity-reduced conditioning regimen for allogeneic hematopoietic stem cell transplantation. Blood. 2004;103:725–31.
Fine JP, Gray RJ. A proportional hazards model for the sub-distribution of a competing risk. J Am Stat Assoc. 1999;94:496–509.
Harrison N, Mitterbauer M, Tobudic S, Kalhs P, Rabitsch W, Greinix H, et al. Incidence and characteristics of invasive fungal diseases in allogeneic hematopoietic stem cell transplant recipients: a retrospective cohort study. BMC Infect Dis. 2015;15:584.
Roth RS, Masouridi-Levrat S, Chalandon Y, Mamez AC, Giannotti F, Riat A, et al. Invasive mold infections in allogeneic hematopoietic cell transplant recipients in 2020: have we made enough progress? Open Forum Infect Dis. 2021;9:ofab596.
Malagola M, Turra A, Signorini L, Corbellini S, Polverelli N, Masina L, et al. Results of an innovative program for surveillance, prophylaxis, and treatment of infectious complications following allogeneic stem cell transplantation in hematological malignancies (BATMO Protocol). Front Oncol. 2022;12:874117.
Busca A, Cinatti N, Gill J, Passera R, Dellacasa CM, Giaccone L, et al. Management of invasive fungal infections in patients undergoing allogeneic hematopoietic stem cell transplantation: the turin experience. Front Cell Infect Microbiol. 2022;11:805514.
Mikuslka M, Bartalucci C, Raiola AM, Oltolini C. Does PTCY increase the risk of infections? Blood Rev. 2023;62:101092.
Polverelli N, Breccia M, Benevolo G, Martino B, Tieghi A, Latagliata R, et al. Risk factors for infections in myelofibrosis: role of disease status and treatment. A multicenter study of 507 patients. Am J Hematol. 2017;92:37–41.
Kröger N, Bacigalupo A, Barbui T, Ditschkowski M, Gagelmann N, Griesshammer M, et al. Indication and management of allogeneic haematopoietic stem-cell transplantation in myelofibrosis: updated recommendations by the EBMT/ELN International Working Group. Lancet Hematol. 2024;11:e62–e74.
Acknowledgements
Authors thank Sara Diotallevi for her contribution to the statistical analyses of the study.
Author information
Authors and Affiliations
Contributions
MC, FF, AA and CO collected the data; MC, FF, AA, LG, RG and CO interpreted the data, CO wrote the manuscript; LG performed statistical analysis and prepared figures; RG, CO, AC and FC designed the study; MC, FF, AA, GM, EX, DC, SM, SP, MTLS, MR, CT, CC, JP, PS, AC, FC and RG contributed to patients’ clinical care; and all authors approved the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chiurlo, M., Galli, L., Farina, F. et al. Risk stratification and impact of donor type on breakthrough invasive fungal infections in haematopoietic cell transplant with post-transplant cyclophosphamide and mould-active prophylaxis. Bone Marrow Transplant 60, 1036–1044 (2025). https://doi.org/10.1038/s41409-025-02605-2
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41409-025-02605-2