Abstract
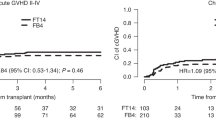
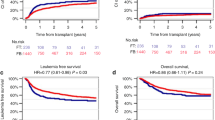
In the past decades, treatment for acute myeloid leukemia (AML) has advanced, but allogeneic hematopoietic stem cell transplantation (allo-HSCT) remains vital for improving survival in most patients. This retrospective study, conducted on behalf of the Acute Leukemia Working Party of the EBMT, examines the impact of fludarabine dose in reduced-intensity conditioning regimens on clinical outcomes in patients over 50 years old with AML in first complete remission, without chronic kidney disease. We analyzed 1907 patients who underwent allo-HSCT between 2010 and 2022, stratifying them into four fludarabine dose groups: 110-130 mg/m2, 140–150 mg/m2, 151–160 mg/m2, and 170–190 mg/m2. Our results suggest that a lower fludarabine dose (≤130 mg/m2) is associated with significantly improved leukemia-free survival (LFS), graft-versus-host disease-free/relapse-free survival (GRFS), overall survival, and reduced non-relapse mortality. Multivariate analysis shows that a lower fludarabine dose (≤130 mg/m2) was associated with significantly improved LFS (HR 1.46, 95% CI: 1.09–1.94) and GRFS (HR 1.50, 95% CI: 1.12–1.99). These findings indicate that using a lower fludarabine dosing in older AML patients may improve the efficacy and tolerability of allo-HSCT. Further studies are needed to validate these observations to confirm and expand upon our results, particularly in diverse patient populations and other indications for allo-HSCT.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
Data availability
The datasets analysed during the current study are available from the corresponding author on reasonable request.
References
Dohner H, Wei AH, Appelbaum FR, Craddock C, DiNardo CD, Dombret H, et al. Diagnosis and management of AML in adults: 2022 recommendations from an international expert panel on behalf of the ELN. Blood. 2022;140:1345–77.
Passweg JR, Baldomero H, Atlija M, Kleovoulou I, Witaszek A, Alexander T, et al. The 2023 EBMT report on hematopoietic cell transplantation and cellular therapies. Increased use of allogeneic HCT for myeloid malignancies and of CAR-T at the expense of autologous HCT. Bone Marrow Transplant. 2025. https://doi.org/10.1038/s41409-025-02524-2
Bacigalupo A, Ballen K, Rizzo D, Giralt S, Lazarus H, Ho V, et al. Defining the intensity of conditioning regimens: working definitions. Biol Blood Marrow Transplant. 2009;15:1628–33.
Sawyer J, Elliott T, Orton L, Sowell H, Gatwood K, Shultes K. Prevention and management of acute toxicities from conditioning regimens during hematopoietic stem cell transplantation. Clin Hematol Int. 2024;6:1–10.
Shah R, Murphy D, Logue M, Jerkins J, Jallouk A, Adetola K, et al. Multidisciplinary management of morbidities associated with chronic graft-versus-host disease. Clin Hematol Int. 2024;6:74–88.
Langenhorst JB, Dorlo TPC, van Maarseveen EM, Nierkens S, Kuball J, Boelens JJ, et al. Population pharmacokinetics of fludarabine in children and adults during conditioning prior to allogeneic hematopoietic cell transplantation. Clin Pharmacokinet. 2019;58:627–37.
Langenhorst JB, van Kesteren C, van Maarseveen EM, Dorlo TPC, Nierkens S, Lindemans CA, et al. Fludarabine exposure in the conditioning prior to allogeneic hematopoietic cell transplantation predicts outcomes. Blood Adv. 2019;3:2179–87.
Dekker L, Calkoen FG, Jiang Y, Blok H, Veldkamp SR, De Koning C, et al. Fludarabine exposure predicts outcome after CD19 CAR T-cell therapy in children and young adults with acute leukemia. Blood Adv. 2022;6:1969–76.
Fabrizio VA, Boelens JJ, Mauguen A, Baggott C, Prabhu S, Egeler E, et al. Optimal fludarabine lymphodepletion is associated with improved outcomes after CAR T-cell therapy. Blood Adv. 2022;6:1961–8.
Sorror M. Impacts of pretransplant comorbidities on allogeneic hematopoietic cell transplantation (HCT) outcomes. Biol Blood Marrow Transplant. 2009;15:149–53.
Ruggeri A, Labopin M, Ciceri F, Mohty M, Nagler A. Definition of GvHD-free, relapse-free survival for registry-based studies: an ALWP-EBMT analysis on patients with AML in remission. Bone Marrow Transplant. 2016;51:610–1.
Dohner H, Estey E, Grimwade D, Amadori S, Appelbaum FR, Buchner T, et al. Diagnosis and management of AML in adults: 2017 ELN recommendations from an international expert panel. Blood. 2017;129:424–47.
Nagler A, Baron F, Labopin M, Polge E, Esteve J, Bazarbachi A, et al. Measurable residual disease (MRD) testing for acute leukemia in EBMT transplant centers: a survey on behalf of the ALWP of the EBMT. Bone Marrow Transplant. 2021;56:218–24.
Langenhorst JB, Dorlo TPC, van Kesteren C, van Maarseveen EM, Nierkens S, de Witte MA, et al. Clinical Trial Simulation To Optimize Trial Design for Fludarabine Dosing Strategies in Allogeneic Hematopoietic Cell Transplantation. CPT Pharmacomet Syst Pharm. 2020;9:272–81.
de Witte MA A prospective randomized controlled trial to address the impact of targeted fludarabine on the incidence of viral complications after tcralphabeta/CD19 depleted ALLO-HCT. In: EBMT Annual Meeting 2024. Glasgow, UK, 2024.
Scordo M, Flynn JR, Gonen M, Devlin SM, Parascondola A, Tomas AA, et al. Identifying an optimal fludarabine exposure for improved outcomes after axi-cel therapy for aggressive B-cell non-Hodgkin lymphoma. Blood Adv. 2023;7:5579–85.
Chung H, Hong KT, Lee JW, Rhee SJ, Kim S, Yoon SH, et al. Pharmacokinetics of fludarabine and its association with clinical outcomes in paediatric haematopoietic stem cell transplantation patients. Bone Marrow Transplant. 2019;54:284–92. e-pub ahead of print 20180618
Hruba L, Das V, Hajduch M, Dzubak P. Nucleoside-based anticancer drugs: mechanism of action and drug resistance. Biochem Pharmacol. 2023;215:115741.
Duy PN, Thuy NT, Trang BK, Giang NH, Van NTH, Xuan NT. Regulation of NF-kappaB- and STAT1-mediated plasmacytoid dendritic cell functions by A20. PLoS ONE. 2019;14:e0222697.
Maloney DG. Graft-vs.-lymphoma effect in various histologies of non-Hodgkin’s lymphoma. Leuk Lymphoma. 2003;44:S99–105.
Weinkove R, Brooks CR, Carter JM, Hermans IF, Ronchese F. Functional invariant natural killer T-cell and CD1d axis in chronic lymphocytic leukemia: implications for immunotherapy. Haematologica. 2013;98:376–84.
Author information
Authors and Affiliations
Contributions
GD, ML, MM designed the study, performed data analysis and interpretation, and wrote the manuscript; GS, CC, EF, IWB, PC, ED, DB, CEB, RV, AH, DB, JM, TS, JOB, BS, AS, FC took care of the patients and collected the data; all authors contributed to critical review and drafting of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
The ALWP review board approved the study protocol. This study was conducted in accordance with all relevant guidelines and regulations. Patients (or their legal guardians) provided informed consent authorizing the use of their personal information for research purposes.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Dachy, G., Labopin, M., Socié, G. et al. Impact of fludarabine dose on outcome after allo-HSCT with reduced intensity conditioning for older patients with AML. Bone Marrow Transplant 60, 1258–1264 (2025). https://doi.org/10.1038/s41409-025-02614-1
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41409-025-02614-1