Abstract
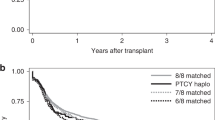
Few studies analyzed the impact of bone marrow (BM) or peripheral blood stem cells (PBSC) on outcomes after haploidentical transplantation using post-cyclophosphamide (Haplo-PTCY). We analyzed 8854 adults with malignant disorders, given a first Haplo-PTCY. BM cells was used in 2914 and PBSC in 5940 patients. Multivariate models were built adjusting for the statistical differences between the 2 groups. Median follow-up time for survivors in the BM and PBSC groups were 48 and 30 months, respectively. Neutrophil Engraftment was observed in 92.4% of BM and 93.7% of PBSC recipients (p = 0.01). In a multivariate analysis, use of PBSC compared to BM, was associated with higher incidence of acute (HR:1.53; p < 0.0001) and chronic GVHD (HR:1.34; p < 10-3), increased non-relapse mortality (HR:1.22; p = 0.002), similar risk of relapse (HR:1.02; p = 0.79), and decreased overall survival (OS)(HR:1.13; p = 0.008); progression-free survival (PFS)(HR:1.11; p = 0.024) and GVHD-Relapse free survival (GRFS) (HR:1.2; p < 10–3). In conclusion, use of BM cells is associated with better outcomes compared to PBSC after Haplo-PTCY. Future studies should investigate better GVHD prophylaxis in the PBSC-Haplo-PTCY setting and the association of measured T-cell or other subpopulations of lymphocyte content in the PBSC graft.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
Data availability
Data cannot be shared unless a specific request is sent to the EBMT. This was a multicenter retrospective study, conducted on behalf of the Cellular therapy and Immunobiology Working Party (CTIWP) of EBMT.
References
Luznik L, O’Donnell PV, Symons HJ, Chen AR, Leffell MS, Zahurak M, et al. HLA-haploidentical bone marrow transplantation for hematologic malignancies using nonmyeloablative conditioning and high-dose, post-transplantation cyclophosphamide. Biol Blood Marrow Transpl. 2008;14:641–50.
Arcuri LJ, Aguiar MTM, Ribeiro AAF, Pacheco AGF, Kerbauy MN, Bonfim CM. Haploidentical transplantation with post-transplant cyclophosphamide versus unrelated donor hematopoietic stem cell transplantation: a systematic review and meta-analysis. Biol Blood Marrow Transpl. 2019;25:2422–30.
Battipaglia G, Galimard JE, Labopin M, Raiola AM, Blaise D, Ruggeri A, et al. Post-transplant cyclophosphamide in one-antigen mismatched unrelated donor transplantation versus haploidentical transplantation in acute myeloid leukemia: a study from the Acute Leukemia Working Party of the EBMT. Bone Marrow Transpl. 2022;57:562–71. https://doi.org/10.1038/s41409-022-01577-x.
Anasetti C, Logan BR, Lee SJ, Waller EK, Weisdorf DJ, Wingard JR, et al. Peripheral-blood stem cells versus bone marrow from unrelated donors. N Engl J Med. 2012;367:1487–96.
Arcuri LJ, Hamerschlak N, Rocha V, Bonfim C, Kerbauy MN, Ribeiro AAF. Outcomes after haploidentical hematopoietic cell transplantation with post transplantation cyclophosphamide: a systematic review and meta-analysis comparing myeloablative with reduced-intensity conditioning regimens and bone marrow with peripheral blood stem cell grafts. Transplantation Cell Ther. 2021;27:782.e1–782.e7.
Ruggeri A, Labopin M, Ciceri F, Mohty M, Nagler A. Definition of GvHD-free, relapse-free survival for registry-based studies: an ALWP-EBMT analysis on patients with AML in remission. Bone Marrow Transpl. 2016;51:610–1.
Martino R, Romero P, Subirá M, Sureda A, Brunet S, Urbano-Ispizua A, et al. Comparison of the classic Glucksberg criteria and the IBMTR Severity Index for grading acute graft-versus-host disease following HLA-identical sibling stem cell transplantation. Bone Marrow Transpl. 1999;24:283–7.
Jagasia MH, Greinix HT, Arora M, Williams KM, Wolff D, Cowen EW, et al. National Institutes of Health consensus development project on criteria for clinical trials in chronic graft-versus-host disease: I. The 2014 Diagnosis and Staging Working Group Report. Biol Blood Marrow Transpl. 2015;21:389–401.
Hougaard P. Frailty models for survival data. Lifetime Data Anal 1995;1:255–73, Andersen PK, Klein JP, Zhang MJ. Testing for centre effects in multi-centre survival studies: a Monte Carlo comparison of fixed and random effects tests. Stat Med. 1999;18:1489–500.
Korbling M, Anderlini P. Peripheral blood stem cell versus bone marrow allotransplantation: does the source of hematopoietic stem cells matter?. Blood. 2001;98:2900–8.
Solomon SR, Sizemore CA, Sanacore M, Zhang X, Brown S, Holland HK, et al. Haploidentical transplantation using T cell replete peripheral blood stem cells and myeloablative conditioning in patients with high-risk hematologic malignancies who lack conventional donors is well tolerated and produces excellent relapse-free survival: results of a prospective phase II trial. Biol Blood Marrow Transpl. 2012;18:1859–66.
Farhan S, Peres E, Janakiraman N, Jagasia M, Bashey A, Solomon SR. Choice of unmanipulated T cell replete graft for haploidentical stem cell transplant and posttransplant cyclophosphamide in hematologic malignancies in adults: peripheral blood or bone marrow—review of published literature. Adv Hematol. 2016;2016:6950346.
Devine SM. Haploidentical hematopoietic cell transplantation using post-transplantation cyclophosphamide: does graft source matter?. J Clin Oncol. 2017;35:2984–6.
Bashey A, Zhang MJ, McCurdy SR, St Martin A, Argall T, Anasetti C, et al. Mobilized peripheral blood stem cells versus unstimulated bone marrow as a graft source for T-cell-replete haploidentical donor transplantation using post-transplant cyclophosphamide. J Clin Oncol. 2017;35:3002–9.
Ruggeri A, Labopin M, Bacigalupo A, Gülbas Z, Koc Y, Blaise D, et al. Bone marrow versus mobilized peripheral blood stem cells in haploidentical transplants using posttransplantation cyclophosphamide. Cancer. 2018;124:1428–37.
Im A, Rashidi A, Wang T, Hemmer M, MacMillan ML, Pidala J, et al. Risk factors for graft-versus-host disease in haploidentical hematopoietic cell transplantation using post-transplant cyclophosphamide. Biol Blood Marrow Transpl. 2020;26:1459–68.
Battipaglia G, Labopin M, Blaise D, Diez-Martin JL, Bazarbachi A, Vitek A, et al. Impact of the addition of antithymocyte globulin to post-transplantation cyclophosphamide in haploidentical transplantation with peripheral blood compared to post-transplantation cyclophosphamide alone in acute myelogenous leukemia: a retrospective study on behalf of the Acute Leukemia Working Party of the European Society for Blood and Marrow Transplantation. Transplant Cell Ther. 2022;28:587.e1–587.e7. https://doi.org/10.1016/j.jtct.2022.06.006.
Admiraal R, Nierkens S, de Witte MA, Bierings MB, van Kesteren C, Versluijs AB, et al. Association between anti-thymocyte globulin exposure and survival outcomes in pediatric unrelated hematopoietic cell transplantation. Lancet Haematol. 2015;2:e194–203. https://doi.org/10.1016/S2352-3026(15)00044-7.
Admiraal R, van Kesteren C, Jol-van der Zijde CM, Lankester AC, Bierings MB, Egeler RM, et al. Association between anti-thymocyte globulin exposure and survival outcomes in adult unrelated hematopoietic cell transplantation. J Clin Oncol. 2017;35:4003–12. https://doi.org/10.1200/JCO.2017.73.6352.
Author information
Authors and Affiliations
Consortia
Contributions
VR and AR designed the study, VR and AR wrote the manuscript; ML performed the statistical analysis. JEG has reviewed the statistical analysis. AMR, RD, SB, FC, JLDM, YK, JT, SS, MM, ZG, AK, LLC, AS provided cases for the study. All authors edited and approved the manuscript. VR: honoraria of Takeda, Janssen, Amgen, Gilead Kite, Sanofi, Roche, Pierre Fabre. AS: Honoraria: Takeda, BMS/Celgene, MSD, Janssen, Amgen, Novartis, Gilead Kite, Sanofi, Roche, Alexion, Pierre Fabre, Jazz Pharmaceuticals, Verte. Consultancy: Takeda, BMS/Celgene, Novartis, Janssen, Gilead, Sanofi, GenMab, Abbvie, MSD, Incyte, Autolus, Regeneron, Pierre Fabre. Speaker’s Bureau: Takeda. Research Support: Takeda. Non-profit organizations: Presidency of the GETH-TC, Presidency of the EBMT. The remaining authors have no conflict of interest to disclose.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
Data were collected according to EBMT rules. All patients or their legal guardians provided written informed consent for data collection and analysis in accordance with the Declaration of Helsinki, and with the centers’ ethical research guidelines. The scientific board of the CTIWP of the EBMT approved this study.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Rocha, V., Labopin, M., Raiola, A.M. et al. Use of bone marrow cells is associated with improved outcomes when compared to peripheral blood stem cell after haplo-identical transplants with post transplant cyclophosphamide, a study from of the CTIWP-EBMT. Bone Marrow Transplant 61, 75–81 (2026). https://doi.org/10.1038/s41409-025-02735-7
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41409-025-02735-7