Abstract
Thrombocytopenia following an allogeneic hematopoietic cell transplant (allo-HCT) is a potentially serious complication, and the efficacy of eltrombopag, a thrombopoietin receptor agonist, in this context is unclear due to inconsistent findings. Additionally, other post-allograft outcomes in eltrombopag treated patients such as transfusion independence, overall survival (OS), and non-relapse mortality have not been systematically reviewed. The aim of this systematic review/meta-analysis (SR/MA) is to evaluate the efficacy of eltrombopag in allo-HCT-induced thrombocytopenia by analyzing data from 16 eligible studies. Pooled rate of response for platelets counts achieving >30 × 109/L and >50 × 109/L were 72% and 56%, respectively. When evaluating the composite endpoint of platelets response and transfusion independence, pooled rates for >30 × 109/L and >50 × 109/L plus transfusion independence were 47% and 56%, respectively. Pooled OS, bleeding-related mortality and mortality from GVHD/infection were 68%, 6%, and 19%, respectively. These findings show that eltrombopag is an effective treatment of allo-HCT-induced thrombocytopenia. Optimal dose and duration of treatment remain to be determined in a large prospective study. Results of this SR/MA suggest a beneficial effect of eltrombopag for thrombocytopenia after allo-HCT. These results could represent the benchmark to be used for future prospective and comparative studies to better understand the benefit of this intervention.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
This work represents the results of a systematic review and meta-analysis. Data were extracted from published information in the public domain. The methodology section explains the criteria used to select studies for inclusion in the meta-analysis. All data generated or analyzed during this study are included in this published article.
References
Ayas M, Nassar A, Hamidieh AA, Kharfan-Dabaja M, Othman TB, Elhaddad A, et al. Reduced intensity conditioning is effective for hematopoietic SCT in dyskeratosis congenita-related BM failure. Bone Marrow Transpl. 2013;48:1168–72. https://doi.org/10.1038/bmt.2013.35.
Gooley TA, Chien JW, Pergam SA, Hingorani S, Sorror ML, Boeckh M, et al. Reduced mortality after allogeneic hematopoietic-cell transplantation. N Engl J Med. 2010;363:2091–101. https://doi.org/10.1056/NEJMoa1004383.
Hansen JA, Gooley TA, Martin PJ, Appelbaum F, Chauncey TR, Clift RA, et al. Bone marrow transplants from unrelated donors for patients with chronic myeloid leukemia. N Engl J Med. 1998;338:962–8. https://doi.org/10.1056/NEJM199804023381405.
Kassim AA, Walters MC, Eapen M, Smith M, Logan BR, Solh M, et al. Haploidentical bone marrow transplantation for sickle cell disease. NEJM Evid. 2025;4:EVIDoa2400192 https://doi.org/10.1056/EVIDoa2400192.
Kharfan-Dabaja MA, Al Malki MM, Deotare U, Raj RV, El-Jurdi N, Majhail N, et al. Haematopoietic cell transplantation for blastic plasmacytoid dendritic cell neoplasm: a North American multicentre collaborative study. Br J Haematol. 2017;179:781–9. https://doi.org/10.1111/bjh.14954.
Kroger N, Bacigalupo A, Barbui T, Ditschkowski M, Gagelmann N, Griesshammer M, et al. Indication and management of allogeneic haematopoietic stem-cell transplantation in myelofibrosis: updated recommendations by the EBMT/ELN International Working Group. Lancet Haematol. 2024;11:e62–e74. https://doi.org/10.1016/S2352-3026(23)00305-8.
Bento L, Canaro M, Bastida JM, Sampol A. Thrombocytopenia and therapeutic strategies after allogeneic hematopoietic stem cell transplantation. J Clin Med. 2022;11; https://doi.org/10.3390/jcm11051364
ASHP. Eltrombopag. In: Bethesda. Maryland: The American Society of Health-System Pharmacists; 2004.
Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372:n71 https://doi.org/10.1136/bmj.n71.
Wells GA, Shea B, O’Connell D, Peterson J, Welch V, Losos M, et al. The Newcastle-Ottawa Scale (NOS) for assessing the quality if nonrandomized studies in meta-analyses. Ottawa, Ontario: University of Ottawa.
STATA, 18.5 ed. College Station, TX: StataCorp LLC; 2023.
Deeks JJHJ, Altman DG, McKenzie JE and Veroniki AA. Chapter 10: Analysing data and undertaking meta-analyses. In: Julian Higgins JT, editor, Cochrane Handbook for Systematic Reviews of Interventions, version 6.5. Cochrane: Chichester (UK): 2024.
Ahmed S, Bashir Q, Bassett R, Poon MC, Valdez B, Konoplev S, et al. Eltrombopag for post-transplantation thrombocytopenia: results of phase II randomized, double-blind, placebo-controlled trial. Transplant Cell Ther. 2021;27:430 e431–430 e437. https://doi.org/10.1016/j.jtct.2021.02.004.
Aguirre L, Lizardi A, Bachiller P, Esteban B, Gonzalez C, Argoitia N, et al. Eltrombopag (EPAG) induces a high percentage of responses in patients with post allo-HSCT poor graft function (PGF) and no active GVHD. Bone Marrow Transplantation 2019;54(supplement 1):228–229.
Arslan Davulcu E, Akad Soyer N, Vural F. Eltrombopag for the treatment of allogeneic hematopoietic stem cell transplantation-related poor graft function. Cureus. 2023;15:e44555 https://doi.org/10.7759/cureus.44555.
Bostankolu Değirmenci B, Yegin ZA, Dikyar A, Aydın Kaynar L, Özkurt ZN, Uyar Göçün P et al. Eltrombopag may induce bone marrow fibrosis in allogeneic hematopoietic stem cell transplant recipients with prolonged thrombocytopenia. Leuk Res. 2022:118:106870.
Fu H, Zhang X, Han T, Mo X, Wang Y, Chen H, et al. Eltrombopag is an effective and safe therapy for refractory thrombocytopenia after haploidentical hematopoietic stem cell transplantation. Bone Marrow Transplant. 2019;54:1310–8. https://doi.org/10.1038/s41409-019-0435-2.
Gao F, Zhou X, Shi J, Luo Y, Tan Y, Fu H, et al. Eltrombopag treatment promotes platelet recovery and reduces platelet transfusion for patients with post-transplantation thrombocytopenia. Ann Hematol. 2020;99:2679–87. https://doi.org/10.1007/s00277-020-04106-2.
Guven ZT, Celik S, Eser B, Cetin M, Unal A, Kaynar L. Eltrombopag for treatment of thrombocytopenia following hematopoietic stem cell transplantation. Turk J Haematol. 2022;39:103–8. https://doi.org/10.4274/tjh.galenos.2022.2021.0675.
He G, Karrison T, Stock W, Larson RA, Kosuri S, LaBelle JL, et al. Eltrombopag is a safe and effective agent to treat thrombocytopenia after allogeneic hematopoietic stem cell transplantation (allo-HSCT). In: 2023 Tandem Meetings. Transplantation & Cellular Therapy Meetings of ASTCT and CIBMTR. Tandem Meetings. 2023;29:S142–S143.
Karatas A, Goker H, Demiroglu H, Malkan UY, Velet M, Cinar OE, et al. Efficacy of eltrombopag in thrombocytopenia after hematopoietic stem celltransplantation. Turk J Med Sci. 2022;52:413–9. https://doi.org/10.55730/1300-0144.5328.
Kilic Gunes E, Yigit Kaya S, Yaman F, Yeniay MK, Vural K, Comert M, et al. Eltrombopag treatment in thrombocytopenia following hematopoietic stem cell transplantation: a multicenter real-world experience. Leuk Res. 2024;140:107484. https://doi.org/10.1016/j.leukres.2024.107484.
Nampoothiri RV, Ho L, McEwan C, Pasic I, Lam W, Law AD, et al. Efficacy and cost analysis of eltrombopag in thrombocytopenia and poor graft function post allogeneic hematopoietic cell transplantation. Bone Marrow Transpl. 2021;56:2471–6. https://doi.org/10.1038/s41409-021-01362-2.
Rivera D, Bastida JM, Lopez-Corral L, Sanchez-Guijo F, Cabrero M, Martin A, et al. Usefulness of eltrombopag for treating thrombocytopenia after allogeneic stem cell transplantation. Bone Marrow Transpl. 2019;54:757–61. https://doi.org/10.1038/s41409-018-0368-1.
Tanaka T, Inamoto Y, Yamashita T, Fuji S, Okinaka K, Kurosawa S, et al. Eltrombopag for treatment of thrombocytopenia after allogeneic hematopoietic cell transplantation. Biol Blood Marrow Transpl. 2016;22:919–24. https://doi.org/10.1016/j.bbmt.2016.01.018.
Wang TP, Lin RY, Pereira D, Beitinjaneh A, Lekakis LJ, Jimenez AM, et al. Eltrombopag may improve platelet recovery in the outpatient setting after allogeneic stem cell transplant. Biology Blood Marrow Transplant. 2020;26:S343.
Yan F, Lu N, Gu Z, Huang W, Wang S, Gao X et al. Eltrombopag in the treatment of patients with persistent thrombocytopenia after haploidentical peripheral blood stem cell transplantation: a single-center experience. Ann Hematol. 2022;101:397–408.
Yuan C, Boyd AM, Nelson J, Patel RD, Varela JC, Goldstein SC, et al. Eltrombopag for treating thrombocytopenia after allogeneic stem cell transplantation. Biol Blood Marrow Transpl. 2019;25:1320–4. https://doi.org/10.1016/j.bbmt.2019.01.027.
Vallejo C, García I, Sanchez Guijo F, Cuesta M, Solano C, Sampol A, et al. P837: Eltrombopag (epag) induces hematologic responses for patients with post-allogenic hematopoietic stem cell transplant poor graft function (post-allo hsct pgf): results of the eltion study in spain. HemaSphere. 2022;6:731–2. https://doi.org/10.1097/01.Hs9.0000846232.58961.88.
Elsayed A, Elsayed B, Elmarasi M, Elsabagh AA, Elsayed E, Elmakaty I, et al. Thrombopoietin receptor agonists in post-hematopoietic cell transplantation complicated by prolonged thrombocytopenia: a comprehensive review. Immunotargets Ther. 2024;13:461–86. https://doi.org/10.2147/ITT.S463384.
Novartis AG PROMACTA (eltrombopag)[package insert. U.S. Food and Drug Administration website; 2008.
Author information
Authors and Affiliations
Contributions
AK, AS, TR, MI, RM, HM, and MAK-D were responsible for designing the study protocol, screening potentially eligible studies, extracting and analyzing the data, interpreting results, and writing the manuscript. TN, RP, EA, VR, and MA were responsible for analyzing the data, interpreting the results and reviewing and provided feedback on the final manuscript.
Corresponding author
Ethics declarations
Competing interests
AK, AS, TR, RM, EA, VR, MA declare no financial conflicts of interest. MI declares consultancy for US Sanofi, Bristol Myers Squibb and ADC therapeutics; HM declares consultancy or advisory board member: Bristol Myers Squibb, CRISPR Therapeutics, Incyte, Jazz Pharmaceuticals, Senti Bioscience, Autolus, Kite/Gilead, Sobi; medical monitor for Bone Marrow Transplant Clinical Trial Network; TN declares clinical trial research support to the institution from Novartis and Karyopharm; RP declares serving on the advisory board for Sanofi Aventis and Astra Zeneca, has received honoraria from Sanofi Aventis, Astra Zeneca and Beigene, and has received research funding from Bristol Myers Squibb Foundation, Beigene, Cullinan Therapeutics, and GlaxoSmithKline; MAK-D declares research/grant from Bristol Myers Squibb, Novartis, and Pharmacyclics and lecture/honoraria from Kite Pharma.
Ethics approval
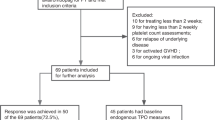
All methods were performed in accordance with the relevant guidelines and regulations. The study selection process was summarized using the PRISMA flow diagram and methodology for conduct of systematic reviews followed the Cochrane Collaboration Handbook.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kumar, A., Singh, A., Reljic, T. et al. Efficacy of eltrombopag for treatment of thrombocytopenia in the setting of allogeneic hematopoietic cell transplantation: a systematic review and meta-analysis. Bone Marrow Transplant 61, 211–218 (2026). https://doi.org/10.1038/s41409-025-02760-6
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41409-025-02760-6