Abstract
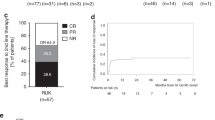
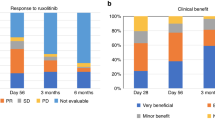
CHRONOS is a multicenter, retrospective, cohort study involving acute graft-versus-host disease (aGvHD) adult patients with gastrointestinal (GI) symptoms, steroid- and ruxolitinib refractory, who initiated third-line therapy between 30 May 2019 and 30 September 2024. Primary endpoints were all-organ overall response rate (ORR) and GI-specific ORR (GI-ORR) around 28 days after treatment initiation. Secondary endpoints included duration of response, real-world progression-free survival (rwPFS) of underlying malignancy, and overall survival (OS). Fifty-nine patients from 16 sites in Europe were included. On Day 28, ORR was 36% (95% CI: 24–49%), GI-ORR was 37% (95% CI: 25–51%); 29% (95% CI: 11–49%) of responders lost response within 30 days, and 52% (95% CI: 29–72%) within 90 days. Median rwPFS and median OS were both 86 days (95% CI: 54–128 days). Median OS was higher in responders than in non-responders (186 versus 45 days). Over the 12-month follow-up period, 41 patients died, mainly due to aGvHD progression (n = 25), and infectious complications (n = 9). Within 3 months, Grade ≥ 2 infectious events occurred in 51% of patients; Grade 3–4 thrombocytopenia and neutropenia in 64% and 32%, respectively. These findings demonstrate limited effectiveness of third-line therapy in this cohort of steroid- and ruxolitinib-refractory aGvHD patients with GI symptoms.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Garcia-Cadenas I, Awol R, Esquirol A, Saavedra S, Bosch-Vilaseca A, Novelli S, et al. Incorporating posttransplant cyclophosphamide-based prophylaxis as standard-of-care outside the haploidentical setting: challenges and review of the literature. Bone Marrow Transpl. 2020;55:1041–9.
Martin PJ, Rizzo JD, Wingard JR, Ballen K, Curtin PT, Cutler C, et al. First- and second-line systemic treatment of acute graft-versus-host disease: recommendations of the American Society of Blood and Marrow Transplantation. Biol Blood Marrow Transpl. 2012;18:1150–63.
Newell LF, Holtan SG. Acute GVHD: think before you treat. Hematol Am Soc Hematol Educ Program. 2021;2021:642–7.
Al Malki MM, Gendzekhadze K, Yang D, Mokhtari S, Parker P, Karanes C, et al. Long-term Outcome of Allogeneic Hematopoietic Stem Cell Transplantation From Unrelated Donor Using Tacrolimus/Sirolimus-based GvHD Prophylaxis: Impact of HLA Mismatch. Transplantation. 2020;104:1070–80.
Ballen K, Logan BR, Chitphakdithai P, Kuxhausen M, Spellman SR, Adams A, et al. Unlicensed Umbilical Cord Blood Units Provide a Safe and Effective Graft Source for a Diverse Population: A Study of 2456 Umbilical Cord Blood Recipients. Biol Blood Marrow Transpl. 2020;26:745–57.
Carreras E, Diaz-Ricart M. Early Complications of Endothelial Origin. In: Carreras E, Dufour C, Mohty M, Kroger N, editors. The EBMT Handbook: Hematopoietic Stem Cell Transplantation and Cellular Therapies. 7th ed. Cham (CH) 2019. 315-22.
Greco R, Lorentino F, Nitti R, Lupo Stanghellini MT, Giglio F, Clerici D, et al. Interleukin-6 as Biomarker for Acute GvHD and Survival After Allogeneic Transplant With Post-transplant Cyclophosphamide. Front Immunol. 2019;10:2319.
Jagasia M, Arora M, Flowers ME, Chao NJ, McCarthy PL, Cutler CS, et al. Risk factors for acute GVHD and survival after hematopoietic cell transplantation. Blood. 2012;119:296–307.
Ferrara JL, Levine JE, Reddy P, Holler E. Graft-versus-host disease. Lancet. 2009;373:1550–61.
Malard F, Huang XJ, Sim JPY. Treatment and unmet needs in steroid-refractory acute graft-versus-host disease. Leukemia. 2020;34:1229–40.
Castilla-Llorente C, Martin PJ, McDonald GB, Storer BE, Appelbaum FR, Deeg HJ, et al. Prognostic factors and outcomes of severe gastrointestinal GVHD after allogeneic hematopoietic cell transplantation. Bone Marrow Transpl. 2014;49:966–71.
Leung ASY, Wai CYY, Leung NYH, Ngai NA, Chua GT, Ho PK, et al. Real-World Sensitization and Tolerance Pattern to Seafood in Fish-Allergic Individuals. J Allergy Clin Immunol Pr. 2024;12:633–42.e9.
Vander Lugt MT, Braun TM, Hanash S, Ritz J, Ho VT, Antin JH, et al. ST2 as a marker for risk of therapy-resistant graft-versus-host disease and death. N Engl J Med. 2013;369:529–39.
Penack O, Marchetti M, Aljurf M, Arat M, Bonifazi F, Duarte RF, et al. Prophylaxis and management of graft-versus-host disease after stem-cell transplantation for haematological malignancies: updated consensus recommendations of the European Society for Blood and Marrow Transplantation. Lancet Haematol. 2024;11:e147–e59.
Axt L, Naumann A, Toennies J, Haen SP, Vogel W, Schneidawind D, et al. Retrospective single center analysis of outcome, risk factors and therapy in steroid refractory graft-versus-host disease after allogeneic hematopoietic cell transplantation. Bone Marrow Transpl. 2019;54:1805–14.
Holtan SG, Yu J, Choe HK, Paranagama D, Tang J, Naim A, et al. Disease progression, treatments, hospitalization, and clinical outcomes in acute GVHD: a multicenter chart review. Bone Marrow Transpl. 2022;57:1581–5.
Asensi Canto P, Gomez-Segui I, Montoro J, Villalba Montaner M, Chorao P, Solves Alcaina P, et al. Incidence, risk factors and therapy response of acute graft-versus-host disease after myeloablative hematopoietic stem cell transplantation with posttransplant cyclophosphamide. Bone Marrow Transpl. 2024;59:1577–84.
Alousi AM, Weisdorf DJ, Logan BR, Bolanos-Meade J, Carter S, Difronzo N, et al. Etanercept, mycophenolate, denileukin, or pentostatin plus corticosteroids for acute graft-versus-host disease: a randomized phase 2 trial from the Blood and Marrow Transplant Clinical Trials Network. Blood. 2009;114:511–7.
Bolanos-Meade J, Logan BR, Alousi AM, Antin JH, Barowski K, Carter SL, et al. Phase 3 clinical trial of steroids/mycophenolate mofetil vs steroids/placebo as therapy for acute GVHD: BMT CTN 0802. Blood. 2014;124:3221–7. quiz 335.
Weisdorf D, Haake R, Blazar B, Miller W, McGlave P, Ramsay N, et al. Treatment of moderate/severe acute graft-versus-host disease after allogeneic bone marrow transplantation: an analysis of clinical risk features and outcome. Blood. 1990;75:1024–30.
Mohty M, Apperley JF. Long-term physiological side effects after allogeneic bone marrow transplantation. Hematol Am Soc Hematol Educ Program. 2010;2010:229–36.
Zeiser R, von Bubnoff N, Butler J, Mohty M, Niederwieser D, Or R, et al. Ruxolitinib for Glucocorticoid-Refractory Acute Graft-versus-Host Disease. N Engl J Med. 2020;382:1800–10.
Biavasco F, Ihorst G, Wasch R, Wehr C, Bertz H, Finke J, et al. Therapy response of glucocorticoid-refractory acute GVHD of the lower intestinal tract. Bone Marrow Transpl. 2022;57:1500–6.
Abedin S, Rashid N, Schroeder M, Romee R, Nauffal M, Alhaj Moustafa M, et al. Ruxolitinib resistance or intolerance in steroid-refractory acute graft-versus-host disease - a real-world outcomes analysis. Br J Haematol. 2021;195:429–32.
Malard F, Holler E, Sandmaier BM, Huang H, Mohty M. Acute graft-versus-host disease. Nat Rev Dis Prim. 2023;9:27.
ISPE. Guidelines for Good Pharmacoepidemiology Practices (GPP): International Society for Pharmacoepidemiology; 2015 [updated June 2015. Revision 3:
Harris AC, Young R, Devine S, Hogan WJ, Ayuk F, Bunworasate U, et al. International, Multicenter Standardization of Acute Graft-versus-Host Disease Clinical Data Collection: A Report from the Mount Sinai Acute GVHD International Consortium. Biol Blood Marrow Transpl. 2016;22:4–10.
Schoemans HM, Lee SJ, Ferrara JL, Wolff D, Levine JE, Schultz KR, et al. EBMT-NIH-CIBMTR Task Force position statement on standardized terminology & guidance for graft-versus-host disease assessment. Bone Marrow Transpl. 2018;53:1401–15.
Mohty M, Holler E, Jagasia M, Jenq R, Malard F, Martin P, et al. Refractory acute graft-versus-host disease: a new working definition beyond corticosteroid refractoriness. Blood. 2020;136:1903–6.
Martin PJ, Bachier CR, Klingemann HG, McCarthy PL, Szabolcs P, Uberti JP, et al. Endpoints for clinical trials testing treatment of acute graft-versus-host disease: a joint statement. Biol Blood Marrow Transpl. 2009;15:777–84.
Michonneau D, Devillier R, Keranen M, Rubio MT, Nicklasson M, Labussiere-Wallet H, et al. Treatment Patterns and Clinical Outcomes of Patients with Moderate to Severe Acute Graft-Versus-Host Disease: A Multicenter Chart Review Study. Hematol Rep. 2024;16:283–94.
Luo C, Huang X, Wei L, Wu G, Huang Y, Ding Y, et al. Second-line therapy for patients with steroid-refractory aGVHD: systematic review and meta-analysis of randomized controlled trials. Front Immunol. 2023;14:1211171.
Holler EG, H; Zeiser, R Acute Graft-Versus-Host Disease In: (eds.) ASea, editor. The EMBT Handbook - Hematopoietic Cell Transplantation and Cellular Therapies: Springer Cham; 2024. 385-93.
Martin PJ. How I treat steroid-refractory acute graft-versus-host disease. Blood. 2020;135:1630–8.
Mori Y, Ikeda K, Inomata T, Yoshimoto G, Fujii N, Ago H, et al. Ruxolitinib treatment for GvHD in patients with myelofibrosis. Bone Marrow Transpl. 2016;51:1584–7.
Ruutu T, Gratwohl A, de Witte T, Afanasyev B, Apperley J, Bacigalupo A, et al. Prophylaxis and treatment of GVHD: EBMT-ELN working group recommendations for a standardized practice. Bone Marrow Transpl. 2014;49:168–73.
Bonig H, Verbeek M, Herhaus P, Braitsch K, Beutel G, Schmid C, et al. Real-world data suggest effectiveness of the allogeneic mesenchymal stromal cells preparation MSC-FFM in ruxolitinib-refractory acute graft-versus-host disease. J Transl Med. 2023;21:837.
Allogeneic Transplantation: Acute and Chronic GVHD and Immune Reconstitution: Clinical and Translational Insights: 67th ASH® Annual Meeting; Available from: https://submit.hematology.org/program/session/117229.
Mohty M. Pooled fecal allogenic microbiotherapy for refractory gastrointestinal acute graft-versus-host disease: results from the early access program in Europe: EHA library; 2025 [Available from: https://library.ehaweb.org/eha/2025/eha2025-congress/4159337/.mohamad.mohty.pooled.fecal.allogenic.microbiotherapy.for.refractory.html?f=listing%3D0%2Abrowseby%3D8%2Asortby%3D1%2Asearch%3Dgvhd.
Acknowledgements
The authors thank Pierre CLERSON (Soladis Clinical Studies) for medical writing support and Marie-Blanche ONSELAER (Soladis Clinical Studies) for scientific writing support.
Funding
MaaT Pharma.
Author information
Authors and Affiliations
Contributions
EP, FM, MM contributed to the conception of the study. EP, HJP, BKM, IF contributed to the design of the study, analysis and interpretation of the data. HJP supervised the project. HJP, FM, MM wrote and critically revised the manuscript. JC, JAPS, MC, CCL, DM, AS, PAC, SF, LG, LLC, A Benzaquen, HL-W, JC, RD, AH, PT, A Bauhofer, HPJ, MB contributed to the collection of data. All authors reviewed and approved the manuscript.
Corresponding authors
Ethics declarations
Competing interests
JAPS received research grants from Sanofi, Incyte and Amge; consulting fees from Gilead, J&J, Novartis, BMS, Sanofi, Jazz pharmaceuticals, Incyte, GSK, Amgen; honoraria or travel support from Gilead, J&J, Novartis, BMS, Sanofi, Jazz pharmaceuticals, Incyte, GSK, Amgen, MDS. CCL received honoraria or travel support from Jazz pharmaceuticals. DM received research grants from Novartis, Sanofi, CSL Behring; consulting fees from Novartis, Incyte, Sanofi, Jazz pharmaceuticals; honoraria or travel support from Sanofi, Novartis, Jazz pharmaceuticals; fees for participation in review activities such as data monitoring boards or advisory boards from Incyte, Sanofi. AS received consulting fees from Alexion, Gilead, MaaT pharma, Novartis, Basilea, Sanofi. AS received honoraria from Pfizer satellite symposia, Pierre Fabre; fees for participation in review activities such as data monitoring boards or advisory boards from Alexion, Gilead, Maat Pharma, Novartis, Basilea, Sanofi. PAC received honoraria or travel support from Kite, Amgen, Macopharma, Gilead, Therakos, Novartis. LG received travel support from Gilead. LG received research grants and consulting fees from Grifols, Abbvie. RD received consulting fees from Sanofi, Novartis. AH received travel support from Sanofi, Novartis. FM received honoraria or travel support from Gilead, Sanofi, Novartis, BMS, Astrazeneca, Therakos, Priothera, MSD, Jazz pharmaceuticals. MM received research grants from Sanofi, Novartis, Janssen, Jazz pharmaceuticals; consulting fees from Adaptive Biotechnologies, Amgen, Astellas, BMS, GSK, Janssen, Jazz Pharmaceuticals, Novartis, Pfizer, Sanofi, Stemline, Takeda; honoraria from Adaptive Biotechnologies, Amgen, Astellas, BMS, GSK, Janssen, Jazz Pharmaceuticals, Novartis, Pfizer, Sanofi, Stemline, Takeda, MaaT pharma; fees for participation in review activities such as data monitoring boards or advisory board from Jenssen; holds a role on the board, society or committee of EBMT, IACH. HJP, IF, BKM, received consulting fees from Maat Pharma. BKM received travel support from Maat Pharma. MB, EP are employees and stockholders of MaaT Pharma. HJP is stockholder of MaaT Pharma. JC, MC, SF, LLC, PT, A Bauhofer and A Benzaquen declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Clausen, J., Pérez Simón, J.A., Carré, M. et al. Clinical outcomes of third-line therapy for aGvHD with gastrointestinal involvement after steroids and ruxolitinib failure. Bone Marrow Transplant (2026). https://doi.org/10.1038/s41409-026-02825-0
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41409-026-02825-0