Abstract
Background
We assessed health-related quality of life (symptoms of therapy/patient functioning/global health status), in APHINITY (pertuzumab/placebo, trastuzumab, and chemotherapy as adjuvant HER2-positive early breast cancer therapy).
Methods
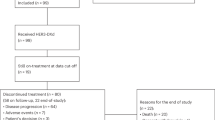
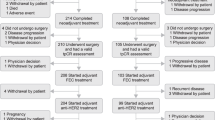
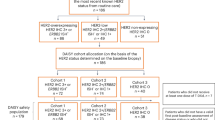
Patients received 1 year/18 cycles of pertuzumab/placebo with trastuzumab and chemotherapy and completed EORTC QLQ-C30 and BR23 questionnaires until 36 months post-randomisation/disease recurrence. Changes ≥10 points from baseline were considered clinically meaningful.
Results
87–97% of patients completed questionnaires. In the pertuzumab versus placebo arms, mean decrease in physical function scores (baseline → end of taxane) was −10.7 (95% CI −11.4, −10.0) versus −10.6 (−11.4, −9.9), mean decrease in global health status was −11.2 (−12.2, −10.2) versus −10.2 (−11.1, −9.2), and mean increase in diarrhoea scores (baseline → end of taxane) was +22.3 (21.0, 23.6) versus +9.2 (8.2, 10.2). Diarrhoea scores remained elevated versus baseline in the pertuzumab arm throughout HER2-targeted treatment (week 25: +13.2; end of treatment: +12.2). Role functioning was maintained in both arms.
Conclusions
Improved invasive disease-free survival achieved by adding pertuzumab to trastuzumab and chemotherapy did not adversely affect the ability to conduct activities of daily living versus trastuzumab and chemotherapy alone. Patient-reported diarrhoea worsened during taxane therapy in both arms, persisting during HER2-targeted treatment in the pertuzumab arm.
ClinicalTrials.gov
NCT01358877.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 24 print issues and online access
$259.00 per year
only $10.79 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Baselga, J., Cortés, J., Kim, S.-B., Im, S.-A., Hegg, R., Roman, L. et al. Pertuzumab plus trastuzumab plus docetaxel for metastatic breast cancer. N. Engl. J. Med. 366, 109–119 (2012).
Swain, S. M., Baselga, J., Kim, S.-B., Ro, J., Semiglazov, V., Campone, M. et al. Pertuzumab, trastuzumab, and docetaxel in HER2-positive metastatic breast cancer. N. Engl. J. Med. 372, 724–734 (2015).
Gianni, L., Pienkowski, T., Im, Y.-H., Roman, L., Tseng, L. M., Liu, M. C. et al. Efficacy and safety of neoadjuvant pertuzumab and trastuzumab in women with locally advanced, inflammatory, or early HER2-positive breast cancer (NeoSphere): a randomised multicentre, open-label, phase 2 trial. Lancet Oncol. 13, 25–32 (2012).
Cortazar, P., Zhang, L., Untch, M., Mehta, K., Costantino, J. P., Wolmark, N. et al. Pathological complete response and long-term clinical benefit in breast cancer: the CTNeoBC pooled analysis. Lancet 384, 164–172 (2014).
von Minckwitz, G., Procter, M., de Azambuja, E., Zardavas, D., Benyunes, M., Viale, G. et al. Adjuvant pertuzumab and trastuzumab in early HER2-positive breast cancer. N. Engl. J. Med. 377, 122–131 (2017).
Piccart, M., Procter, M., Fumagalli, D. de Azambuja, E., Clark, E., Ewer, M. S. et al. Adjuvant Pertuzumab and Trastuzumab in Early HER2-Positive Breast Cancer in the APHINITY Trial: 6 Years' Follow-Up. J. Clin. Oncol. JCO2001204 (2021). https://doi.org/10.1200/JCO.20.01204. Epub ahead of print.
Roche Registration Ltd. Perjeta® (pertuzumab). Summary of Product Characteristics. https://www.ema.europa.eu/en/documents/product-information/perjeta-epar-product-information_en.pdf (2019).
Genentech Inc. Perjeta™ (pertuzumab). Prescribing Information (USA). https://www.accessdata.fda.gov/drugsatfda_docs/label/2018/125409s123lbl.pdf (2019).
Aaronson, N. K., Ahmedzai, S., Bergman, B., Bullinger, M., Cull, A., Duez, N. J. et al. The European Organization for Research and Treatment of Cancer QLQ-C30: a quality-of-life instrument for use in international clinical trials in oncology. J. Natl. Cancer Inst. 85, 365–376 (1993).
Sprangers, M. A., Groenvold, M., Arraras, J. I., Franklin, J., te Velde, A., Muller, M. et al. The European Organization for Research and Treatment of Cancer breast cancer-specific quality-of-life questionnaire module: first results from a three-country field study. J. Clin. Oncol. Cancer 14, 2756–2768 (1996).
Fayers, P. M. Interpreting quality of life data: population-based reference data for the EORTC QLQ-C30. Eur. J. Cancer 37, 1331–1334 (2001).
Osoba, D., Rodrigues, G., Myles, J., Zee, B. & Pater, J. Interpreting the significance of changes in health-related quality-of-life scores. J. Clin. Oncol. 16, 139–144 (1998).
Bines, J., Procter, M., Restuccia, E., Viale, G., Zardavas, D., Suter, T. et al. Incidence and Management of Diarrhea With Adjuvant Pertuzumab and Trastuzumab in Patients With Human Epidermal Growth Factor Receptor 2-Positive Breast Cancer. Clin. Breast Cancer 20, 174–181.e3 (2020).
Johansson, B., Brandberg, Y., Hellbom, M., Persson, C., Petersson, L. M., Berglund, G. et al. Health-related quality of life and distress in cancer patients: results from a large randomised study. Br. J. Cancer 99, 1975–1983 (2008).
Cortés, J., Baselga, J., Im, Y.-H., Im, S.-A., Pivot, X., Ross, G. et al. Health-related quality-of-life assessment in CLEOPATRA, a phase III study combining pertuzumab with trastuzumab and docetaxel in metastatic breast cancer. Ann. Oncol. 24, 2630–2635 (2013).
Dueck, A. C., Hillman, D. W., Kottschade, L. A., Halyard, M. Y., Sloan, J. A., Flickinger, L. M. et al. Quality of life (QOL) among patients (pts) with HER2+ breast cancer (bc) treated with adjuvant lapatinib and/or trastuzumab in the ALTTO study (BIG 2-06, Alliance N063D). J. Clin. Oncol. 32(suppl), 647 (2014).
Delaloge, S., Cella, D., Ye, Y., Buyse, M., Chan, A., Barrios, C. H. et al. Effects of neratinib on health-related quality of life in women with HER2-positive early-stage breast cancer: longitudinal analyses from the randomized phase III ExteNET trial. Ann. Oncol. 30, 567–574 (2019).
Barcenas, C. H., Hurvitz, S. A., Di Palma, J. A., Bose, R., Chien, A. J., Iannotti N., Marx G. et al. Improved tolerability of neratinib in patients with HER2-positive early-stage breast cancer: the CONTROL trial. Ann. Oncol. 31, 1223–1230 (2020).
Cocks, K., King, M. T., Velikova, G., Martyn St-James, M., Fayers, P. M. & Brown, J. M. Evidence-based guidelines for determination of sample size and interpretation of the European Organisation for the Research and Treatment of Cancer Quality of Life Questionnaire Core 30. J. Clin. Oncol. 29, 89–96 (2011).
Acknowledgements
The study partners: BIG, BrEAST, FSS, and F. Hoffmann-La Roche Ltd. The study sponsor: F. Hoffmann-La Roche Ltd. The Central Lab: IEO. The Steering Committee members. All members of the other committees involved in APHINITY: Translational Advisory Committee, Independent Data Monitoring Committee, Interface Committee, Cardiac Advisory Board. Support for third-party writing assistance for this manuscript, furnished by John Carron, Ph.D., of Health Interactions, was provided by F. Hoffmann-La Roche Ltd.
Author information
Authors and Affiliations
Contributions
J.Bi., E.R., J.Ba., and M.Pi. contributed to the conception of the work. J.Bi., E.R., J.Ba., and M.Pi. contributed to the design of the work. E.R., A.S., D.P., J.Ba., and C.J. contributed to the acquisition of data for the work. J.Bi., J.Ba., and C.J. contributed to the analysis of data for the work. J.Bi., E.Cl., C.B., E.R., M.Pr., A.S., A.A., D.F., J.Ba., G.V., L.R., E.F., R.D.G., M.Pi., C.J., and J.A.P. contributed to interpretation data for the work. All authors contributed to drafting the work or revising it critically for important intellectual content. All authors gave final approval of the version to be published. All authors agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
APHINITY was conducted in accordance with the Declaration of Helsinki; all patients provided written informed consent. The protocol was approved by the appropriate institutional review board at each centre. A full list of ethical committees and institutional review boards is listed in the supplementary material.
Data availability
Qualified researchers may request access to individual patient-level data through the clinical study data request platform: https://vivli.org. Further details on Roche’s criteria for eligible studies are available here: https://vivli.org/members/ourmembers. For further details on Roche’s Global Policy on the Sharing of Clinical Information and how to request access to related clinical study documents, see here: https://www.roche.com/research_and_development/who_we_are_how_we_work/clinical_trials/our_commitment_to_data_sharing.htm.
Competing interests
All authors received support for third-party writing assistance for this manuscript, provided by F. Hoffmann-La Roche Ltd. J.Bi. reports consulting/advisory roles for AbbVie, Genomic Health, Libbs, Lilly, Pfizer, F. Hoffmann-La Roche Ltd, and travel/accommodations/expenses from AstraZeneca and F. Hoffmann-La Roche Ltd. E.C. is an employee of Roche Products Limited and is named on issued patent ‘Uses for and article of manufacture including HER2 dimerisation inhibitor pertuzumab, 13/649591’. C.B. is a freelance pharmaceutical physician/medical advisor with Barton Oncology Ltd and has undertaken paid consultancy work with Roche Products Limited and many other companies and organisations, including (in the last ~5 years) Apeiron Biologics AG, Cancer Research UK Centre for Drug Development, Cancer Targeting Systems Inc, CellCentric Ltd, Certara LP, Innate Pharma SA, Macrophage Pharma Ltd, MorphoSys AG, Mosaic Biomedicals SL, Norgine Pharmaceuticals Ltd, Ona Therapeutics SL, Orion Clinical Services Ltd, Piqur Therapeutics AG, PTEN Research Foundation, SFL Services GmbH, T3 Pharmaceuticals AG, UCB Biopharma SPRL, and the Wellcome Trust Ltd. She is on the advisory board for SFL Services GmbH and owns shares in GlaxoSmithKline. E.R. is an employee of, and holds shares in, F. Hoffmann-La Roche Ltd. M.Pr.’s institution received funding from F. Hoffmann-La Roche Ltd with respect to the APHINITY study. A.S. reports a consulting/advisory role with Eli Lilly, Pfizer, and Novartis; travel/accommodations/expenses from Neopharm and Celgene; and has been on speaker bureaus for Teva, F. Hoffmann-La Roche Ltd, and Pfizer (all unrelated to the APHINITY study). D.F.’s and A.A.’s institution has received research funding to support the conduct of APHINITY from F. Hoffmann-La Roche Ltd/Genentech, Inc.; and research funding from AstraZeneca, Tesaro, Novartis, Pfizer, F. Hoffmann-La Roche Ltd/Genentech, Inc., and Servier, outside the submitted work. D.P. is a member of the APHINITY Joint Study Management Team and has never received any personal fees for the APHINITY study or outside of the APHINITY study from F. Hoffmann-La Roche Ltd. His institution, Institut Jules Bordet, has received both financial and non-financial (e.g., provision of drugs for study) support for the conduct of the APHINITY study. Institut Jules Bordet has received and still receives research grants or non-financial support outside of APHINITY from F. Hoffmann-La Roche Ltd. J.Ba. was an employee of AstraZeneca, served on the Board of Directors of Foghorn, and was a past board member of Varian Medical Systems, Bristol-Myers Squibb, Grail, Aura Biosciences, and Infinity Pharmaceuticals. He had performed consulting and/or advisory work for Grail, PMV Pharma, ApoGen, Juno, Lilly, Seragon, Novartis, and Northern Biologics. He had stock or other ownership interests in PMV Pharma, Grail, Juno, Varian, Foghorn, Aura, Infinity, and ApoGen, as well as Tango and Venthera, for which he is a co-founder. He had previously received honoraria or travel expenses from F. Hoffmann-La Roche Ltd, Novartis, and Lilly. He had also received from F. Hoffmann-La Roche Ltd non-financial support for studies’ drug supplies and conduct. G.V. has received travel grants and remuneration for advisory board meetings from F. Hoffmann-La Roche Ltd. His institution received investigators’ fees for the APHINITY study. L.L.R. has received airfare, taxi fare, and accommodation payments to attend APHINITY Steering Committee meetings since 2012. E.F. has received <$500 airfare and accommodation payments to attend APHINITY Steering Committee meetings. R.D.G.’s institution receives support for his salary from F. Hoffmann-La Roche Ltd, Pfizer, AstraZeneca, Merck, Ipsen, Ferring, Celgene, and Novartis. He has received travel support payments to attend APHINITY Steering Committee meetings from F. Hoffmann-La Roche Ltd. M.Pi. serves on the Scientific Board for Oncolytic. She has received honoraria for consulting roles for AstraZeneca, Camel-IDS, Crescendo Biologics, Debiopharm, G1 Therapeutics, Genentech, Inc., Huya, Immunomedics, Lilly, Menarini, MSD, Novartis, Odonate, Periphagen, Pfizer, F. Hoffmann-La Roche Ltd, and Seattle Genetics, and her institute has received research grants from AstraZeneca, Lilly, MSD, Novartis, Pfizer, Radius, F. Hoffmann-La Roche Ltd/Genentech, Inc., Servier, and Synthon. C.J. has received travel support payments for Steering Committee meetings, consulting fees, and honoraria from F. Hoffmann-La Roche Ltd for the APHINITY study. His institution received investigators’ fees for the APHINITY study. J.A.P. is an employee of Genentech, Inc. and has shares in F. Hoffmann-La Roche Ltd.
Funding
This work was supported by F. Hoffmann-La Roche Ltd, Basel, Switzerland/Genentech, Inc., South San Francisco, CA, USA (no grant number applicable). F. Hoffmann-La Roche Ltd/Genentech, Inc. was involved in the study design, data interpretation, and the decision to submit for publication in conjunction with the authors.
Consent for publication
Not required.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Bines, J., Clark, E., Barton, C. et al. Patient-reported function, health-related quality of life, and symptoms in APHINITY: pertuzumab plus trastuzumab and chemotherapy in HER2-positive early breast cancer. Br J Cancer 125, 38–47 (2021). https://doi.org/10.1038/s41416-021-01323-y
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41416-021-01323-y
This article is cited by
-
Patterns of self-reported diarrhea in patients with cancer receiving chemotherapy
Supportive Care in Cancer (2025)