Abstract
Background
Despite differences in tumour behaviour and characteristics between duodenal adenocarcinoma (DAC), the intestinal (AmpIT) and pancreatobiliary (AmpPB) subtype of ampullary adenocarcinoma and distal cholangiocarcinoma (dCCA), the effect of adjuvant chemotherapy (ACT) on these cancers, as well as the optimal ACT regimen, has not been comprehensively assessed. This study aims to assess the influence of tailored ACT on DAC, dCCA, AmpIT, and AmpPB.
Patients and methods
Patients after pancreatoduodenectomy for non-pancreatic periampullary adenocarcinoma were identified and collected from 36 tertiary centres between 2010 - 2021. Per non-pancreatic periampullary tumour type, the effect of adjuvant chemotherapy and the main relevant regimens of adjuvant chemotherapy were compared. The primary outcome was overall survival (OS).
Results
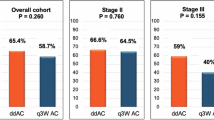
The study included a total of 2866 patients with DAC (n = 330), AmpIT (n = 765), AmpPB (n = 819), and dCCA (n = 952). Among them, 1329 received ACT, and 1537 did not. ACT was associated with significant improvement in OS for AmpPB (P = 0.004) and dCCA (P < 0.001). Moreover, for patients with dCCA, capecitabine mono ACT provided the greatest OS benefit compared to gemcitabine (P = 0.004) and gemcitabine – cisplatin (P = 0.001). For patients with AmpPB, no superior ACT regime was found (P > 0.226). ACT was not associated with improved OS for DAC and AmpIT (P = 0.113 and P = 0.445, respectively).
Discussion
Patients with resected AmpPB and dCCA appear to benefit from ACT. While the optimal ACT for AmpPB remains undetermined, it appears that dCCA shows the most favourable response to capecitabine monotherapy. Tailored adjuvant treatments are essential for enhancing prognosis across all four non-pancreatic periampullary adenocarcinomas.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 24 print issues and online access
$259.00 per year
only $10.79 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
Due to the sensitive nature of the research and the privacy of participants involved, the data supporting the findings of this study are not publicly available. Data can be made available upon reasonable request to the corresponding author, subject to compliance with applicable privacy laws and ethical guidelines.
References
Adsay V, Ohike N, Tajiri T, Kim GE, Krasinskas A, Balci S, et al. Ampullary region carcinomas: definition and site specific classification with delineation of four clinicopathologically and prognostically distinct subsets in an analysis of 249 cases. Am J Surg Pathol. 2012;36:1592–608.
Lerch MM, Domschke W. Abraham Vater of the ampulla (papilla) of Vater. Gastroenterology. 2000;118:379.
Sarmiento JM, Nagomey DM, Sarr MG, Farnell MB. Periampullary cancers: are there differences? Surg Clin North Am. 2001;81:543–55.
Schnelldorfer T, Ware AL, Sarr MG, Smyrk TC, Zhang L, Qin R, et al. Long-term survival after pancreatoduodenectomy for pancreatic adenocarcinoma: is cure possible? Ann Surg. 2008;247:456–62.
Han SS, Jang JY, Kim SW, Kim WH, Lee KU, Park YH. Analysis of long-term survivors after surgical resection for pancreatic cancer. Pancreas. 2006;32:271–5.
Pawlik TM, Gleisner AL, Cameron JL, Winter JM, Assumpcao L, Lillemoe KD, et al. Prognostic relevance of lymph node ratio following pancreaticoduodenectomy for pancreatic cancer. Surgery. 2007;141:610–8.
Zacharias T, Jaeck D, Oussoultzoglou E, Neuville A, Bachellier P. Impact of lymph node involvement on long-term survival after R0 Pancreaticoduodenectomy for ductal Adenocarcinoma of the pancreas. J Gastrointest Surg. 2007;11:350–6.
Neoptolemos JP, Stocken DD, Friess H, Bassi C, Dunn JA, Hickey H, et al. A randomized trial of chemoradiotherapy and chemotherapy after resection of pancreatic cancer. New Engl J Med. 2004;350:1200–10.
Stark AP, Sacks GD, Rochefort MM, Donahue TR, Reber HA, Tomlinson JS, et al. Long-term survival in patients with pancreatic ductal adenocarcinoma. Surgery. 2016;159:1520–7.
Erdmann JI, Eskens FA, Vollmer CM, Kok NF, Groot Koerkamp B, Biermann K, et al. Histological and molecular subclassification of pancreatic and nonpancreatic periampullary cancers: Implications for (Neo) adjuvant systemic treatment. Ann Surg Oncol. 2015;22:2401–7.
Kimura W, Futakawa N, Yamagata S, Wada Y, Kuroda A, Muto T, et al. Different clinicopathologic findings in two histologic types of carcinoma of Papilla of Vater. Japanese Journal of Cancer Research. 1994;85:161–6.
Kimura W, Futakawa N, Zhao B Neoplastic diseases of the papilla of Vater. J Hepatobiliary Pancreat Surg; 11. https://doi.org/10.1007/s00534-004-0894-7 (2004).
Moekotte AL, Lof S, van Roessel S, Fontana M, Dreyer S, Shablak A, et al. Histopathologic predictors of survival and recurrence in resected ampullary Adenocarcinoma: International Multicenter Cohort Study. Ann Surg. 2020;272:1086–93.
Hatzaras I, George N, Muscarella P, Melvin WS, Ellison EC, Bloomston M. Predictors of survival in periampullary cancers following pancreaticoduodenectomy. Ann Surg Oncol. 2010;17:991–7.
Riall TS, Cameron JL, Lillemoe KD, Winter JM, Campbell KA, Hruban RH, et al. Resected periampullary adenocarcinoma: 5-year survivors and their 6- to 10-year follow-up. Surgery. 2006;140:764–72.
Ahn DH, Bekaii-Saab T Ampullary cancer: an overview. Am Soc Clin Oncol Educ Book: 112-5 (2014).
Conroy T, Castan F, Lopez A, Turpin A, Ben Abdelghani M, Wei AC, et al. Five-year outcomes of FOLFIRINOX vs Gemcitabine as adjuvant therapy for pancreatic cancer. JAMA Oncol. 2022;8:1571.
Rizzo A, Brandi G. BILCAP trial and adjuvant capecitabine in resectable biliary tract cancer: reflections on a standard of care. Expert Rev Gastroenterol Hepatol. 2021;15:483–5.
Overman MJ, Varadhachary GR, Kopetz S, Adinin R, Lin E, Morris JS, et al. Phase II study of capecitabine and oxaliplatin for advanced adenocarcinoma of the small bowel and ampulla of Vater. J Clin Oncol. 2009;27:2598–603.
Neoptolemos JP, Moore MJ, Cox TF, Valle JW, Palmer DH, McDonald AC, et al. Effect of adjuvant chemotherapy with fluorouracil plus folinic acid or gemcitabine vs observation on survival in patients with resected periampullary adenocarcinoma: the ESPAC-3 periampullary cancer randomized trial. JAMA. 2012;308:147–56.
Bolm L, Ohrner K, Nappo G, Ruckert F, Zimmermann C, Rau BM, et al. Adjuvant therapy is associated with improved overall survival in patients with pancreatobiliary or mixed subtype ampullary cancer after pancreatoduodenectomy - A multicenter cohort study. Pancreatology. 2020;20:433–41.
Moekotte AL, Malleo G, van Roessel S, Bonds M, Halimi A, Zarantonello L, et al. Gemcitabine-based adjuvant chemotherapy in subtypes of ampullary adenocarcinoma: international propensity score-matched cohort study. Br J Surg. 2020;107:1171–82.
Weiss JM, Gupta S, Burke CA, Axell L, Chen LM, Chung DC, et al. Genetic/familial high-risk assessment: Colorectal, version 1.2021 featured updates to the NCCN Guidelines. JNCCN J Natl Compr Cancer Netw. 2021;19:1122–32.
von Elm E, Altman DG, Egger M, Pocock SJ, Gotzsche PC, Vandenbroucke JP, et al. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) Statement: guidelines for reporting observational studies. Int J Surg. 2014;12:1495–9.
Cuschieri S. The STROBE guidelines. Saudi J Anaesth. 2019;13:S31–S34.
Ament R. Origin of the ASA classification. Anesthesiology. 1979;51:179.
Nagtegaal ID, Odze RD, Klimstra D, Paradis V, Rugge M, Schirmacher P, et al. The 2019 WHO classification of tumours of the digestive system. Histopathology. 2020;76:182–8.
WHO Classiication of Tumours Editorial Board. Digestive system tumours. Lyon (France): International Agency for Research on Cancer; 2019. (WHO classification of tumours series, 5th ed.; vol. 1)…
Edge SB, Compton CC. The American Joint Committee on Cancer: the 7th edition of the AJCC cancer staging manual and the future of TNM. Ann Surg Oncol. 2010;17:1471–4.
Markov P, Satoi S, Kon M. Redefining the R1 resection in patients with pancreatic ductal adenocarcinoma. J Hepatobiliary Pancreat Sci. 2016;23:523–32.
Meijer LL, Alberga AJ, de Bakker JK, van der Vliet HJ, Le Large TYS, van Grieken NCT, et al. Outcomes and treatment options for duodenal Adenocarcinoma: A systematic review and meta-analysis. Ann Surg Oncol. 2018;25:2681–92.
Conroy T, Hammel P, Hebbar M, Ben Abdelghani M, Wei AC, Raoul JL, et al. FOLFIRINOX or Gemcitabine as adjuvant therapy for pancreatic cancer. N Engl J Med. 2018;379:2395–406.
Valle J, Wasan H, Palmer DH, Cunningham D, Anthoney A, Maraveyas A, et al. Cisplatin plus Gemcitabine versus Gemcitabine for biliary tract cancer. New Engl J Med. 2010;362:1273–81.
Ebata T, Hirano S, Konishi M, Uesaka K, Tsuchiya Y, Ohtsuka M, et al. Randomized clinical trial of adjuvant gemcitabine chemotherapy versus observation in resected bile duct cancer. Br J Surg. 2018;105:192–202.
Lonjon G, Porcher R, Ergina P, Fouet M, Boutron I. Potential pitfalls of reporting and bias in observational studies with propensity score analysis assessing a surgical procedure: a methodological systematic review. Ann Surg. 2017;265:901–9.
Sterne JA, White IR, Carlin JB, Spratt M, Royston P, Kenward MG, et al. Multiple imputation for missing data in epidemiological and clinical research: potential and pitfalls. BMJ. 2009;338:b2393.
de Bakker JK, Meijer LL, Zonderhuis BM, van der Vliet HJ, Daams F, van Grieken NCT et al. Adjuvant chemotherapy for resected duodenal adenocarcinoma: a case-matched analysis in nation wide cohort. Acta Chir Belg: 1–7 (2022).
Chiorean EG, Chiaro MDel, Tempero MA, Malafa MP, Benson AB, Cardin DB, et al. Ampullary Adenocarcinoma, Version 1.2023, NCCN Clinical Practice Guidelines in Oncology. J Natl Compr Cancer Netw. 2023;21:753–82.
Roos E, Strijker M, Franken LC, Busch OR, van Hooft JE, Klümpen H-J, et al. Comparison of short- and long-term outcomes between anatomical subtypes of resected biliary tract cancer in a Western high-volume center. HPB. 2020;22:405–14.
Belkouz A, Van Roessel S, Strijker M, van Dam JL, Daamen L, van der Geest LG, et al. Development and external validation of a prediction model for overall survival after resection of distal cholangiocarcinoma. Br J Cancer. https://doi.org/10.1038/s41416-021-01687-1 (2022).
Wellner UF, Shen Y, Keck T, Jin W, Xu Z. The survival outcome and prognostic factors for distal cholangiocarcinoma following surgical resection: a meta-analysis for the 5-year survival. Surg Today. 2017;47:271–9.
Edeline J, Benabdelghani M, Bertaut A, Watelet J, Hammel P, Joly JP, et al. Gemcitabine and Oxaliplatin chemotherapy or surveillance in resected biliary tract cancer (PRODIGE 12-ACCORD 18-UNICANCER GI): A Randomized Phase III Study. J Clin Oncol. 2019;37:658–67.
Edeline J, Hirano S, Bertaut A, Konishi M, Benabdelghani M, Uesaka K, et al. Individual patient data meta-analysis of adjuvant gemcitabine-based chemotherapy for biliary tract cancer: combined analysis of the BCAT and PRODIGE-12 studies. Eur J Cancer. 2022;164:80–87.
Shroff RT, Kennedy EB, Bachini M, Bekaii-Saab T, Crane C, Edeline J, et al. Adjuvant therapy for resected biliary tract cancer: ASCO Clinical Practice Guideline. J Clin Oncol. 2019;37:1015–27.
Nakachi K, Konishi M, Ikeda M, Mizusawa J, Eba J, Okusaka T, et al. A randomized Phase III trial of adjuvant S-1 therapy vs. observation alone in resected biliary tract cancer: Japan Clinical Oncology Group Study (JCOG1202, ASCOT). Jpn J Clin Oncol. 2018;48:392–5.
Stein A, Arnold D, Bridgewater J, Goldstein D, Jensen LH, Klumpen HJ, et al. Adjuvant chemotherapy with gemcitabine and cisplatin compared to observation after curative intent resection of cholangiocarcinoma and muscle invasive gallbladder carcinoma (ACTICCA-1 trial) - a randomized, multidisciplinary, multinational phase III trial. BMC Cancer. 2015;15:564.
Body A, Prenen H, Latham S, Lam M, Tipping-Smith S, Raghunath A, et al. The role of neoadjuvant chemotherapy in locally advanced colon cancer. Cancer Manag Res. 2021;ume 13:2567–79.
Gumbs AA, Croner R, Abu-Hilal M, Bannone E, Ishizawa T, Spolverato G, et al. Surgomics and the Artificial intelligence, Radiomics, Genomics, Oncopathomics and Surgomics (AiRGOS) Project. Artificial Intelligence Surgery. 2023;3:180–5.
Funding
No specific funding needed.
Author information
Authors and Affiliations
Consortia
Contributions
Study conception and design: BU, DL, MG, HW, AZ, CF, MB, MA. Acquisition of data: GF, AZ, BK, ML, PG, RS, SW, BI, WG, UB, GK, MH, VM, BB, MM, MS, DK, AA, KR, ZS, PP, WF, SK, TK, MV, LB, NJ, RD, JK, AM, MM, SC, CB, AB, Analysis and interpretation of data: BU, DL, MG, HW, AZ, CF, MB, MA. Drafting of manuscript: BU, DL, MG, HW, AZ, CF, MB, MA. Critical revision: BU, DL, MG, HW, AZ, GF, AZ, BK, ML, PG, RS, SW, BI, WG, UB, GK, MH, VM, BB, MM, MS, DK, AA, KR, ZS, PP, WF, SK, TK, MV, LB, NJ, RD, JK, AM, MM, SC, CB, AB, CF, MB, MA.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
The study was approved by the Ethical Committee of Brescia, Italy (number NP 5269 - STUDIO NPPC 15.03.2022). The need for written informed consent was waived due to the retrospective nature of the research.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Uijterwijk, B.A., Lemmers, D.H., Ghidini, M. et al. The road to tailored adjuvant chemotherapy for all four non-pancreatic periampullary cancers: An international multimethod cohort study. Br J Cancer 131, 117–125 (2024). https://doi.org/10.1038/s41416-024-02692-w
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41416-024-02692-w
This article is cited by
-
The Landscape of Genomic Alterations in Receptor Tyrosine Kinase Pathways in Biliary Cancers: Implications for Targeted Therapies
Journal of Gastrointestinal Cancer (2025)