Abstract
Background
Poly (ADP-ribose) polymerase inhibitors (PARPis) can effectively treat ovarian cancer patients with defective homologous recombination (HR). Loss or dysfunction of PTEN, a typical tumour suppressor, impairs double-strand break (DSB) repair. Hence, we explored the possibility of inhibiting PTEN to induce HR deficiency (HRD) for PARPi application.
Methods
Functional studies using PTEN inhibitor VO-OHpic and PARPi olaparib were performed to explore the molecular mechanisms in vitro and in vivo.
Results
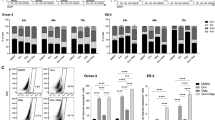
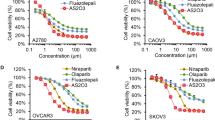
In this study, the combination of VO-OHpic with olaparib exhibited synergistic inhibitory effects on ovarian cancer cells was demonstrated. Furthermore, VO-OHpic was shown to enhance DSBs by reducing nuclear expression of PTEN and inhibiting HR repair through the modulation of MRE11-RAD50-NBN (MRN) complex, critical for DSB repair. TCGA and GTEx analysis revealed a strong correlation between PTEN and MRN in ovarian cancer. Mechanistic studies indicated that VO-OHpic reduced expression of MRN, likely by decreasing PTEN/E2F1-mediated transcription. Moreover, PTEN-knockdown inhibited expression of MRN, increased sensitivities to olaparib, and induced DSBs. In vivo experiments showed that the combination of VO-OHpic with olaparib exhibited enhanced inhibitory effects on tumour growth.
Conclusions
Collectively, this study highlights the potential of PTEN inhibitors in combination therapy with PARPis to create HRD for HRD-negative ovarian cancers.

This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 24 print issues and online access
$259.00 per year
only $10.79 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Zhang H, Lu B. microRNAs as biomarkers of ovarian cancer. Expert Rev Anticancer Ther. 2020;20:373–85.
Dion L, Carton I, Jaillard S, Nyangoh Timoh K, Henno S, Sardain H, et al. The landscape and therapeutic implications of molecular profiles in epithelial ovarian cancer. J Clin Med. 2020;9:2239.
Liu L, Liu P, Liang Z, Li R, Shen M, Xu H, et al. Poly (ADP-ribose) polymerase inhibitors combined with other small-molecular compounds for the treatment of ovarian cancer. Anticancer Drugs. 2019;30:554–61.
Lee A. Fuzuloparib: first approval. Drugs. 2021;81:1221–6.
Yamashita T. [Treatment of delusions of parasitosis: efficacy of transcutaneous electrical stimulation]. Seishin Shinkeigaku Zasshi. 1986;88:1–13.
Bryant HE, Schultz N, Thomas HD, Parker KM, Flower D, Lopez E, et al. Specific killing of BRCA2-deficient tumours with inhibitors of poly(ADP-ribose) polymerase. Nature. 2005;434:913–7.
Gelmon KA, Tischkowitz M, Mackay H, Swenerton K, Robidoux A, Tonkin K, et al. Olaparib in patients with recurrent high-grade serous or poorly differentiated ovarian carcinoma or triple-negative breast cancer: a phase 2, multicentre, open-label, non-randomised study. Lancet Oncol. 2011;12:852–61.
Bitler BG, Watson ZL, Wheeler LJ, Behbakht K. PARP inhibitors: clinical utility and possibilities of overcoming resistance. Gynecol Oncol. 2017;147:695–704.
Chow JT, Salmena L. Recent advances in PTEN signalling axes in cancer. Fac Rev. 2020;9:31.
Shen WH, Balajee AS, Wang J, Wu H, Eng C, Pandolfi PP, et al. Essential role for nuclear PTEN in maintaining chromosomal integrity. Cell. 2007;128:157–70.
Planchon SM, Waite KA, Eng C. The nuclear affairs of PTEN. J Cell Sci. 2008;121:249–53.
Ming M, He YY. PTEN in DNA damage repair. Cancer Lett. 2012;319:125–9.
Mendes-Pereira AM, Martin SA, Brough R, McCarthy A, Taylor JR, Kim JS, et al. Synthetic lethal targeting of PTEN mutant cells with PARP inhibitors. EMBO Mol Med. 2009;1:315–22.
McEllin B, Camacho CV, Mukherjee B, Hahm B, Tomimatsu N, Bachoo RM, et al. PTEN loss compromises homologous recombination repair in astrocytes: implications for glioblastoma therapy with temozolomide or poly(ADP-ribose) polymerase inhibitors. Cancer Res. 2010;70:5457–64.
Dedes KJ, Wetterskog D, Mendes-Pereira AM, Natrajan R, Lambros MB, Geyer FC, et al. PTEN deficiency in endometrioid endometrial adenocarcinomas predicts sensitivity to PARP inhibitors. Sci Transl Med. 2010;2:53ra75.
Hopkins BD, Hodakoski C, Barrows D, Mense SM, Parsons RE. PTEN function: the long and the short of it. Trends Biochem Sci. 2014;39:183–90.
Tamura M, Gu J, Matsumoto K, Aota S, Parsons R, Yamada KM. Inhibition of cell migration, spreading, and focal adhesions by tumor suppressor PTEN. Science. 1998;280:1614–7.
Liu H, Lu Z, Shi X, Liu L, Zhang P, Golemis EA, et al. HSP90 inhibition downregulates DNA replication and repair genes via E2F1 repression. J Biol Chem. 2021;297:100996.
Zhao Z, Shang D, Qiu L, Guo C, Li Y, Liu H, et al. 4,5-Diphenyl-2-methyl picolinate induces cellular senescence by accumulating DNA damage and activating associated signaling pathways in gastric cancer. Life Sci. 2019;238:116973.
Chen S, Gu T, Lu Z, Qiu L, Xiao G, Zhu X, et al. Roles of MYC-targeting long non-coding RNA MINCR in cell cycle regulation and apoptosis in non-small cell lung Cancer. Respir Res. 2019;20:202.
Shang DS, Zhou TC, Zhuang XY, Wu YF, Liu HQ, Tu ZG. Molecular dissection on inhibition of Ras-induced cellular senescence by small t antigen of SV40. Cell Mol Life Sci. 2022;79:242.
Tang Z, Kang B, Li C, Chen T, Zhang Z. GEPIA2: an enhanced web server for large-scale expression profiling and interactive analysis. Nucleic Acids Res. 2019;47:W556–60.
Lanczky A, Gyorffy B. Web-based survival analysis tool tailored for medical research (KMplot): development and implementation. J Med Internet Res. 2021;23:e27633.
Chandrashekar DS, Bashel B, Balasubramanya SAH, Creighton CJ, Ponce-Rodriguez I, Chakravarthi B, et al. UALCAN: a portal for facilitating tumor subgroup gene expression and survival analyses. Neoplasia. 2017;19:649–58.
Tu Z, Aird KM, Bitler BG, Nicodemus JP, Beeharry N, Xia B, et al. Oncogenic Ras regulates BRIP1 expression to induce dissociation of BRCA1 from chromatin, inhibit DNA repair, and promote senescence. Dev Cell. 2011;21:1077–91.
Lu Z, Yuan S, Ruan L, Tu Z, Liu H. Partitioning defective 6 homolog alpha (PARD6A) promotes epithelial-mesenchymal transition via integrin β1-ILK-SNAIL1 pathway in ovarian cancer. Cell Death Dis. 2022;13:304.
Ledermann JA, Drew Y, Kristeleit RS. Homologous recombination deficiency and ovarian cancer. Eur J Cancer. 2016;60:49–58.
Boussios S, Karihtala P, Moschetta M, Karathanasi A, Sadauskaite A, Rassy E, et al. Combined strategies with poly (ADP-ribose) polymerase (PARP) inhibitors for the treatment of ovarian cancer: a literature review. Diagnostics. 2019;9:87.
Arora S, Balasubramaniam S, Zhang H, Berman T, Narayan P, Suzman D, et al. FDA approval summary: olaparib monotherapy or in combination with bevacizumab for the maintenance treatment of patients with advanced ovarian cancer. Oncologist. 2021;26:e164–72.
Alfieri R, Giovannetti E, Bonelli M, Cavazzoni A. New treatment opportunities in phosphatase and tensin homolog (PTEN)-deficient tumors: focus on PTEN/focal adhesion kinase pathway. Front Oncol. 2017;7:170.
Lee YR, Chen M, Pandolfi PP. The functions and regulation of the PTEN tumour suppressor: new modes and prospects. Nat Rev Mol Cell Biol. 2018;19:547–62.
Li S, Zhu M, Pan R, Fang T, Cao YY, Chen S, et al. The tumor suppressor PTEN has a critical role in antiviral innate immunity. Nat Immunol. 2016;17:241–9.
Bian L, Meng Y, Zhang M, Li D. MRE11-RAD50-NBS1 complex alterations and DNA damage response: implications for cancer treatment. Mol Cancer. 2019;18:169.
Elkholi IE, Foulkes WD, Rivera B. MRN complex and cancer risk: old bottles, new wine. Clin Cancer Res. 2021;27:5465–71.
Syed A, Tainer JA. The MRE11-RAD50-NBS1 complex conducts the orchestration of damage signaling and outcomes to stress in DNA replication and repair. Annu Rev Biochem. 2018;87:263–94.
Yuan J, Song Y, Pan W, Li Y, Xu Y, Xie M, et al. LncRNA SLC26A4-AS1 suppresses the MRN complex-mediated DNA repair signaling and thyroid cancer metastasis by destabilizing DDX5. Oncogene. 2020;39:6664–76.
Li J, Yen C, Liaw D, Podsypanina K, Bose S, Wang SI, et al. PTEN, a putative protein tyrosine phosphatase gene mutated in human brain, breast, and prostate cancer. Science. 1997;275:1943–7.
Steck PA, Pershouse MA, Jasser SA, Yung WK, Lin H, Ligon AH, et al. Identification of a candidate tumour suppressor gene, MMAC1, at chromosome 10q23.3 that is mutated in multiple advanced cancers. Nat Genet. 1997;15:356–62.
Rosivatz E, Matthews JG, McDonald NQ, Mulet X, Ho KK, Lossi N, et al. A small-molecule inhibitor for phosphatase and tensin homologue deleted on chromosome 10 (PTEN). ACS Chem Biol. 2006;1:780–90.
Bassi C, Ho J, Srikumar T, Dowling RJ, Gorrini C, Miller SJ, et al. Nuclear PTEN controls DNA repair and sensitivity to genotoxic stress. Science. 2013;341:395–9.
Ho J, Cruise ES, Dowling RJO, Stambolic V. PTEN nuclear functions. Cold Spring Harb Perspect Med. 2020;10:a036079.
Shi LG, Li BC, Zhang BN, Zhen CY, Zhou JD, Tang SJ. Mouse embryonic palatal mesenchymal cells maintain stemness through the PTEN-Akt-mTOR autophagic pathway. Stem Cell Res Ther. 2019;10:217.
Kim SJ, Lee HW, Baek JH, Cho YH, Kang HG, Jeong JS, et al. Activation of nuclear PTEN by inhibition of Notch signaling induces G2/M cell cycle arrest in gastric cancer. Oncogene. 2016;35:251–60.
Wu Y, Zhou H, Wu K, Lee S, Li RJ, Liu X. PTEN phosphorylation and nuclear export mediate free fatty acid-induced oxidative stress. Antioxid Redox Signal. 2014;20:1382–95.
Salmena L, Carracedo A, Pandolfi PP. Tenets of PTEN tumor suppression. Cell. 2008;133:403–14.
The Cancer Genome Atlas Research Network. Integrated genomic analyses of ovarian carcinoma. Nature. 2011;474:609–15.
Pennington KP, Walsh T, Harrell MI, Lee MK, Pennil CC, Rendi MH, et al. Germline and somatic mutations in homologous recombination genes predict platinum response and survival in ovarian, fallopian tube, and peritoneal carcinomas. Clin Cancer Res. 2014;20:764–75.
Brandt S, Samartzis EP, Zimmermann AK, Fink D, Moch H, Noske A, et al. Lack of MRE11-RAD50-NBS1 (MRN) complex detection occurs frequently in low-grade epithelial ovarian cancer. BMC Cancer. 2017;17:44.
Koppensteiner R, Samartzis EP, Noske A, von Teichman A, Dedes I, Gwerder M, et al. Effect of MRE11 loss on PARP-inhibitor sensitivity in endometrial cancer in vitro. PLoS ONE. 2014;9:e100041.
Lu HM, Li S, Black MH, Lee S, Hoiness R, Wu S, et al. Association of breast and ovarian cancers with predisposition genes identified by large-scale sequencing. JAMA Oncol. 2019;5:51–7.
Lajud SA, Nagda DA, Yamashita T, Zheng J, Tanaka N, Abuzeid WM, et al. Dual disruption of DNA repair and telomere maintenance for the treatment of head and neck cancer. Clin Cancer Res. 2014;20:6465–78.
Oplustilova L, Wolanin K, Mistrik M, Korinkova G, Simkova D, Bouchal J, et al. Evaluation of candidate biomarkers to predict cancer cell sensitivity or resistance to PARP-1 inhibitor treatment. Cell Cycle. 2012;11:3837–50.
De Lorenzo SB, Patel AG, Hurley RM, Kaufmann SH. The elephant and the blind men: making sense of PARP inhibitors in homologous recombination deficient tumor cells. Front Oncol. 2013;3:228.
Miller RE, El-Shakankery KH, Lee JY. PARP inhibitors in ovarian cancer: overcoming resistance with combination strategies. J Gynecol Oncol. 2022;33:e44.
Shibata A, Moiani D, Arvai AS, Perry J, Harding SM, Genois MM, et al. DNA double-strand break repair pathway choice is directed by distinct MRE11 nuclease activities. Mol Cell. 2014;53:7–18.
Yang YG, Saidi A, Frappart PO, Min W, Barrucand C, Dumon-Jones V, et al. Conditional deletion of Nbs1 in murine cells reveals its role in branching repair pathways of DNA double-strand breaks. EMBO J. 2006;25:5527–38.
Karakashev S, Fukumoto T, Zhao B, Lin J, Wu S, Fatkhutdinov N, et al. EZH2 inhibition sensitizes CARM1-high, homologous recombination proficient ovarian cancers to PARP inhibition. Cancer Cell. 2020;37:157–67.e6.
Piscitello D, Varshney D, Lilla S, Vizioli MG, Reid C, Gorbunova V, et al. AKT overactivation can suppress DNA repair via p70S6 kinase-dependent downregulation of MRE11. Oncogene. 2018;37:427–38.
Funding
This work was supported by the National Natural Science Foundation of China [Grant number 81672582 (to HL) and 31771521 (to ZT)]; Top Talent of Innovative Research Team of Jiangsu Province (to HL and ZT); the Natural Science Foundation of Jiangsu Province [Grant number BE2016718 (to ZT)]; the Training Project for Young Backbone Teachers of Jiangsu University (to LQ).
Author information
Authors and Affiliations
Contributions
Experimental design and supervision: ZT and HL; Data analysis and interpretation: LQ, RL, and YW; Bioinformatic correlation analysis: LQ; In vitro assays: RL, YW, and LQ; In vivo experiments: ZL; Paper writing: LQ, ZT, and HL.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
The animal study was reviewed and approved by Bioethics Committee of Jiangsu University. All methods were performed in accordance with the relevant guidelines and regulations.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Qiu, L., Li, R., Wang, Y. et al. PTEN inhibition enhances sensitivity of ovarian cancer cells to the poly (ADP-ribose) polymerase inhibitor by suppressing the MRE11-RAD50-NBN complex. Br J Cancer 131, 577–588 (2024). https://doi.org/10.1038/s41416-024-02749-w
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41416-024-02749-w