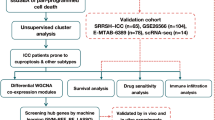
Abstract
Background
Targeted therapy for intrahepatic cholangiocarcinoma (ICC) shows superior survival outcomes but patients with certain targetable alterations are no more than 20%. Genetic alteration screening for all ICC patients is of high cost and not routinely performed. This study intends to develop a histopathology-based artificial intelligence (AI)-assisted system for predicting genetic alteration of ICC.
Methods
We constructed a Genetic Alteration Prediction (GAP) system based on multi-instance learning and self-supervised learning to predict genetic alterations using whole-slide images (WSIs) of H&E-stained slides. A total of 2069 WSIs from 232 ICC patients underwent surgery of the FAH-SYSU dataset were used for model construction and adjustment by five-fold cross-validation. Another 150 patients from three medical centres were used as independent external validations. We also compared the cost-effectiveness of GAP-assisted precise treatment and all-sequencing strategy to non-sequencing strategy.
Results
The GAP was able to predict actionable genetic alterations of ICC, including FGFR2 and IDH. The area under the receiver operating characteristic curves (AUC) for FGFR2 and IDH were 0.754 and 0.713 in the internal dataset, and 0.724 and 0.656 in the external dataset, respectively. Furthermore, compared to giving chemotherapy without sequencing for every patient, GAP-assisted precise treatment could increase 1 progression-free quality-adjusted life month with a cost of $13871.72, the co-responding figure for all-sequencing strategy is $44538.93. Decision curve analysis showed that AI-assisted strategy provides better clinical benefits.
Conclusions
We constructed an AI-assisted genetic alteration screening system which is predictable to ICC actionable targets and has potential to assist precise targeted treatment of advanced ICC.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 24 print issues and online access
$259.00 per year
only $10.79 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Valle JW, Kelley RK, Nervi B, Oh DY, Zhu AX. Biliary tract cancer. Lancet. 2021;397:428–44.
Sirica AE, Gores GJ, Groopman JD, Selaru FM, Strazzabosco M, Wei Wang X, et al. Intrahepatic cholangiocarcinoma: continuing challenges and translational advances. Hepatology. 2019;69:1803–15.
Kelley RK, Bridgewater J, Gores GJ, Zhu AX. Systemic therapies for intrahepatic cholangiocarcinoma. J Hepatol. 2020;72:353–63.
Verlingue L, Malka D, Allorant A, Massard C, Ferté C, Lacroix L, et al. Precision medicine for patients with advanced biliary tract cancers: an effective strategy within the prospective MOSCATO-01 trial. Eur J Cancer. 2017;87:122–30.
Lamarca A, Barriuso J, McNamara MG, Valle JW. Molecular targeted therapies: ready for “prime time” in biliary tract cancer. J Hepatol. 2020;73:170–85.
Silverman IM, Hollebecque A, Friboulet L, Owens S, Newton RC, Zhen H, et al. Clinicogenomic analysis of FGFR2-rearranged cholangiocarcinoma identifies correlates of response and mechanisms of resistance to Pemigatinib. Cancer Discov. 2021;11:326–39.
Rusch M, Nakitandwe J, Shurtleff S, Newman S, Zhang Z, Edmonson MN, et al. Clinical cancer genomic profiling by three-platform sequencing of whole genome, whole exome and transcriptome. Nat Commun. 2018;9:3962.
Wentzensen N, Lahrmann B, Clarke MA, Kinney W, Tokugawa D, Poitras N, et al. Accuracy and efficiency of deep-learning-based automation of dual stain cytology in cervical cancer screening. J Natl Cancer Inst. 2021;113:72–9.
Kather JN, Pearson AT, Halama N, Jäger D, Krause J, Loosen SH, et al. Deep learning can predict microsatellite instability directly from histology in gastrointestinal cancer. Nat Med. 2019;25:1054–6.
Shia J, Schultz N, Kuk D, Vakiani E, Middha S, Segal NH, et al. Morphological characterization of colorectal cancers in The Cancer Genome Atlas reveals distinct morphology-molecular associations: clinical and biological implications. Mod Pathol. 2017;30:599–609.
Kather JN, Heij LR, Grabsch HI, Loeffler C, Echle A, Muti HS, et al. Pan-cancer image-based detection of clinically actionable genetic alterations. Nat Cancer. 2020;1:789–99.
Chen M, Zhang B, Topatana W, Cao J, Zhu H, Juengpanich S, et al. Classification and mutation prediction based on histopathology H&E images in liver cancer using deep learning. NPJ Precis Oncol. 2020;4:14.
Coudray N, Ocampo PS, Sakellaropoulos T, Narula N, Snuderl M, Fenyö D, et al. Classification and mutation prediction from non-small cell lung cancer histopathology images using deep learning. Nat Med. 2018;24:1559–67.
Nagtegaal ID, Odze RD, Klimstra D, Paradis V, Rugge M, Schirmacher P, et al. The 2019 WHO classification of tumours of the digestive system. Histopathology. 2020;76:182–8.
Huang G, Liu Z, van der Maaten L, Weinberger KQ, Ieee. Densely connected convolutional networks. 30th IEEE/CVF Conference on Computer Vision and Pattern Recognition (CVPR) 2017:2261–9.
Szegedy C, Vanhoucke V, Ioffe S, Shlens J, Wojna Z, Ieee. Rethinking the inception architecture for computer vision. 2016 IEEE Conference on Computer Vision and Pattern Recognition (CVPR) 2016:2818–26.
He KM, Zhang XY, Ren SQ, Sun J, Ieee. Deep residual learning for image recognition. 2016 IEEE Conference on Computer Vision and Pattern Recognition (CVPR). 2016:770–8.
Simonyan K, Zisserman A. Very deep convolutional networks for large-scale image recognition. arXiv: 1409.1556, Preprint posted online 4 September 2014, http://arxiv.org/abs/1409.1556 (2014).
van der Maaten L. Accelerating t-SNE using tree-based algorithms. J Mach Learn Res. 2014;15:3221–45.
Zhao J, Lu D, Ma K, Zhang Y, Zheng Y. Deep image clustering with category-style representation. European conference on computer vision(ECCV 2020). 2020;54-70.
Zhou B, Khosla A, Lapedriza A, Oliva A, Torralba A, Ieee. Learning deep features for discriminative localization. 2016 IEEE Conference on Computer Vision and Pattern Recognition (CVPR) 2016:2921–9.
Pedregosa F, Varoquaux G, Gramfort A, Michel V, Thirion B, Grisel O, et al. Scikit-learn: machine learning in Python. J Mach Learn Res. 2011;12:2825–30.
Mosele F, Remon J, Mateo J, Westphalen CB, Barlesi F, Lolkema MP, et al. Recommendations for the use of next-generation sequencing (NGS) for patients with metastatic cancers: a report from the ESMO Precision Medicine Working Group. Ann Oncol. 2020;31:1491–505.
Saha SK, Parachoniak CA, Ghanta KS, Fitamant J, Ross KN, Najem MS, et al. Mutant IDH inhibits HNF-4α to block hepatocyte differentiation and promote biliary cancer. Nature. 2014;513:110–4.
Javle M, Bekaii-Saab T, Jain A, Wang Y, Kelley RK, Wang K, et al. Biliary cancer: utility of next-generation sequencing for clinical management. Cancer. 2016;122:3838–47.
Helsten T, Elkin S, Arthur E, Tomson BN, Carter J, Kurzrock R. The FGFR landscape in cancer: analysis of 4,853 tumors by next-generation sequencing. Clin Cancer Res. 2016;22:259–67.
Zhang X, Liu J, Liang X, Chen J, Hong J, Li L, et al. History and progression of Fat cadherins in health and disease. Onco Targets Ther. 2016;9:7337–43.
Luchini C, Robertson SA, Hong SM, Felsenstein M, Anders RA, Pea A, et al. PBRM1 loss is a late event during the development of cholangiocarcinoma. Histopathology. 2017;71:375–82.
Jiao Y, Pawlik TM, Anders RA, Selaru FM, Streppel MM, Lucas DJ, et al. Exome sequencing identifies frequent inactivating mutations in BAP1, ARID1A and PBRM1 in intrahepatic cholangiocarcinomas. Nat Genet. 2013;45:1470–3.
Guardia T, Eason M, Kontrogianni-Konstantopoulos A. Obscurin: a multitasking giant in the fight against cancer. Biochim Biophys Acta Rev Cancer. 2021;1876:188567.
Calderaro J, Ziol M, Paradis V, Zucman-Rossi J. Molecular and histological correlations in liver cancer. J Hepatol. 2019;71:616–30.
Kendall T, Verheij J, Gaudio E, Evert M, Guido M, Goeppert B, et al. Anatomical, histomorphological and molecular classification of cholangiocarcinoma. Liver Int. 2019;39:7–18.
Wang T, Drill E, Vakiani E, Pak LM, Boerner T, Askan G, et al. Distinct histomorphological features are associated with IDH1 mutation in intrahepatic cholangiocarcinoma. Hum Pathol. 2019;91:19–25.
Acknowledgements
We thank all participants for their endeavour and contribution in this study. We thank all medical centres involved for their support of this research project.
Funding
The work was supported by the National Key Research and Development Program of China (No. 2020AAA0109504), the National Science Foundation for Distinguished Young Scholars (Nos. 81825013, 82322034), the National Natural Science Foundation of China (Nos. 82272942, 82302283, 82172047), the Guangdong Natural Science Foundation of Distinguished Youth Scholar (No. 2022B1515020060), the Natural Science Foundation of Guangdong Province (No. 2021A1515010450), and the Kelin Outstanding Young Scientist of the First Affiliated Hospital, Sun Yat-sen University (No. R08030).
Author information
Authors and Affiliations
Contributions
Project administration: Peng S; Conceptualisation: Xiao H, Wang. JP, Weng ZP, Chen SL and Peng S; Data curation: Xiao H, Wang. JP, Weng ZP, Shu M, Shen JX, Sun P, Cai MY, Xiang X, Wei LH, Lai JM and Yun JP; Writing—original draft: Xiao H, Wang JP and Weng ZP; Writing—review & editing: Xiao H, Wang JP, Chen SL, Kuang M and Peng S; Formal analysis: Weng ZP, Lin XX, Li B and Shi YY; Methodology: Weng ZP, Lin XX, Li B and Shi YY; Investigation: Xiao H, Wang JP, Chen SL and Peng S; Resources: Shi YY, Kuang M and Peng S; Validation: Shen JX, Sun P, Cai MY, Xiang X, Lai JM and Yun JP; Supervision: Yun JP, Chen SL and Peng S; Funding acquisition: Xiao H, Kuang M, Chen SL and Peng S.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Ethical approval
This study was conducted ethically in accordance with the World Medical Association Declaration of Helsinki. The protocol was reviewed and approved by Ethics Committee of the First Affiliated Hospital of Sun Yat-sen University, approval number No. [2023]107.
Informed consent
Informed consent was waived since it’s a retrospective study.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Xiao, H., Wang, J., Weng, Z. et al. A histopathology-based artificial intelligence system assisting the screening of genetic alteration in intrahepatic cholangiocarcinoma. Br J Cancer 132, 195–202 (2025). https://doi.org/10.1038/s41416-024-02910-5
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41416-024-02910-5
This article is cited by
-
Smart Diagnosis of Cholangiocarcinoma from Microscopic Images Using a Modified Visual Geometry Group Network with Adaptive Augmentation
International Journal of Computational Intelligence Systems (2025)
-
From slide analysis to precision strategy: the pathologist in the artificial intelligence loop for liver disease diagnosis and patient management
Surgical and Experimental Pathology (2025)