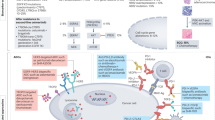
Abstract
Background
Although immune checkpoint inhibitors (ICIs) have been successfully utilized in patients with non-small cell lung cancer (NSCLC), EGFR-mutated patients didn’t benefit from ICIs. The underlying mechanisms for the poor efficacy of this subgroup remain unclear.
Methods
CD8+T cells cytotoxicity, DCs phagocytosis and immunofluorescence assay were applied to examine the immunosuppressive microenvironment of NSCLC. m6A RNA immunoprecipitation, luciferase assay and immunohistochemistry were used to explore the relationship between CD47 and ALKBH5 in EGFR-TKI resistant NSCLC. Autochthonous EGFR-driven lung tumor mouse model and PDXs were performed to explore the therapeutic potential of CD47 antibody and EGFR-TKI combination.
Results
We found that EGFR-TKI resistance promoted a more immunosuppressive tumor microenvironment and inhibited anti-tumor functions of CD8+ T cells. Mechanistically, the m6A eraser ALKBH5 was inhibited in EGFR-TKI resistant NSCLC, which subsequently upregulates CD47 by catalyzing m6A demethylation and causes immunosuppression. Combined treatment with EGFR-TKI and inhibitors of CD47 enhances antitumor immunity and EGFR-TKI efficacy in vivo.
Conclusions
Collectively, our findings reveal the possible underlying mechanism for poor immune response of ICIs in EGFR-TKI resistant NSCLC and provide preclinical evidence that targeted therapy combined with innate immune checkpoint blockade may provide synergistic effects in NSCLC treatment.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 24 print issues and online access
$259.00 per year
only $10.79 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Gahr S, Stoehr R, Geissinger E, Ficker JH, Brueckl WM, Gschwendtner A, et al. EGFR mutational status in a large series of Caucasian European NSCLC patients: data from daily practice. Br J Cancer. 2013;109:1821–28.
Shi Y, Au JSK, Thongprasert S, Srinivasan S, Tsai CM, Khoa MT, et al. A prospective, molecular epidemiology study of egfr mutations in asian patients with advanced non-small-cell lung cancer of adenocarcinoma histology (PIONEER). J Thorac Oncol. 2014;9:154–62.
Ohashi K, Maruvka YE, Michor F, Pao W. Epidermal growth factor receptor tyrosine kinase inhibitor-resistant disease. J Clin Oncol. 2013;31:1070–80.
Brahmer J, Reckamp KL, Baas P, Crinò L, Eberhardt WEE, Poddubskaya E, et al. Nivolumab versus Docetaxel in Advanced Squamous-Cell Non–Small-Cell Lung Cancer. N Engl J Med. 2015;373:123–35.
Borghaei H, Paz-Ares L, Horn L, Spigel DR, Steins M, Ready NE, et al. Nivolumab versus docetaxel in advanced nonsquamous non-small-cell lung cancer. N Engl J Med. 2015;373:1627–39.
Herbst RS, Baas P, Kim DW, Felip E, Pérez-Gracia JL, Han JY, et al. Pembrolizumab versus docetaxel for previously treated, PD-L1-positive, advanced non-small-cell lung cancer (KEYNOTE-010): a randomised controlled trial. Lancet. 2016;387:1540–50.
Fehrenbacher L, Spira A, Ballinger M, Kowanetz M, Vansteenkiste J, Mazieres J, et al. Atezolizumab versus docetaxel for patients with previously treated non-small-cell lung cancer (POPLAR): a multicentre, open-label, phase 2 randomised controlled trial. Lancet. 2016;387:1837–46.
Rittmeyer A, Barlesi F, Waterkamp D, Park K, Ciardiello F, von Pawel J, et al. Atezolizumab versus docetaxel in patients with previously treated non-small-cell lung cancer (OAK): a phase 3, open-label, multicentre randomised controlled trial. Lancet. 2017;389:255–65.
Reck M, Rodríguez-Abreu D, Robinson AG, Hui RN, Csoszi T, Fülöp A, et al. Pembrolizumab versus chemotherapy for PD-L1-positive non-small-cell lung cancer. N Engl J Med. 2016;375:1823–33.
Lee CK, Man J, Lord S, Links M, Gebski V, Mok T, et al. Checkpoint inhibitors in metastatic EGFR- mutated non–small cell lung cancer—a meta-analysis. J Thorac Oncol. 2017;12:403–7.
Garassino MC, Gelibter AJ, Grossi F, Chiari R, Parra HS, Cascinu S, et al. Italian nivolumab expanded access program in nonsquamous non-small cell lung cancer patients: results in never-smokers and EGFR-Mutant Patients. J Thorac Oncol. 2018;13:1146–55.
Mazieres J, Drilon A, Lusque A, Mhanna L, Cortot AB, Mezquita L, et al. Immune checkpoint inhibitors for patients with advanced lung cancer and oncogenic driver alterations: results from the IMMUNOTARGET registry. Ann Oncol. 2019;30:1321–8.
Matlung HL, Szilagyi K, Barclay NA, van den Berg TK. The CD47-SIRPα signaling axis as an innate immune checkpoint in cancer. Immunological Rev. 2017;276:145–64.
Willingham SB, Volkmer JP, Gentles AJ, Sahoo D, Dalerba P, Mitra SS, et al. The CD47-signal regulatory protein alpha (SIRPa) interaction is a therapeutic target for human solid tumors. Proc Natl Acad Sci USA. 2012;109:6662–7.
Hu L-Y, Zhuang W-T, Chen M-J, Liao J, Wu D-F, Zhang Y-X, et al. EGFR oncogenic mutations in NSCLC impair macrophage phagocytosis and mediate innate immune evasion through up-regulation of CD47. J Thorac Oncol. 2024;19:1186–200.
Lin HY, Fang ZX, Su YH, Li PH, Wang JK, Liao HF, et al. DHX32 promotes angiogenesis in colorectal cancer through augmenting β-catenin signaling to induce expression of VEGFA. EBioMedicine. 2017;18:62–72.
Su SC, Zhao JH, Xing Y, Zhang XQ, Liu J, Ouyang Q, et al. Immune checkpoint inhibition overcomes ADCP-induced immunosuppression by macrophages. Cell. 2018;175:442–57.
Su SC, Liao JY, Liu J, Huang D, He CH, Chen F, et al. Blocking the recruitment of naive CD4 T cells reverses immunosuppression in breast cancer. Cell Res. 2017;27:461–82.
Pirazzoli V, Ayeni D, Meador CB, Sanganahalli BG, Hyder F, de Stanchina E, et al. Afatinib plus cetuximab delays resistance compared to single-agent erlotinib or afatinib in mouse models of TKI-Naive EGFR L858R-Induced Lung Adenocarcinoma. Clin Cancer Res. 2016;22:426–35.
Ji HB, Li DN, Chen L, Shimamura T, Kobayashi S, McNamara K, et al. The impact of human kinase domain mutations on lung tumorigenesis and in vivo sensitivity to EGFR-targeted therapies. Cancer Cell. 2006;9:485–95.
Charoentong P, Finotello F, Angelova M, Mayer C, Efremova M, Rieder D, et al. Pan-cancer immunogenomic analyses reveal genotype-immunophenotype relationships and predictors of response to checkpoint blockade. Cell Rep. 2017;18:248–62.
Maemondo M, Inoue A, Kobayashi K, Sugawara S, Oizumi S, Isobe H, et al. Gefitinib or chemotherapy for non–small-cell lung cancer with mutated EGFR. N Engl J Med. 2010;362:2380–8.
Tang Q, Li L, Wang Y, Wu P, Hou X, Ouyang J, et al. RNA modifications in cancer. Br J Cancer. 2023;129:204–1.
He PC, He C. m6A RNA methylation: from mechanisms to therapeutic potential. EMBO J. 2021;40:e105977.
Sun WJ, Li JH, Liu S, Wu J, Zhou H, Qu LH, et al. RMBase: a resource for decoding the landscape of RNA modifications from high-throughput sequencing data. Nucleic Acids Res. 2016;44:D259–65.
Byrne AT, Alférez DG, Amant F, Annibali D, Arribas J, Biankin AV, et al. Interrogating open issues in cancer precision medicine with patient-derived xenografts. Nat Rev Cancer. 2017;17:254–68.
Kirchhammer N, Trefny MP, Maur PAD, Läubli H, Zippelius A. Combination cancer immunotherapies: Emerging treatment strategies adapted to the tumor microenvironment. Sci Transl Med. 2022;14:eabo3605.
Chatterjee N, Bivona TG. Polytherapy and targeted cancer drug resistance. Trends Cancer. 2019;5:170–82.
Ayeni D, Miller B, Kuhlmann A, Ho PC, Robles-Oteiza C, Gaefele M, et al. Tumor regression mediated by oncogene withdrawal or erlotinib stimulates infiltration of inflammatory immune cells in EGFR mutant lung tumors. J Immunother Cancer. 2019;7:172.
Chen X, Pan XH, Zhang WX, Guo HJ, Cheng SY, He QJ, et al. Epigenetic strategies synergize with PD-L1/PD-1 targeted cancer immunotherapies to enhance antitumor responses. Acta Pharmaceutica Sin B. 2020;10:723–33.
Funding
This work was supported by grants from the National Natural Science Foundation of China (No. 82073121) and Guangdong Basic and Applied Basic Research Foundation (No. 2020A1515010041).
Author information
Authors and Affiliations
Contributions
KW and HL conceived and designed the experiments. WZ, JW, JL and ZH performed the experiments. WZ and JW analyzed the data. HL and KW wrote the paper. All authors approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Consent for publication
All authors have reviewed the final version of the manuscript and are in agreement its content and submission.
Ethics approval and consent to participate
The study was conducted following the principles outlined in the Declaration of Helsinki and was approved by the Ethics Committees of Sun Yat-Sen Memorial Hospital (SYSEC-KY-KS-2019-189). The animal procedures were approved by the Institutional Animal Care and Use Committee of Sun Yat-sen University (AP20220148). All methods were carried out in accordance with relevant guidelines and regulations.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, W., Wang, J., Liang, J. et al. RNA methylation of CD47 mediates tumor immunosuppression in EGFR-TKI resistant NSCLC. Br J Cancer 132, 569–579 (2025). https://doi.org/10.1038/s41416-025-02945-2
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41416-025-02945-2
This article is cited by
-
Pathobiology and clinical significance of malignant pleural effusions
EMBO Molecular Medicine (2026)