Abstract
Background
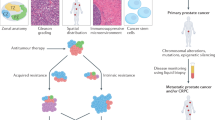
Metastatic, castration-resistant prostate cancer (mCRPC) directly contributes to the mortality and morbidity of prostate cancer. It is imperative to identify new molecular targets and discover effective therapeutic agents against lethal mCRPC.
Methods
The anticancer activities and mechanism of action of the small-molecule lead compound were investigated in preclinical models of human prostate cancer. Immunohistochemistry was employed to determine the expression of S-phase kinase-associated protein 1 (Skp1) in human prostate tissues.
Results
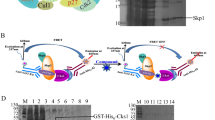
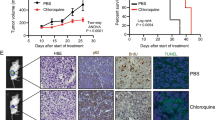
GH501 demonstrates nanomolar potency in the NCI-60 human cancer cell panel and multiple mCRPC cell lines with diverse genetic backgrounds, including those resistant to androgen deprivation therapy drugs. Mechanistically, GH501 may bind Skp1 and disrupt the physical interaction between Skp1 and S-phase kinase-associated protein 2 (Skp2) within the Skp1-Cullin1-F-box protein ubiquitin ligase complexes (SCF), thereby affecting multiple oncogenic signals implicated in mCRPC progression, including p21, p27, β-catenin, cyclin D1, enhancer of zeste homolog 2 (EZH2), c-Myc, and survivin. GH501 exhibits excellent in vitro and in vivo safety pharmacology, and GH501 monotherapy effectively inhibits the in vivo growth of cell- and patient-derived xenografts in intraosseous and subcutaneous models. Skp1 expression is significantly increased in human prostate cancer specimens.
Conclusion
These results indicate that interrupting Skp1-Skp2 interaction is an effective approach to target mCRPC and warrant further preclinical development of GH501 as a promising therapeutic candidate.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 24 print issues and online access
$259.00 per year
only $10.79 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
RNA sequencing data are deposited in the Gene Expression Omnibus database under the accession number GSE244150. Other relevant data supporting the findings of this study are included in this article and its Supplementary Information.
References
Siegel RL, Kratzer TB, Giaquinto AN, Sung H, Jemal A. Cancer statistics, 2025. CA Cancer J Clin. 2025;75:10–45.
Posdzich P, Darr C, Hilser T, Wahl M, Herrmann K, Hadaschik B, et al. Metastatic prostate cancer-a review of current treatment options and promising new approaches. Cancers. 2023;15:461.
Kwan EM, Wyatt AW. Androgen receptor genomic alterations and treatment resistance in metastatic prostate cancer. Prostate. 2022;82:S25–S36.
Xu K, Wu ZJ, Groner AC, He HH, Cai C, Lis RT, et al. EZH2 oncogenic activity in castration-resistant prostate cancer cells is Polycomb-independent. Science. 2012;338:1465–9.
Li X, Gera L, Zhang S, Chen Y, Lou L, Wilson LM, et al. Pharmacological inhibition of noncanonical EED-EZH2 signaling overcomes chemoresistance in prostate cancer. Theranostics. 2021;11:6873–90.
Wang J, Ben-David R, Mehrazin R, Yang W, Tewari AK, Kyprianou N. Novel signatures of prostate cancer progression and therapeutic resistance. Expert Opin Ther Targets. 2023;27:1195–206.
Thompson LL, Rutherford KA, Lepage CC, McManus KJ. The SCF complex is essential to maintain genome and chromosome stability. Int J Mol Sci. 2021;22:8544.
Nguyen KM, Busino L. The Biology of F-box proteins: the SCF family of E3 ubiquitin ligases. Adv Exp Med Biol. 2020;1217:111–22.
Liu J, Peng Y, Zhang J, Long J, Liu J, Wei W. Targeting SCF E3 ligases for cancer therapies. Adv Exp Med Biol. 2020;1217:123–46.
Yang G, Ayala G, De Marzo A, Tian W, Frolov A, Wheeler TM, et al. Elevated Skp2 protein expression in human prostate cancer: association with loss of the cyclin-dependent kinase inhibitor p27 and PTEN and with reduced recurrence-free survival. Clin Cancer Res. 2002;8:3419–26.
Zhao H, Bauzon F, Fu H, Lu Z, Cui J, Nakayama K, et al. Skp2 deletion unmasks a p27 safeguard that blocks tumorigenesis in the absence of pRb and p53 tumor suppressors. Cancer Cell. 2013;24:645–59.
Lin HK, Chen Z, Wang G, Nardella C, Lee SW, Chan CH, et al. Skp2 targeting suppresses tumorigenesis by Arf-p53-independent cellular senescence. Nature. 2010;464:374–9.
Lu W, Liu S, Li B, Xie Y, Izban MG, Ballard BR, et al. SKP2 loss destabilizes EZH2 by promoting TRAF6-mediated ubiquitination to suppress prostate cancer. Oncogene. 2017;36:1364–73.
Wang J, Han F, Lee SW, Wu J, Chan CH, Zhang X, et al. E3-ligase Skp2 regulates beta-catenin expression and maintains hematopoietic stem cell homing. Biochem Biophys Res Commun. 2014;445:566–71.
Wang Z, Zhao T, Zhang S, Wang J, Chen Y, Zhao H, et al. The Wnt signaling pathway in tumorigenesis, pharmacological targets, and drug development for cancer therapy. Biomark Res. 2021;9:68.
Tekcham DS, Chen D, Liu Y, Ling T, Zhang Y, Chen H, et al. F-box proteins and cancer: an update from functional and regulatory mechanism to therapeutic clinical prospects. Theranostics. 2020;10:4150–67.
Viegas-Junior C, Danuello A, da Silva Bolzani V, Barreiro EJ, Fraga CA. Molecular hybridization: a useful tool in the design of new drug prototypes. Curr Med Chem. 2007;14:1829–52.
Stewart JM, Gera L, Chan DC, York EJ, Simkeviciene V, Bunn PA Jr., et al. Combination cancer chemotherapy with one compound: pluripotent bradykinin antagonists. Peptides. 2005;26:1288–91.
Gera L, Stewart J, Chung LW, Wu D. Compositions and methods for treating bone cancer. 2010; US Patent 2010;20100144678.
Gera L, Chan D, Hodges R, Bunn P. Flurbiprofen analogs and methods of use in treating cancer. US Patent 2013;8,575,170 B2.
Seo SI, Gera L, Zhau HE, Qian WP, Iqbal S, Johnson NA, et al. BKM1740, an acyl-tyrosine bisphosphonate amide derivative, inhibits the bone metastatic growth of human prostate cancer cells by inducing apoptosis. Clin Cancer Res. 2008;14:6198–206.
Zhang S, Gera L, Mamouni K, Li X, Chen Z, Kucuk O, et al. Inhibition of skeletal growth of human prostate cancer by the combination of docetaxel and BKM1644: an aminobisphosphonate derivative. Oncotarget. 2016;7:27489–98.
Chen Y, Gera L, Zhang S, Li X, Yang Y, Mamouni K, et al. Small molecule BKM1972 inhibits human prostate cancer growth and overcomes docetaxel resistance in intraosseous models. Cancer Lett. 2019;446:62–72.
Chan CH, Morrow JK, Li CF, Gao Y, Jin G, Moten A, et al. Pharmacological inactivation of Skp2 SCF ubiquitin ligase restricts cancer stem cell traits and cancer progression. Cell. 2013;154:556–68.
Liu YQ, Wang XL, Cheng X, Lu YZ, Wang GZ, Li XC, et al. Skp1 in lung cancer: clinical significance and therapeutic efficacy of its small molecule inhibitors. Oncotarget. 2015;6:34953–67.
Ellis WJ, Vessella RL, Buhler KR, Bladou F, True LD, Bigler SA, et al. Characterization of a novel androgen-sensitive, prostate-specific antigen-producing prostatic carcinoma xenograft: LuCaP 23. Clin Cancer Res. 1996;2:1039–48.
Ramos-Inza S, Ruberte AC, Sanmartin C, Sharma AK, Plano D. NSAIDs: old acquaintance in the pipeline for cancer treatment and prevention horizontal line structural modulation, mechanisms of action, and bright future. J Med Chem. 2021;64:16380–421.
Wittich S, Scherf H, Xie C, Brosch G, Loidl P, Gerhauser C, et al. Structure-activity relationships on phenylalanine-containing inhibitors of histone deacetylase: in vitro enzyme inhibition, induction of differentiation, and inhibition of proliferation in Friend leukemic cells. J Med Chem. 2002;45:3296–309.
Avdieiev S, Gera L, Havrylyuk D, Hodges RS, Lesyk R, Ribrag V, et al. Bradykinin antagonists and thiazolidinone derivatives as new potential anti-cancer compounds. Bioorg Med Chem. 2014;22:3815–23.
Thalmann GN, Anezinis PE, Chang SM, Zhau HE, Kim EE, Hopwood VL, et al. Androgen-independent cancer progression and bone metastasis in the LNCaP model of human prostate cancer. Cancer Res. 1994;54:2577–81.
Liu C, Armstrong CM, Lou W, Lombard AP, Cucchiara V, Gu X, et al. Niclosamide and bicalutamide combination treatment overcomes enzalutamide- and bicalutamide-resistant prostate cancer. Mol Cancer Ther. 2017;16:1521–30.
Zhu Y, Liu C, Nadiminty N, Lou W, Tummala R, Evans CP, et al. Inhibition of ABCB1 expression overcomes acquired docetaxel resistance in prostate cancer. Mol Cancer Ther. 2013;12:1829–36.
Liu C, Armstrong C, Zhu Y, Lou W, Gao AC. Niclosamide enhances abiraterone treatment via inhibition of androgen receptor variants in castration resistant prostate cancer. Oncotarget. 2016;7:32210–20.
Wu TT, Sikes RA, Cui Q, Thalmann GN, Kao C, Murphy CF, et al. Establishing human prostate cancer cell xenografts in bone: induction of osteoblastic reaction by prostate-specific antigen-producing tumors in athymic and SCID/bg mice using LNCaP and lineage-derived metastatic sublines. Int J Cancer. 1998;77:887–94.
Holleran JL, Miller CJ, Culp LA. Tracking micrometastasis to multiple organs with lacZ-tagged CWR22R prostate carcinoma cells. J Histochem Cytochem. 2000;48:643–51.
Martinez Molina D, Jafari R, Ignatushchenko M, Seki T, Larsson EA, Dan C, et al. Monitoring drug target engagement in cells and tissues using the cellular thermal shift assay. Science. 2013;341:84–7.
Tiffen JC, Gunatilake D, Gallagher SJ, Gowrishankar K, Heinemann A, Cullinane C, et al. Targeting activating mutations of EZH2 leads to potent cell growth inhibition in human melanoma by derepression of tumor suppressor genes. Oncotarget. 2015;6:27023–36.
Chen H, Tu SW, Hsieh JT. Down-regulation of human DAB2IP gene expression mediated by polycomb Ezh2 complex and histone deacetylase in prostate cancer. J Biol Chem. 2005;280:22437–44.
Zheng Z, Li J, Liu Y, Shi Z, Xuan Z, Yang K, et al. The crucial role of AR-V7 in enzalutamide-resistance of castration-resistant prostate cancer. Cancers (Basel). 2022;14:4877.
Sudsakorn S, Bahadduri P, Fretland J, Lu C. 2020 FDA drug-drug interaction guidance: a comparison analysis and action plan by pharmaceutical industrial scientists. Curr Drug Metab. 2020;21:403–26.
Lester RM, Paglialunga S, Johnson IA. QT assessment in early drug development: the long and the short of it. Int J Mol Sci. 2019;20:1324.
Haffner MC, Zwart W, Roudier MP, True LD, Nelson WG, Epstein JI, et al. Genomic and phenotypic heterogeneity in prostate cancer. Nat Rev Urol. 2021;18:79–92.
Kirk PS, Koreckij T, Nguyen HM, Brown LG, Snyder LA, Vessella RL, et al. Inhibition of CCL2 signaling in combination with docetaxel treatment has profound inhibitory effects on prostate cancer growth in bone. Int J Mol Sci. 2013;14:10483–96.
Bai C, Sen P, Hofmann K, Ma L, Goebl M, Harper JW, et al. SKP1 connects cell cycle regulators to the ubiquitin proteolysis machinery through a novel motif, the F-box. Cell. 1996;86:263–74.
Xing S, Nong F, Wang Y, Huang D, Qin J, Chen YF, et al. Brusatol has therapeutic efficacy in non-small cell lung cancer by targeting Skp1 to inhibit cancer growth and metastasis. Pharm Res. 2022;176:106059.
Hussain M, Lu Y, Tariq M, Jiang H, Shu Y, Luo S, et al. A small-molecule Skp1 inhibitor elicits cell death by p53-dependent mechanism. iScience. 2022;25:104591.
Hwang N, Pei Y, Clement J, Robertson ES, Du Y. Identification of a 3-beta-homoalanine conjugate of brusatol with reduced toxicity in mice. Bioorg Med Chem Lett. 2020;30:127553.
Ikediobi ON, Davies H, Bignell G, Edkins S, Stevens C, O’Meara S, et al. Mutation analysis of 24 known cancer genes in the NCI-60 cell line set. Mol Cancer Ther. 2006;5:2606–12.
Hooshfar S, Linzey MR, Wu D, Gera L, Mamouni K, Li X, et al. Sensitive liquid chromatography/tandem mass spectrometry method for the determination of two novel highly lipophilic anticancer drug candidates in rat plasma and tissues. Biomed Chromatogr. 2018;32:10.1002/bmc.4064.
Acknowledgements
The authors are grateful to the National Cancer Institute Developmental Therapeutics Program for the NCI-60 screening. We thank Emory University Winship Cancer Institute Cancer Tissue and Pathology core for histopathological studies, and Dr. Rhea-Beth Markowitz at Georgia Cancer Center, Augusta University, for editorial assistance.
Funding
This work was supported by National Cancer Institute grants R42CA217491 and R01CA256058 (DW), R01CA266452, R21CA277368, and P50CA097186 (PSN and IMC), R50CA274336 (IMC), National Institute on Minority Health and Health Disparities grant U54MD007590 (Project 1), Department of Education Title III Program at Clark Atlanta University, Georgia Research Alliance VentureLab grant, Emory University Winship Cancer Institute-Roswell Country Club Prostate Cancer Research Award, and Augusta University-Clark Atlanta University Prostate Cancer Pilot grant (DW), the John D. Stobo, M.D. Distinguished Chair Endowment, and the Edith & Robert Zinn Chair Endowment in Drug Discovery (JZ).
Author information
Authors and Affiliations
Contributions
XL: Conducted investigation, performed data curation and formal analysis, and contributed to manuscript drafting and revision. KM: Conducted investigation, performed data curation and formal analysis, and contributed to manuscript drafting. RZ: Conducted investigation, performed data curation and formal analysis. LB: Conducted investigation, performed data curation and formal analysis. YC: Conducted investigation, performed data curation and formal analysis. YW: Conducted investigation, performed data curation and formal analysis, and contributed to manuscript drafting and figure preparation. ZRX: Contributed to conceptualization, supervision, project administration, methodology, investigation, formal analysis, and manuscript editing. GAS: Contributed to conceptualization, supervision, project administration, methodology, data curation and formal analysis, and manuscript drafting and revision. DLiu: Contributed to conceptualization, methodology, investigation, formal analysis, and manuscript drafting. NJB: Contributed to conceptualization, methodology, investigation, formal analysis, and manuscript editing. AD: Conducted investigation, assisted in data curation, formal analysis, and manuscript editing. DLi: Conducted investigation, assisted in data curation and formal analysis. NC: Conducted investigation, assisted in data curation and formal analysis. SG: Conducted investigation, assisted in data curation and formal analysis. JZhu: Conducted investigation, assisted in methodology and data analysis. IMC: Contributed to conceptualization, supervision, project administration, funding acquisition, data curation and formal analysis, manuscript drafting and revision. PSN: Contributed to conceptualization, supervision, project administration, funding acquisition, and manuscript editing. QB: Conducted investigation, assisted in data curation and formal analysis. JZhou: Contributed to conceptualization, supervision, project administration, funding acquisition, and manuscript preparation. AOO: Conducted investigation, data curation and formal analysis, and contributed to manuscript preparation. OK: Reviewed and provided critical feedback on the manuscript. LG: Contributed to conceptualization, investigation, supervision, project administration, data curation and formal analysis, manuscript drafting and revision. DW: Conceptualized the study, supervised the project, conducted investigation, data curation and formal analysis, provided funding support, drafted and finalized the manuscript.
Corresponding authors
Ethics declarations
Competing interests
Lajos Gera and Daqing Wu have intellectual property interests in GH501 and related compounds. Daqing Wu has ownership interests in MetCure Therapeutics LLC. Lajos Gera and Omer Kucuk provided consultation services to MetCure Therapeutics LLC. No potential conflicts of interest were disclosed by the other authors.
Ethics approval and consent to participate
The Institutional Animal Care and Use Committee (IACUC) at Augusta University approved the animal protocol (AUP# 2014-0626) used in this study. All animal procedures were subjected to National Institutes of Health guidelines.
Consent for publication
Not applicable.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, X., Mamouni, K., Zhao, R. et al. Novel Skp1 inhibitor has potent preclinical efficacy against castration-resistant prostate cancer. Br J Cancer 132, 1188–1199 (2025). https://doi.org/10.1038/s41416-025-02993-8
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41416-025-02993-8