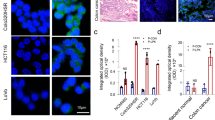
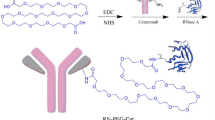
Abstract
Background
Antibody-drug conjugates (ADCs) are a significant advancement in targeted cancer therapy, but none are approved for colorectal cancer (CRC). LGR4/5/6, highly expressed in most CRCs, are promising targets. While LGR5-targeting ADCs show strong anti-tumor effects, their efficacy is limited by LGR5 loss in some CRC cells. RSPO4, a natural ligand for LGR4/5/6, binds all three receptors with high affinity. This research develops RSPO4-based peptibody drug-conjugates (PDCs) to simultaneously target LGR4/5/6, offering a novel therapeutic approach for CRC.
Methods
LGR4/5/6 expression in CRCs was analysed using RNA-seq datasets and Western blot. Peptibody binding affinities were measured, conjugated to camptothecin analog, CPT2, and tested for cytotoxicity in CRC cell lines. Antitumor efficacy was evaluated in vivo using CRC cell line and patient-derived xenograft (PDX) models.
Results
Peptibody was engineered by fusing a mutant RSPO4 furin-domain to human IgG1 Fc, retaining high-affinity LGR4/5/6 binding without enhancing Wnt/β-catenin signalling. Conjugated with CPT2 molecules, the PDC showed strong antitumor activity in CRC cell lines and dose-dependent tumor growth inhibition in xenograft and patient-derived models.
Conclusion
Preclinical data showed that LGR4/5/6-targeting PDC exhibited potent cytotoxicity in vitro and robust antitumor efficacy in CRC xenograft and PDX models, making its potential as a promising therapeutic approach for CRC.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 24 print issues and online access
$259.00 per year
only $10.79 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
All the relevant data generated in this manuscript that support the findings of this study are available upon request from the authors.
References
Dekker E, Tanis PJ, Vleugels JLA, Kasi PM, Wallace MB. Colorectal cancer. Lancet. 2019;394:1467–80. https://doi.org/10.1016/S0140-6736(19)32319-0.
Morris VK, Kennedy EB, Baxter NN, Benson AB 3rd, Cercek A, Cho M, et al. Treatment of metastatic colorectal cancer: ASCO guideline. J Clin Oncol. 2023;41:678–700. https://doi.org/10.1200/JCO.22.01690.
Vogelstein B, Kinzler KW. Cancer genes and the pathways they control. Nat Med. 2004;10:789–99. https://doi.org/10.1038/nm1087nm1087.
de Lau W, Barker N, Clevers H. WNT signaling in the normal intestine and colorectal cancer. Front Biosci. 2007;12:471–91.
Carmon KS, Gong X, Lin Q, Thomas A, Liu Q. R-spondins function as ligands of the orphan receptors LGR4 and LGR5 to regulate Wnt/beta-catenin signaling. Proc Natl Acad Sci USA. 2011;108:11452–7. https://doi.org/10.1073/pnas.1106083108.
de Lau W, Barker N, Low TY, Koo BK, Li VS, Teunissen H, et al. Lgr5 homologues associate with Wnt receptors and mediate R-spondin signalling. Nature. 2011;476:293–7. https://doi.org/10.1038/nature10337.
Seshagiri S, Stawiski EW, Durinck S, Modrusan Z, Storm EE, Conboy CB, et al. Recurrent R-spondin fusions in colon cancer. Nature. 2012;488:660–4. https://doi.org/10.1038/nature11282.
de Lau WB, Snel B, Clevers HC. The R-spondin protein family. Genome Biol. 2012;13:242 https://doi.org/10.1186/gb-2012-13-3-242.
Nam JS, Park E, Turcotte TJ, Palencia S, Zhan X, Lee J, et al. Mouse R-spondin2 is required for apical ectodermal ridge maintenance in the hindlimb. Dev Biol. 2007;311:124–35. https://doi.org/10.1016/j.ydbio.2007.08.023.
Wei Q, Yokota C, Semenov MV, Doble B, Woodgett J, He X. R-spondin1 is a high affinity ligand for LRP6 and induces LRP6 phosphorylation and beta-catenin signaling. J Biol Chem. 2007;282:15903–11. https://doi.org/10.1074/jbc.M701927200.
Park S, Cui J, Yu W, Wu L, Carmon KS, Liu QJ. Differential activities and mechanisms of the four R-spondins in potentiating Wnt/beta-catenin signaling. J Biol Chem. 2018;293:9759–69. https://doi.org/10.1074/jbc.RA118.002743.
Dubey R, van Kerkhof P, Jordens I, Malinauskas T, Pusapati GV, McKenna JK, et al. R-spondins engage heparan sulfate proteoglycans to potentiate WNT signaling. eLife. 2020; 9. https://doi.org/10.7554/eLife.54469.
Chen PH, Chen X, Lin Z, Fang D, He X. The structural basis of R-spondin recognition by LGR5 and RNF43. Genes Dev. 2013;27:1345–50. https://doi.org/10.1101/gad.219915.113.
Zebisch M, Jones EY. Crystal structure of R-spondin 2 in complex with the ectodomains of its receptors LGR5 and ZNRF3. J Struct Biol. 2015;191:149–55. https://doi.org/10.1016/j.jsb.2015.05.008.
Peng WC, de Lau W, Forneris F, Granneman JC, Huch M, Clevers H, et al. Structure of stem cell growth factor R-spondin 1 in complex with the ectodomain of its receptor LGR5. Cell Rep. 2013;3:1885–92. https://doi.org/10.1016/j.celrep.2013.06.009.
Xu K, Xu Y, Rajashankar KR, Robev D, Nikolov DB. Crystal structures of Lgr4 and its complex with R-spondin1. Structure. 2013;21:1683–9. https://doi.org/10.1016/j.str.2013.07.001.
Hao HX, Xie Y, Zhang Y, Charlat O, Oster E, Avello M, et al. ZNRF3 promotes Wnt receptor turnover in an R-spondin-sensitive manner. Nature. 2012;485:195–200. https://doi.org/10.1038/nature11019.
Koo BK, Spit M, Jordens I, Low TY, Stange DE, van de Wetering M, et al. Tumour suppressor RNF43 is a stem-cell E3 ligase that induces endocytosis of Wnt receptors. Nature. 2012;488:665–9. https://doi.org/10.1038/nature11308.
Wang L, Hu F, Cui Q, Qiao H, Li L, Geng T, et al. Structural insights into the LGR4-RSPO2-ZNRF3 complexes regulating WNT/beta-catenin signaling. Nat Commun. 2025;16:362 https://doi.org/10.1038/s41467-024-55431-3.
Park S, Wu L, Tu J, Yu W, Toh Y, Carmon KS, et al. Unlike LGR4, LGR5 potentiates Wnt-beta-catenin signaling without sequestering E3 ligases. Sci Signal. 2020;13. https://doi.org/10.1126/scisignal.aaz4051.
Toh Y, Wu L, Park S, Wang A, Tu J, Yu W, et al. LGR4 and LGR5 form distinct homodimers that only LGR4 complexes with RNF43/ZNRF3 to provide high affinity binding of R-spondin ligands. Sci Rep. 2023;13:10796 https://doi.org/10.1038/s41598-023-37856-w.
Gao Y, Kitagawa K, Hiramatsu Y, Kikuchi H, Isobe T, Shimada M, et al. Up-regulation of GPR48 induced by down-regulation of p27Kip1 enhances carcinoma cell invasiveness and metastasis. Cancer Res. 2006;66:11623–31.
Yi J, Xiong W, Gong X, Bellister S, Ellis LM, Liu Q. Analysis of LGR4 receptor distribution in human and mouse tissues. PLoS ONE. 2013;8:e78144 https://doi.org/10.1371/journal.pone.0078144.
Gugger M, White R, Song S, Waser B, Cescato R, Riviere P, et al. GPR87 is an overexpressed G-protein coupled receptor in squamous cell carcinoma of the lung. Dis Markers. 2008;24:41–50.
Gong X, Yi J, Carmon KS, Crumbley CA, Xiong W, Thomas A, et al. Aberrant RSPO3-LGR4 signaling in Keap1-deficient lung adenocarcinomas promotes tumor aggressiveness. Oncogene. 2015;34:4692–701. https://doi.org/10.1038/onc.2014.417.
Yue Z, Yuan Z, Zeng L, Wang Y, Lai L, Li J, et al. LGR4 modulates breast cancer initiation, metastasis, and cancer stem cells. FASEB J. 2018;32:2422–37. https://doi.org/10.1096/fj.201700897R.
Junttila MR, Mao W, Wang X, Wang BE, Pham T, Flygare J, et al. Targeting LGR5+ cells with an antibody-drug conjugate for the treatment of colon cancer. Sci Transl Med. 2015;7:314ra186 https://doi.org/10.1126/scitranslmed.aac7433.
Vermeulen L, Todaro M, de Sousa Mello F, Sprick MR, Kemper K, Perez Alea M, et al. Single-cell cloning of colon cancer stem cells reveals a multi-lineage differentiation capacity. Proc Natl Acad Sci USA. 2008;105:13427–32. https://doi.org/10.1073/pnas.0805706105.
Kobayashi S, Yamada-Okabe H, Suzuki M, Natori O, Kato A, Matsubara K, et al. LGR5-positive colon cancer stem cells interconvert with drug-resistant LGR5-negative cells and are capable of tumor reconstitution. Stem Cells. 2012;30:2631–44. https://doi.org/10.1002/stem.1257.
Kemper K, Prasetyanti PR, De Lau W, Rodermond H, Clevers H, Medema JP. Monoclonal antibodies against Lgr5 identify human colorectal cancer stem cells. Stem Cells. 2012;30:2378–86. https://doi.org/10.1002/stem.1233.
Huang PY, Kandyba E, Jabouille A, Sjolund J, Kumar A, Halliwill K, et al. Lgr6 is a stem cell marker in mouse skin squamous cell carcinoma. Nat Genet. 2017;49:1624–32. https://doi.org/10.1038/ng.3957.
Shimokawa M, Ohta Y, Nishikori S, Matano M, Takano A, Fujii M, et al. Visualization and targeting of LGR5(+) human colon cancer stem cells. Nature. 2017;545:187–92. https://doi.org/10.1038/nature22081.
de Sousa e Melo F, Kurtova AV, Harnoss JM, Kljavin N, Hoeck JD, Hung J, et al. A distinct role for Lgr5(+) stem cells in primary and metastatic colon cancer. Nature. 2017;543:676–80. https://doi.org/10.1038/nature21713.
Kim SK, Kim SY, Kim JH, Roh SA, Cho DH, Kim YS, et al. A nineteen gene-based risk score classifier predicts prognosis of colorectal cancer patients. Mol Oncol. 2014;8:1653–66. https://doi.org/10.1016/j.molonc.2014.06.016.
Leto SM, Grassi E, Avolio M, Vurchio V, Cottino F, Ferri M, et al. XENTURION is a population-level multidimensional resource of xenografts and tumoroids from metastatic colorectal cancer patients. Nat Commun. 2024:15:7495–506. https://doi.org/10.1038/s41467-024-51909-2.
Carter PJ, Senter PD. Antibody-drug conjugates for cancer therapy. Cancer J. 2008;14:154–69. https://doi.org/10.1097/PPO.0b013e318172d704.
Dumontet C, Reichert JM, Senter PD, Lambert JM, Beck A. Antibody-drug conjugates come of age in oncology. Nat Rev Drug Discov. 2023;22:641–61. https://doi.org/10.1038/s41573-023-00709-2. e-pub ahead of print 20230612
Cui J, Toh Y, Park S, Yu W, Tu J, Wu L, et al. Drug conjugates of antagonistic R-spondin 4 mutant for simultaneous targeting of leucine-rich repeat-containing G protein-coupled receptors 4/5/6 for cancer treatment. J Med Chem. 2021;64:12572–81. https://doi.org/10.1021/acs.jmedchem.1c00395.
Tolcher AW. Antibody drug conjugates: lessons from 20 years of clinical experience. Annals of oncology: official journal of the European Society for Medical Oncology/ESMO. Ann Oncol. 2016;27:2168–72. https://doi.org/10.1093/annonc/mdw424.
Bailly C. Irinotecan: 25 years of cancer treatment. Pharmacol Res. 2019;148:104398. https://doi.org/10.1016/j.phrs.2019.104398.
Lyski RD, Bou LB, Lau UY, Meyer DW, Cochran JH, Okeley NM, et al. Development of novel antibody-camptothecin conjugates. Mol Cancer Ther. 2021;20:329–39. https://doi.org/10.1158/1535-7163.MCT-20-0526.
Dennler P, Chiotellis A, Fischer E, Bregeon D, Belmant C, Gauthier L, et al. Transglutaminase-based chemo-enzymatic conjugation approach yields homogeneous antibody-drug conjugates. Bioconjug Chem. 2014;25:569–78. https://doi.org/10.1021/bc400574z.
Lhospice F, Bregeon D, Belmant C, Dennler P, Chiotellis A, Fischer E, et al. Site-specific conjugation of monomethyl auristatin E to Anti-CD30 antibodies improves their pharmacokinetics and therapeutic index in rodent models. Mol Pharm. 2015;12:1863–71. https://doi.org/10.1021/mp500666j.
Gong X, Carmon KS, Lin Q, Thomas A, Yi J, Liu Q. LGR6 is a high affinity receptor of R-spondins and potentially functions as a tumor suppressor. PLoS ONE. 2012;7:e37137 https://doi.org/10.1371/journal.pone.0037137.
Jacob J, Francisco LE, Chatterjee T, Liang Z, Subramanian S, Liu QJ, et al. An antibody-drug conjugate targeting GPR56 demonstrates efficacy in preclinical models of colorectal cancer. Br J Cancer. 2023;128:1592–602. https://doi.org/10.1038/s41416-023-02192-3.
Strop P, Liu SH, Dorywalska M, Delaria K, Dushin RG, Tran TT, et al. Location matters: site of conjugation modulates stability and pharmacokinetics of antibody drug conjugates. Chem Biol. 2013;20:161–7. https://doi.org/10.1016/j.chembiol.2013.01.010.
Ogitani Y, Aida T, Hagihara K, Yamaguchi J, Ishii C, Harada N, et al. DS-8201a, a novel HER2-targeting ADC with a novel DNA topoisomerase I inhibitor, demonstrates a promising antitumor efficacy with differentiation from T-DM1. Clin Cancer Res. 2016;22:5097–108. https://doi.org/10.1158/1078-0432.CCR-15-2822.
Peng WC, de Lau W, Madoori PK, Forneris F, Granneman JC, Clevers H, et al. Structures of Wnt-antagonist ZNRF3 and its complex with R-spondin 1 and implications for signaling. PLoS One. 2013;8:e83110 https://doi.org/10.1371/journal.pone.0083110.
Anami Y, Xiong W, Gui X, Deng M, Zhang CC, Zhang N, et al. Enzymatic conjugation using branched linkers for constructing homogeneous antibody-drug conjugates with high potency. Org Biomol Chem. 2017;15:5635–42. https://doi.org/10.1039/c7ob01027c.
Saber H, Leighton JK. An FDA oncology analysis of antibody-drug conjugates. Regul Toxicol Pharmacol. 2015;71:444–52. https://doi.org/10.1016/j.yrtph.2015.01.014.
Gong X, Azhdarinia A, Ghosh SC, Xiong W, An Z, Liu Q, et al. LGR5-targeted antibody-drug conjugate eradicates gastrointestinal tumors and prevents recurrence. Mol Cancer Ther. 2016;15:1580–90. https://doi.org/10.1158/1535-7163.MCT-16-0114.
Fumagalli A, Oost KC, Kester L, Morgner J, Bornes L, Bruens L, et al. Plasticity of Lgr5-Negative Cancer Cells Drives Metastasis in Colorectal Cancer. Cell Stem Cell. 2020;26:569–578 e567. https://doi.org/10.1016/j.stem.2020.02.008.
Nessler I, Menezes B, Thurber GM. Key metrics to expanding the pipeline of successful antibody-drug conjugates. Trends Pharm Sci. 2021;42:803–12. https://doi.org/10.1016/j.tips.2021.07.005.
Tarcsa E, Guffroy MR, Falahatpisheh H, Phipps C, Kalvass JC. Antibody-drug conjugates as targeted therapies: Are we there yet? A critical review of the current clinical landscape. Drug Discov Today Technol. 2020;37:13–22. https://doi.org/10.1016/j.ddtec.2020.07.002.
Acknowledgements
The authors would like to thank Mr. Jack Adams of UTHealth-Houston for drawing the structure of PEG2-bis-PE3-azido-DBCO-PEG8-VKG-CPT2.
Funding
This work was supported in part by funding from the Cancer Prevention and Research Institute of Texas (CPRIT) RP220169 (to QJL and KSC), RP210119 (to QJL), National Cancer Institute (NCI) R01CA226894 and R21 CA282378 (to KSC), and the Janice David Gordon for Bowel Cancer Research Endowment (to QJL).
Author information
Authors and Affiliations
Contributions
YT, KSC and QJL devised the concept. YT, LW, JT, ZL, AMA, JJW, LL, SP, JHR, MEH, CLH, RAF, KSC and QJL designed, performed, and analyzed experiments. YT and QJL wrote the manuscript. All coauthors reviewed and approved of the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare the following competing financial interest(s): KSC and QJL and the Regents of the University of Texas have filed a patent application related to this project.
Ethics approval
All methods in this study were performed in accordance with the relevant guidelines and regulations. All animal experiments were approved by the Institutional Animal Care and Use Committee of the University of Texas at Houston (Protocol number AWC-22-0080, AWC-20-0144, and AWC-23-0106).
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
41416_2025_3121_MOESM1_ESM.pdf (download PDF )
Supplementary Figure S1. Coomassie blue staining image of purified R462 and R465 under reducing (left side) and non reduction condition (right side).
41416_2025_3121_MOESM2_ESM.pdf (download PDF )
Supplementary Figure S2. In vitro cytotoxicity of R462-CPT2 and R465-CPT2 in HEK293T cells and HEK293T cells expressing LGR4, LGR5, or LGR6. a
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Toh, Y., Wu, L., Tu, J. et al. Anti-tumor activity of camptothecin analog conjugate of an RSPO4-based peptibody targeting LGR4/5/6 in preclinical models of colorectal cancer. Br J Cancer 133, 1218–1228 (2025). https://doi.org/10.1038/s41416-025-03121-2
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41416-025-03121-2