Abstract
Background
Ewing sarcoma (ES) is a rare tumour with metastatic spread in ~25% of cases at diagnosis. Extrapulmonary disseminated disease defines very high-risk (VHR) patients, with frequent relapse and poor overall survival (OS, ~30%).
Methods
The phase II CombinaiR3 trial (NCT03011528) enroled 45 VHR ES patients across 15 French centres (2017–2021) to evaluate a strategy combining dose-dense induction chemotherapy, high-dose consolidation, and prolonged maintenance therapy. The primary endpoint was median event-free survival (EFS). Exploratory endpoints included full-body-fluorine-18-fluorodeoxyglucose-positron emission tomography/computed tomography (PET/CT) and circulating tumour DNA (ctDNA) detection during treatment.
Results
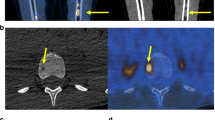
Among 42 analysed patients (median age 14 years, range 6–47), 29 had a primary tumour volume ≥200 ml, and 35 presented with bone ± bone marrow metastatic lesions, 18 exhibiting more than 5 bone lesions. At 48-month follow-up, 18- and 36-month EFS rates were 63.4% and 53.7%, respectively, with 3-year OS at 65.5%. Toxicity was as expected, with no treatment-related deaths or maintenance therapy discontinuations due to toxicity. PET/CT and ctDNA monitoring showed strong correlation at diagnosis and relapse.
Discussion
This study supports the proposed experimental strategy as a first-line option for selected VHR ES patients, warranting integration into international therapeutic discussions.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 24 print issues and online access
$259.00 per year
only $10.79 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The dataset generated and analysed during the current study is available from the corresponding author on reasonable request.
References
Kovar H, Dworzak M, Strehl S, Schnell E, Ambros IM, Ambros PF, et al. Overexpression of the pseudoautosomal gene MIC2 in Ewing’s sarcoma and peripheral primitive neuroectodermal tumor. Oncogene. 1990;5:1067–70.
Delattre O, Zucman J, Plougastel B, Desmaze C, Melot T, Peter M, et al. Gene fusion with an ETS DNA-binding domain caused by chromosome translocation in human tumours. Nature. 1992;359:162–5.
Delattre O, Zucman J, Melot T, Garau XS, Zucker JM, Lenoir GM, et al. The Ewing family of tumors–a subgroup of small-round-cell tumors defined by specific chimeric transcripts. N Engl J Med. 1994;331:294–9.
Zoubek A, Dockhorn-Dworniczak B, Delattre O, Christiansen H, Niggli F, Gatterer-Menz I, et al. Does expression of different EWS chimeric transcripts define clinically distinct risk groups of Ewing tumor patients? J Clin Oncol. 1996;14:1245–51.
Riggi N, Suvà ML, Stamenkovic I. Ewing’s Sarcoma. N Engl J Med. 2021;384:154–64.
Ladenstein R, Pötschger U, Le Deley MC, Whelan J, Paulussen M, Oberlin O, et al. Primary disseminated multifocal Ewing sarcoma: results of the Euro-EWING 99 trial. J Clin Oncol. 2010;28:3284–91.
Grünewald TGP, Cidre-Aranaz F, Surdez D, Tomazou EM, De Álava E, Kovar H, et al. Ewing sarcoma. Nat Rev Dis Prim. 2018;4:5.
Womer RB, West DC, Krailo MD, Dickman PS, Pawel BR, Grier HE, et al. Randomized controlled trial of interval-compressed chemotherapy for the treatment of localized Ewing sarcoma: a report from the Children’s Oncology Group. J Clin Oncol. 2012;30:4148–54.
Whelan J, Khan A, Sharma A, Rothermundt C, Dileo P, Michelagnoli M, et al. Interval compressed vincristine, doxorubicin, cyclophosphamide alternating with ifosfamide, etoposide in patients with advanced Ewing’s and other Small Round Cell Sarcomas. Clin Sarcoma Res. 2012;2:12.
Brennan B, Kirton L, Marec-Bérard P, Gaspar N, Laurence V, Martín-Broto J, et al. Comparison of two chemotherapy regimens in patients with newly diagnosed Ewing sarcoma (EE2012): an open-label, randomised, phase 3 trial. Lancet. 2022;400:1513–21.
Wagner LM, McAllister N, Goldsby RE, Rausen AR, McNall-Knapp RY, McCarville MB, et al. Temozolomide and intravenous irinotecan for treatment of advanced Ewing sarcoma. Pediatr Blood Cancer. 2007;48:132–9.
Casey DA, Wexler LH, Merchant MS, Chou AJ, Merola PR, Price AP, et al. Irinotecan and temozolomide for Ewing sarcoma: the Memorial Sloan-Kettering experience. Pediatr Blood Cancer. 2009;53:1029–34.
Koch R, Gelderblom H, Haveman L, Brichard B, Jürgens H, Cyprova S, et al. High-dose treosulfan and melphalan as consolidation therapy versus standard therapy for high-risk (Metastatic) Ewing sarcoma. J Clin Oncol. 2022;40:2307–20.
Hanahan D, Bergers G, Bergsland E. Less is more, regularly: metronomic dosing of cytotoxic drugs can target tumor angiogenesis in mice. J Clin Investig. 2000;105:1045–7.
Ferrara N, Kerbel RS. Angiogenesis as a therapeutic target. Nature. 2005;438:967–74.
Zapletalova D, André N, Deak L, Kyr M, Bajciova V, Mudry P, et al. Metronomic chemotherapy with the COMBAT regimen in advanced pediatric malignancies: a multicenter experience. Oncology. 2012;82:249–60.
Pasquier E, Kavallaris M, André N. Metronomic chemotherapy: new rationale for new directions. Nat Rev Clin Oncol. 2010;7:455–65.
Casanova M, Ferrari A, Spreafico F, Terenziani M, Massimino M, Luksch R, et al. Vinorelbine in previously treated advanced childhood sarcomas: evidence of activity in rhabdomyosarcoma. Cancer. 2002;94:3263–8.
Casanova M, Ferrari A, Bisogno G, Merks JHM, De Salvo GL, Meazza C, et al. Vinorelbine and low-dose cyclophosphamide in the treatment of pediatric sarcomas: pilot study for the upcoming European Rhabdomyosarcoma Protocol. Cancer. 2004;101:1664–71.
Bisogno G, Chisholm J, Hladun R, De Salvo GL, Guerin F, Casanova M, et al. Maintenance chemotherapy in patients with high-risk rhabdomyosarcoma: long-term survival analysis of the European paediatric soft tissue sarcoma Study Group RMS 2005 trial. J Clin Oncol. 2025;43:1856–62.
Krumbholz M, Hellberg J, Steif B, Bäuerle T, Gillmann C, Fritscher T, et al. Genomic EWSR1 fusion sequence as highly sensitive and dynamic plasma tumor marker in Ewing sarcoma. Clin Cancer Res. 2016;22:4356–65.
Krumbholz M, Eiblwieser J, Ranft A, Zierk J, Schmidkonz C, Stütz AM, et al. Quantification of translocation-specific ctDNA provides an integrating parameter for early assessment of treatment response and risk stratification in Ewing sarcoma. Clin Cancer Res. 2021;27:5922–30.
Shulman DS, Klega K, Imamovic-Tuco A, Clapp A, Nag A, Thorner AR, et al. Detection of circulating tumour DNA is associated with inferior outcomes in Ewing sarcoma and osteosarcoma: a report from the Children’s Oncology Group. Br J Cancer. 2018;119:615–21.
Gupta A, Riedel RF, Shah C, Borinstein SC, Isakoff MS, Chugh R, et al. Consensus recommendations in the management of Ewing sarcoma from the National Ewing Sarcoma Tumor Board. Cancer. 2023;129:3363–71.
Strauss SJ, Frezza AM, Abecassis N, Bajpai J, Bauer S, Biagini R, et al. Bone sarcomas: ESMO-EURACAN-GENTURIS-ERN PaedCan Clinical Practice Guideline for diagnosis, treatment and follow-up. Ann Oncol. 2021;32:1520–36.
Seddon BM, Cassoni AM, Galloway MJ, Rees JH, Whelan JS. Fatal radiation myelopathy after high-dose busulfan and melphalan chemotherapy and radiotherapy for Ewing’s sarcoma: a review of the literature and implications for practice. Clin Oncol. 2005;17:385–90.
Bölling T, Dirksen U, Ranft A, Ernst I, Jürgens H, Willich N. Radiation toxicity following busulfan/melphalan high-dose chemotherapy in the EURO-EWING-99-trial: review of GPOH data. Strahlenther Onkol. 2009;185(Suppl 2):21–2.
Carrie C, Le Deley M, Claude L, Alapetite C, Marandet J, Habrand J, et al. The radiosensitization effect and toxicity of busulfan containing chemotherapy before radiotherapy for Ewing’s sarcomas. SPRINGER HEIDELBERG TIERGARTENSTRASSE 17, D-69121 HEIDELBERG, GERMANY, 2009, pp 31–31.
Young H, Baum R, Cremerius U, Herholz K, Hoekstra O, Lammertsma AA, et al. Measurement of clinical and subclinical tumour response using [18F]-fluorodeoxyglucose and positron emission tomography: review and 1999 EORTC recommendations. European Organization for Research and Treatment of Cancer (EORTC) PET Study Group. Eur J Cancer. 1999;35:1773–82.
Kwak M, Jung S-H. Phase II clinical trials with time-to-event endpoints: optimal two-stage designs with one-sample log-rank test. Stat Med. 2014;33:2004–16.
Choderlos de Laclos X, Risbourg S, Brennan B, Bertucci F, Gaspar N, Gelderblom H, et al. Impact of age on safety of Busulfan-Melphalan followed by autologous hematopoietic stem-cell transplantation versus standard chemotherapy in the patients of the EURO-E.W.I.N.G. 99 and Ewing 2008 clinical trials. Eur J Cancer. 2024;208:114229.
Brahmi M, Gautier J, Dufresne A, Marec-Berard P, Cropet C, Vizoso S, et al. REGOMAIN: A randomized, placebo-controlled, double-blinded, multicenter, comparative phase II study of the efficacy of regorafenib as maintenance treatment in patients (pts) with high-grade bone sarcomas (HGBS) at diagnosis or relapse and without complete remission after standard treatment. JCO. 2022;40:TPS11585–TPS11585.
Schmidkonz C, Krumbholz M, Atzinger A, Cordes M, Goetz TI, Prante O, et al. Assessment of treatment responses in children and adolescents with Ewing sarcoma with metabolic tumor parameters derived from 18F-FDG-PET/CT and circulating tumor DNA. Eur J Nucl Med Mol Imaging. 2020;47:1564–75.
Guinot A, Tabone-Eglinger S, Isnardi V, Bahri H, Surdez D, Delattre O, et al. Staging of newly diagnosed Ewing sarcoma: results of bone marrow aspiration and biopsy versus (18)FDG-PET/CT imaging for bone marrow involvement. Eur J Cancer. 2023;179:56–64.
Yadgarov M, Berikashvili L, Rakova E, Likar Y. 18 F-FDG PET metabolic parameters for the prediction of histological response to induction chemotherapy in osteosarcoma and Ewing sarcoma: a systematic review and network meta-analysis. Clin Nucl Med. 2024;49:e640–9.
Acknowledgement
We thank Dr. Erika Vacchelli for her assistance in writing, editing, and critically reviewing the manuscript. We also acknowledge Lisa Belin for her methodologic help.
Funding
This work was supported by the French Society for Childhood Cancer (SFCE, Société Française des Cancers de l’Enfant) and the ‘Princesse Margot’ Association. The authors also thank Mr. Wisnia for his private donation. We would like to acknowledge La Ligue Contre le Cancer for supporting the project through the 2017 ‘Enfants, Adolescents et Cancer’ grant titled: Étude de l’ADN tumoral circulant, des cellules tumorales circulantes, de la biologie des tumeurs primaires et métastatiques dans les sarcomes d’Ewing à la rechute ou de mauvais pronostic (compilation des études ancillaires EuroEwing 2012, rEECur et CombinaiR3).
Author information
Authors and Affiliations
Contributions
Valérie Laurence: Conceptualisation, methodology, investigation, writing-original draft, writing-reviewing and editing, funding acquisition. Nina Jehanno: Methodology, investigation, writing-original draft, writing—reviewing and editing. Sylvain Dureau: Methodology, Statistical analysis, writing-original draft, writing—reviewing and editing. Lieke Mous: Methodology, investigation, writing-original draft, writing—reviewing and editing. Line Claude Conceptualisation, writing-original draft, writing-reviewing and editing. Stelly Ballet: Methodology, investigation, writing-original draft, writing—reviewing and editing. Gaëlle Pierron: Resources, writing—review and editing. Nathalie Gaspar: Resources, writing-review and editing. Mehdi Brahmi: Resources, writing—review and editing. Thibaud Valentin: Resources, writing—review and editing. Gabriel Revon Riviere: Resources, writing—review and editing. Cyril Lervat: Resources, writing—review and editing. Jamie Probert: Resources, writing—review and editing. Natacha Entz-Werle: Resources, writing—review and editing. Ludovic Mansuy: Resources, writing-review and editing. Dominique Plantaz: Resources, writing—review and editing. Maria Rios: Resources, writing—review and editing. Laure Saumet: Resources, writing—review and editing. Cécile Vérité: Resources, writing—review and editing. Marie-Pierre Castex: Resources, writing-review and editing. Estelle Thebaud: Resources, writing—review and editing. Thibaut Cassou-Mounat: Resources, writing—review and editing. Anne-Sophie Plissonnier: Project administration. Jennifer Dieppedale: Resources and data monitoring. Olivier Delattre: Resources, writing—review and editing. Marie-Emmanuelle Legrier: Resources and data monitoring. Perrine Marec-Bérard: Resources, writing—review and editing. François Gouin: Resources, writing—review and editing. Pablo Berlanga: Resources, writing—review and editing. Camille Cordero: Resources, writing-review and editing. Didier Surdez: Conceptualisation, resources, writing—reviewing and editing. Nadège Corradini: Conceptualisation, methodology, investigation, writing-original draft, writing—reviewing and editing, funding acquisition.
Corresponding author
Ethics declarations
Competing interests
PB has consulting role (institutional funding) for Recordati and Merck, drugs for trials from Bayer, participation in educational activities organised by Recordati and is Member of the Executive Committee of the SIOPEN (European neuroblastoma research cooperative group), organisation which receives royalties for the sales of dinutuximab beta.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Laurence, V., Jehanno, N., Dureau, S. et al. Interest of a sequential multimodal approach for the treatment of newly diagnosed patients with multimetastatic Ewing sarcoma: results of the French prospective CombinaiR3 phase II trial. Br J Cancer 134, 279–288 (2026). https://doi.org/10.1038/s41416-025-03263-3
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41416-025-03263-3