Abstract
Purpose
Evidence on the prognostic and staging effects of lymphovascular invasion (LVI) after neoadjuvant chemoradiotherapy for esophageal squamous cell carcinoma (ESCC) is limited. We aimed to determine the prognostic value of LVI and develop a modified post-neoadjuvant pathologic staging (ypStage) system integrating LVI and ypTNM stage to improve risk stratification.
Methods
This multi-institutional study included patients with ESCC receiving neoadjuvant chemoradiotherapy and R0 resection. Recursive partitioning analysis (RPA) was conducted to derive prognostic groupings. A modified ypStage system was developed, validated, and compared with ypTNM stage.
Results
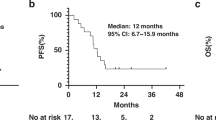
A total of 931 patients were divided into training (n = 565) and external validation (n = 366) cohorts. LVI was present in 115 patients (12.4%). LVI was an independent predictor of survival and disease recurrence, with hazard ratios of 1.70 for overall survival and 1.74 for recurrence-free survival. By integrating LVI status and ypTNM stage, nonmetastatic ESCC were classified into three stages with distinct prognoses. The proposed RPA stage provided superior hazard consistency, hazard discrimination, sample size balance, and outcome prediction over ypTNM stage.
Conclusions
LVI was a strong prognostic factor, independent of the current ypTNM stage in ESCC. We developed an RPA-based ypStage system integrating LVI status and ypTNM stage that exhibited good prognostic performance.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 24 print issues and online access
$259.00 per year
only $10.79 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
Deidentified patient data will be available by reasonable request to the corresponding author after institutional approval and with signed access agreement, with no time limits after publication.
References
Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, et al. Global Cancer Statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2021;71:209–49.
van Hagen P, Hulshof MC, van Lanschot JJ, Steyerberg EW, van Berge, Henegouwen MI, et al. Preoperative chemoradiotherapy for esophageal or junctional cancer. N Engl J Med. 2012;366:2074–84.
Yang H, Liu H, Chen Y, Zhu C, Fang W, Yu Z, et al. Neoadjuvant chemoradiotherapy followed by surgery versus surgery alone for locally advanced squamous cell carcinoma of the esophagus (NEOCRTEC5010): a phase III multicenter, randomized, open-label clinical trial. J Clin Oncol. 2018;36:2796–803.
Shah MA, Kennedy EB, Catenacci DV, Deighton DC, Goodman KA, Malhotra NK, et al. Treatment of locally advanced esophageal carcinoma: ASCO guideline. J Clin Oncol. 2020;38:2677–94.
Rice TW, Ishwaran H, Ferguson MK, Blackstone EH, Goldstraw P. Cancer of the esophagus and esophagogastric junction: an eighth edition staging primer. J Thorac Oncol. 2017;12:36–42.
Park SY, Park B, Yun JK, Kim HR, Kim YH, Jeon YJ, et al. Proposal of new ypstage grouping system for esophageal squamous cell carcinoma patients who underwent neoadjuvant chemoradiotherapy followed by surgery. Ann Surg. 2024;281:288–98.
Kang J, Lee HP, Kim HR, Kim JH, Jung HY, Lee GH, et al. Validation of the post-neoadjuvant staging system of the American joint committee on cancer, 8th edition, in patients treated with neoadjuvant chemoradiotherapy followed by curative esophagectomy for localized esophageal squamous cell carcinoma. Surg Oncol. 2020;35:491–7.
Huang Q, Luo K, Chen C, Wang G, Jin J, Kong M, et al. Identification and validation of lymphovascular invasion as a prognostic and staging factor in node-negative esophageal squamous cell carcinoma. J Thorac Oncol. 2016;11:583–92.
Hsu CP, Chuang CY, Hsu PK, Chien LI, Lin CH, Yeh YC, et al. Lymphovascular invasion as the major prognostic factor in node-negative esophageal cancer after primary esophagectomy. J Gastrointest Surg. 2020;24:1459–68.
Zhuang W, Wu H, Chen R, Ben X, Huang S, Zhou Z, et al. The staging performance of a modified tumor-node-metastasis staging system incorporated with lymphovascular invasion in patients with esophageal squamous cell carcinoma. Front Oncol. 2022;12:1018827.
Padera TP, Kadambi A, di Tomaso E, Carreira CM, Brown EB, Boucher Y, et al. Lymphatic metastasis in the absence of functional intratumor lymphatics. Science. 2002;296:1883–6.
Liebig C, Ayala G, Wilks JA, Berger DH, Albo D. Perineural invasion in cancer: a review of the literature. Cancer. 2009;115:3379–91.
Wang A, Tan Y, Wang S, Chen X. The prognostic value of separate lymphatic invasion and vascular invasion in oesophageal squamous cell carcinoma: a meta-analysis and systematic review. BMC Cancer. 2022;22:1329.
Tu CC, Hsu PK, Chien LI, Liu WC, Huang CS, Hsieh CC, et al. Prognostic histological factors in patients with esophageal squamous cell carcinoma after preoperative chemoradiation followed by surgery. BMC Cancer. 2017;17:62.
Chen WH, Huang YL, Chao YK, Yeh CJ, Chang HK, Tseng CK, et al. Prognostic significance of lymphovascular invasion in patients with esophageal squamous cell carcinoma treated with neoadjuvant chemoradiotherapy. Ann Surg Oncol. 2015;22:338–43.
Lagarde SM, Philips AW, Navidi M, Disep B, Immanuel A, Griffin SM. The presence of lymphovascular and perineural infiltration after neoadjuvant therapy and oesophagectomy identifies patients at high risk for recurrence. Br J Cancer. 2015;113:1427–33.
Chen JW, Xie JD, Ling YH, Li P, Yan SM, Xi SY, et al. The prognostic effect of perineural invasion in esophageal squamous cell carcinoma. BMC Cancer. 2014;14:313.
Groome PA, Schulze K, Boysen M, Hall SF, Mackillop WJ. A comparison of published head and neck stage groupings in carcinomas of the oral cavity. Head Neck. 2001;23:613–24.
Xie Y, Luo X, Li H, Xu Q, He Z, Zhao Q, et al. autoRPA: a web server for constructing cancer staging models by recursive partitioning analysis. Comput Struct Biotechnol J. 2020;18:3361–7.
Kelly RJ, Ajani JA, Kuzdzal J, Zander T, Van Cutsem E, Piessen G, et al. Adjuvant nivolumab in resected esophageal or gastroesophageal junction cancer. N Engl J Med. 2021;384:1191–203.
Oguma J, Ishiyama K, Kurita D, Kanematsu K, Kubo K, Utsunomiya D, et al. Significance of lymphovascular invasion in esophageal squamous cell carcinoma undergoing neoadjuvant chemotherapy followed by esophagectomy. Esophagus. 2023;20:215–24.
Rice TW, Ishwaran H, Kelsen DP, Hofstetter WL, Apperson-Hansen C, Blackstone EH, et al. Recommendations for neoadjuvant pathologic staging (ypTNM) of cancer of the esophagus and esophagogastric junction for the 8th edition AJCC/UICC staging manuals. Dis Esophagus. 2016;29:906–12.
Wong IYH, Chung JCY, Zhang RQ, Gao X, Lam KO, Kwong DLW, et al. A novel tumor staging system incorporating Tumor Regression Grade (TRG) with lymph node status (ypN-category) results in better prognostication than ypTNM stage groups after neoadjuvant therapy for esophageal squamous cell carcinoma. Ann Surg. 2022;276:784–91.
Jiang Y, Huang Y, Wang Z, Xu W, Xu J, Teng F, et al. Comparisons of prognosis prediction accuracy between modified and unmodified versions of 8th edition ypTNM. Ann Transl Med. 2022;10:600.
Albers P, Siener R, Kliesch S, Weissbach L, Krege S, Sparwasser C, et al. Risk factors for relapse in clinical stage I nonseminomatous testicular germ cell tumors: results of the German Testicular Cancer Study Group Trial. J Clin Oncol. 2003;21:1505–12.
Vauthey JN, Lauwers GY, Esnaola NF, Do KA, Belghiti J, Mirza N, et al. Simplified staging for hepatocellular carcinoma. J Clin Oncol. 2002;20:1527–36.
Acknowledgements
We thank Prof. Jibin Li for statistical consultation.
Funding
This work was supported by the grants from the National Natural Science Foundation of China (No.82172669; 82373214), Basic Public Welfare Research Program of Zhejiang Province (LGF21H160005), Beijing Xisike Clinical Oncology Research Foundation (Y-MSDZD2022-0878), Guangdong Basic and Applied Basic Research Foundation (2025B1515020090), and Young Talents Program of Sun Yat-sen University Cancer Center (PT22121101).
Author information
Authors and Affiliations
Contributions
Conception and design: Qiaoqiao Li, Hong Yang, and Mian Xi. Provision of study material or patients: Yujin Xu, Xufeng Guo, Wencheng Zhang, Geng Wang, Qiaoqiao Li, Hong Yang, and Mian Xi. Collection and assembly of data: Shiliang Liu, Gang Zhao, Ruiqi Wang, Yihong Ling, Baoqing Chen, and Mian Xi. Data analysis and interpretation: Shiliang Liu, Li Zhang, Baoqing Chen, Qiaoqiao Li, and Mian Xi. Manuscript writing and final approval: All authors. All authors had full access to the data, participated in reviewing and editing of the manuscript, and approved the final version before submission. Three principal investigators (QL, HY, and MX) verified the raw data of the study and had final responsibility for the decision to submit for publication.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
No patient’s personal identifying information is included in this manuscript. This study was approved by the Institutional Review Boards of each participating center and informed consent was waived owing to its retrospective design. The study was performed in accordance with the Declaration of Helsinki.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Liu, S., Xu, Y., Guo, X. et al. Integrating lymphovascular invasion and ypTNM staging system for esophageal squamous cell carcinoma undergoing neoadjuvant chemoradiotherapy and surgery: a multi-institutional analysis. Br J Cancer 134, 608–617 (2026). https://doi.org/10.1038/s41416-025-03314-9
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41416-025-03314-9