Abstract
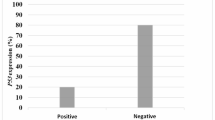
Alpha-synuclein (α-syn), encoded by the SNCA gene, is a major participant in the pathophysiology of Parkinson’s disease (PD). Its functions have been reported to be related to apoptosis induction, the elevation of oxidative stress, mitochondrial homeostasis, cell-cycle aberrations, and DNA-related interactions. Evidence obtained in recent studies suggests a possible link between α-syn and cancer development. Bladder cancer (BCa) is the second most common genitourinary malignancy, with the population of survivors of BCa increasing worldwide. In this study, we show that α-syn expression was significantly downregulated in BCa. In vitro and in vivo experiments showed that α-syn could significantly inhibit BCa cell proliferation by arresting the cell cycle in the S phase via upregulation of p53 expression mediated by DNA damages. Further experiments showed that overexpression of α-syn delivered by adeno-associated viruses (AAVs) exerted inhibitory effects on the growth of BCa tumors. These findings indicate that αα-syn is a functional tumor suppressor that can inhibit the proliferation of BCa cells by activating the p53/p21 signaling pathway. Our present study provides insights into the roles of α-syn in BCa and suggests that α-syn may be a novel therapeutic target for the treatment of BCa.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Burger M, Catto JWF, Dalbagni G, Grossman HB, Herr H, Karakiewicz P, et al. Epidemiology and risk factors of urothelial bladder cancer. Eur Urol. 2013;63:234–41.
Lin P, Chang S, Hsiao T, Huang B, Lin C, Yang P. Association between parkinson disease and risk of cancer in Taiwan. JAMA Oncol. 2015;1:633.
Catalá-López F, Suárez-Pinilla M, Suárez-Pinilla P, Valderas JM, Gómez-Beneyto M, Martinez S, et al. Inverse and direct cancer comorbidity in people with central nervous system disorders: a meta-analysis of cancer incidence in 577,013 participants of 50 observational studies. Psychother Psychosom. 2014;83:89–105.
Ong EL, Goldacre R, Goldacre M. Differential risks of cancer types in people with Parkinson’s disease: a national record-linkage study. Eur J Cancer. 2014;50:2456–62.
Zhang P, Liu B. Association between Parkinson’s disease and risk of cancer: a PRISMA-compliant meta-analysis. ACS Chem Neurosci. 2019;10:4430–9.
Feng DD, Cai W, Chen X. The associations between Parkinson’s disease and cancer: the plot thickens. Transl Neurodegener. 2015;4:20.
Polymeropoulos MH, Lavedan C, Leroy E, Ide SE, Dehejia A, Dutra A, et al. Mutation in the alpha-synuclein gene identified in families with Parkinson’s disease. Science. 1997;276:2045–7.
Spillantini MG, Schmidt ML, Lee VM, Trojanowski JQ, Jakes R, Goedert M. Alpha-synuclein in Lewy bodies. Nature. 1997;388:839–40.
Burré J, Sharma M, Südhof TC. Cell biology and pathophysiology of α-synuclein. CSH Perspect Med. 2018;8:a24091.
Wales P, Pinho R, Lázaro DF, Outeiro TF. Limelight on alpha-synuclein: pathological and mechanistic implications in neurodegeneration. J Parkinson’s Dis. 2013;3:415–59.
Bruening W, Giasson BI, Klein-Szanto AJ, Lee VM, Trojanowski JQ, Godwin AK. Synucleins are expressed in the majority of breast and ovarian carcinomas and in preneoplastic lesions of the ovary. Cancer Am Cancer Soc. 2000;88:2154–63.
Ye Q, Wang TF, Peng YF, Xie J, Feng B, Qiu MY, et al. Expression of alpha-, beta- and gamma-synuclein in colorectal cancer, and potential clinical significance in progression of the disease. Oncol Rep. 2010;23:429–36.
Turriani E, Lázaro DF, Ryazanov S, Leonov A, Giese A, Schön M, et al. Treatment with diphenyl–pyrazole compound anle138b/c reveals that α-synuclein protects melanoma cells from autophagic cell death. Proc Natl Acad Sci USA. 2017;114:E4971–7.
Kawashima M, Suzuki SO, Doh-ura K, Iwaki T. alpha-Synuclein is expressed in a variety of brain tumors showing neuronal differentiation. Acta Neuropathol. 2000;99:154–60.
Yan Y, Xu Z, Hu X, Qian L, Li Z, Zhou Y, et al. SNCA is a functionally low-expressed gene in lung adenocarcinoma. Genes. 2018;9:16.
Tang Z, Kang B, Li C, Chen T, Zhang Z. GEPIA2: an enhanced web server for large-scale expression profiling and interactive analysis. Nucleic Acids Res. 2019;47:W556–60.
Chandrashekar DS, Bashel B, Balasubramanya S, Creighton CJ, Ponce-Rodriguez I, Chakravarthi B, et al. UALCAN: a portal for facilitating tumor subgroup gene expression and survival analyses. Neoplasia. 2017;19:649–58.
Li M, Gu FL, Li WB, Song YS, Zhou AR, Guo YL. Introduction of wild-type p53 gene downregulates the expression of H-ras gene and suppresses the growth of bladder cancer cells. Urol Res. 1995;23:311–4.
Trapnell C, Roberts A, Goff L, Pertea G, Kim D, Kelley DR, et al. Differential gene and transcript expression analysis of RNA-seq experiments with TopHat and Cufflinks. Nat Protoc. 2012;7:562–78.
Anders S, Pyl PT, Huber W. HTSeq-a Python framework to work with high-throughput sequencing data. Bioinformatics. 2015;31:166–9.
Anders S, Huber W. Differential expression analysis for sequence count data. Genome Biol. 2010;11:R106.
Benjamini Y, Drai D, Elmer G, Kafkafi N, Golani I. Controlling the false discovery rate in behavior genetics research. Behav Brain Res. 2001;125:279–84.
Zeng K, Wang Z, Ohshima K, Liu Y, Zhang W, Wang L, et al. BRAF V600E mutation correlates with suppressive tumor immune microenvironment and reduced disease-free survival in Langerhans cell histiocytosis. Oncoimmunology. 2016;5:e1185582.
Du T, Wu Z, Luo H, Lu S, Ma K. Injection of α-syn-98 aggregates into the brain triggers α-synuclein pathology and an inflammatory response. Front Mol Neurosci. 2019;12:189.
Paiva I, Pinho R, Pavlou MA, Hennion M, Wales P, Schütz A, et al. Sodium butyrate rescues dopaminergic cells from alpha-synuclein-induced transcriptional deregulation and DNA damage. Hum Mol Genet. 2017;26:2231–46.
Derech-Haim S, Friedman Y, Hizi A, Bakhanashvili M. p53 regulates its own expression by an intrinsic exoribonuclease activity through AU-rich elements. J Mol Med. 2020;98:437–49.
Milanese C, Cerri S, Ulusoy A, Gornati SV, Plat A, Gabriels S, et al. Activation of the DNA damage response in vivo in synucleinopathy models of Parkinson’s disease. Cell Death Dis. 2018;9:818.
Viswambaram P, Hayne D. Gender discrepancies in bladder cancer: potential explanations. Expert Rev Anticanc. 2020;20:841–9.
Li W, Zhang H, Guo Q, Wu X, Xu Z, Dang C. et al. Detection of SNCA and FBN1 methylation in the stool as a biomarker for colorectal cancer. Dis Markers. 2015;2015:1–6.
Zou J, Yin F, Wang Q, Zhang W, Li L. Analysis of microarray-identified genes and microRNAs associated with drug resistance in ovarian cancer. Int J Clin Exp Pathol. 2015;8:6847.
Li YX, Yu ZW, Jiang T, Shao LW, Liu Y, Li N, et al. SNCA, a novel biomarker for Group 4 medulloblastomas, can inhibit tumor invasion and induce apoptosis. Cancer Sci. 2018;109:1263–75.
Israeli E, Yakunin E, Zarbiv Y, Hacohen-Solovich A, Kisos H, Loeb V, et al. α-synuclein expression selectively affects tumorigenesis in mice modeling Parkinson’s disease. PLoS ONE. 2011;6:e19622.
Ge Y, Xu K. Alpha-synuclein contributes to malignant progression of human meningioma via the Akt/mTOR pathway. Cancer Cell Int. 2016;16:86.
Shekoohi S, Rajasekaran S, Patel D, Yang S, Liu W, Huang S, et al. Knocking out alpha-synuclein in melanoma cells dysregulates cellular iron metabolism and suppresses tumor growth. Sci Rep. 2021;11:5267.
Vousden KH. p53: death star. Cell. 2000;103:691–4.
Yogosawa S, Yoshida K. Tumor suppressive role for kinases phosphorylating p53 inDNA damage‐induced apoptosis. Cancer Sci. 2018;109:3376–82.
Alves DCC, Checler F. Apoptosis in Parkinson’s disease: is p53 the missing link between genetic and sporadic Parkinsonism? Cell Signal. 2011;23:963–8.
Karunakaran S, Saeed U, Mishra M, Valli RK, Datt Joshi S, Meka DP, et al. Selective activation of p38 mitogen-activated protein kinase in dopaminergic neurons of substantia nigra leads to nuclear translocation of p53 in 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine-treated mice. J Neurosci. 2008;28:12500–9.
Martin LJ. Parkinson’s disease-synuclein transgenic mice develop neuronal mitochondrial degeneration and cell death. J Neurosci. 2006;26:41–50.
Karim MR, Liao EE, Kim J, Meints J, Martinez HM, Pletnikova O et al. α-Synucleinopathy associated c-Abl activation causes p53-dependent autophagy impairment. Mol Neurodegener. 2020;15:27.
Sang Q, Liu X, Wang L, Qi L, Sun W, Wang W, et al. CircSNCA downregulation by pramipexole treatment mediates cell apoptosis and autophagy in Parkinson’s disease by targeting miR-7. Aging. 2018;10:1281–93.
Duplan E, Giordano C, Checler F, Alves DCC. Direct alpha-synuclein promoter transactivation by the tumor suppressor p53. Mol Neurodegener. 2016;11:13.
Checler F, Alves DCC. p53 in neurodegenerative diseases and brain cancers. Pharm Ther. 2014;142:99–113.
Chen HY, Lin CH, Teng SC. Stress-induced p53 drives BAG5 cochaperone expression to control alpha-synuclein aggregation in Parkinson’s disease. Aging. 2020;12:20702–27.
Alves Da Costa C, Paitel E, Vincent B, Checler F. α-synuclein lowers p53-dependent apoptotic response of neuronal cells. J Biol Chem. 2002;277:50980–4.
Xu J, Kao SY, Lee FJ, Song W, Jin LW, Yankner BA. Dopamine-dependent neurotoxicity of alpha-synuclein: a mechanism for selective neurodegeneration in Parkinson disease. Nat Med. 2002;8:600–6.
Jones RG, Plas DR, Kubek S, Buzzai M, Mu J, Xu Y, et al. AMP-activated protein kinase induces a p53-dependent metabolic checkpoint. Mol Cell. 2005;18:283–93.
Das U, Manna K, Khan A, Sinha M, Biswas S, Sengupta A, et al. Ferulic acid (FA) abrogates γ-radiation induced oxidative stress and DNA damage by up-regulating nuclear translocation of Nrf2 and activation of NHEJ pathway. Free Radic. Res. 2016;51:47–63.
Dhuppar S, Mazumder A. Measuring cell cycle-dependent DNA damage responses and p53 regulation on a cell-by-cell basis from image analysis. Cell Cycle. 2018;17:1358–71.
Stewart-Ornstein J, Lahav G. p53 dynamics in response to DNA damage vary across cell lines and are shaped by efficiency of DNA repair and activity of the kinase ATM. Sci Signal. 2017;10:h6671.
Vasquez V, Mitra J, Hegde PM, Pandey A, Sengupta S, Mitra S, et al. Chromatin-bound oxidized α-synuclein causes strand breaks in neuronal genomes in in vitro models of Parkinson’s disease. J Alzheimer’s Dis. 2017;60:S133–50.
Hegde ML, Gupta VB, Anitha M, Harikrishna T, Shankar SK, Muthane U, et al. Studies on genomic DNA topology and stability in brain regions of Parkinson’s disease. Arch Biochem Biophys. 2006;449:143–56.
Wang D, Yu T, Liu Y, Yan J, Guo Y, Jing Y, et al. DNA damage preceding dopamine neuron degeneration in A53T human α-synuclein transgenic mice. Biochem Biophys Res Commun. 2016;481:104–10.
Schaser AJ, Osterberg VR, Dent SE, Stackhouse TL, Wakeham CM, Boutros SW et al. Alpha-synuclein is a DNA binding protein that modulates DNA repair with implications for Lewy body disorders. Sci Rep. 2019;9:10919.
He P, Li Z, Xu F, Ru G, Huang Y, Lin E et al. AMPK activity contributes to G2 arrest and DNA damage decrease via p53/p21 pathways in oxidatively damaged mouse zygotes. Front Cell Dev Biol. 2020;8:539485.
Wang Y, Wu H, Dong N, Su X, Duan M, Wei Y et al. Sulforaphane induces S-phase arrest and apoptosis via p53-dependent manner in gastric cancer cells. Sci Rep. 2021;11:2504.
Wu G, Lin N, Xu L, Liu B, Feitelson MA. UCN-01 induces S and G2/M cell cycle arrest through the p53/p21(waf1) or CHK2/CDC25C pathways and can suppress invasion in human hepatoma cell lines. BMC Cancer. 2013;13:167.
Liu H, Luo Q, Cui H, Deng H, Kuang P, Lu Y, et al. Sodium fluoride causes hepatocellular S-phase arrest by activating ATM-p53-p21 and ATR-Chk1-Cdc25A pathways in mice. Oncotarget. 2018;9:4318–37.
Su Y, Reshi L, Chen L, Li W, Chiu H, Hong J. Nuclear targeting of the betanodavirus B1 protein via two arginine-rich domains induces G1/S cell cycle arrest mediated by upregulation of p53/p21. Sci Rep. 2018;8:3079.
Gudkov AV, Komarova EA. Pathologies associated with the p53 response. CSH Perspect Biol. 2010;2:a1180.
Hacker UT, Bentler M, Kaniowska D, Morgan M, Büning H. Towards clinical implementation of adeno-associated virus (AAV) vectors for cancer gene therapy: current status and future perspectives. Cancers. 2020;12:1889.
Santiago-Ortiz JL, Schaffer DV. Adeno-associated virus (AAV) vectors in cancer gene therapy. J Control Release. 2016;240:287–301.
Wu CL, Shieh GS, Chang CC, Yo YT, Su CH, Chang MY, et al. Tumor-selective replication of an oncolytic adenovirus carrying Oct-3/4 response elements in murine metastatic bladder cancer models. Clin Cancer Res. 2008;14:1228–38.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (No. 81860453), Natural Science Foundation of Yunnan Province (Nos. 2018ZF009; 2018FE001 (-006); 202001AT070144), and CAMS Innovation Fund for Medical Sciences (No. 2016-I2M-2-001).
Author information
Authors and Affiliations
Contributions
KM and DY discussed and designed this study. ZW, CX, FL, YO, and JG performed all experiments. The manuscript was written by ZW and CX. CZ and DT revised the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Wu, Z., Xia, C., Zhang, C. et al. Adeno-associated virus-delivered alpha synuclein inhibits bladder cancer growth via the p53/p21 signaling pathway. Cancer Gene Ther 29, 1193–1206 (2022). https://doi.org/10.1038/s41417-022-00425-w
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41417-022-00425-w
This article is cited by
-
Exploring α-Syn’s Functions Through Ablation Models: Physiological and Pathological Implications
Cellular and Molecular Neurobiology (2025)