Abstract
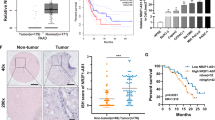
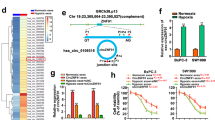
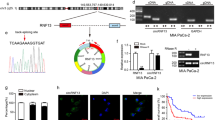
This study revealed the prospective biological role and fundamental mechanisms of hypoxia-induced lncRNA-MANCR (MANCR), which is notably upregulated in pancreatic cancer (PC). This work uncovered the potential biological function and underlying mechanisms of hypoxia-induced MANCR, which is significantly elevated in PC. Microarray assays confirmed MANCR expression in the tissues of patients with PC and patients with chronic pancreatitis (CP), which positively correlated with sirtuin-1 (SIRT1) mRNA levels. Chromatin immunoprecipitation and luciferase assays were employed to gauge binding within the hypoxia-inducible factor-1α (HIF-1α)/MANCR/miRNA-494/SIRT1 pathway. Additionally, the association between MANCR expression and the clinical outcomes of patients with PC was confirmed. MANCR is significantly upregulated in PC cells under hypoxic conditions, which is closely linked to poor prognosis in patients with PC. Depletion of MANCR repressed in vitro proliferation, migration, and invasion of PC cells and in vivo growth of PC xenograft tumours. We further demonstrated that MANCR is localised in the cytoplasm and competitively binds miR-494, which directly targets SIRT1. Mechanically, the overexpression of SIRT1 improved the stability of the HIF-1α protein through deacetylation, leading to enhanced HIF-1α assembly. Moreover, MANCR underwent transcriptional regulation by HIF-1α in a hypoxic setting. This modulation was ascribed to HIF-1α binding to hypoxia response elements present in the MANCR promoter sequence. Data revealed the potential possibility of feedback between MANCR and HIF-1α, which may be conducive to hypoxia-induced oncogenicity and PC tumorigenesis, thereby providing a suitable therapeutic target.

This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
Data availability
Upon reasonable request, the corresponding authors will provide all data that supports the findings of this study.
References
Halbrook CJ, Lyssiotis CA, Pasca di Magliano M, Maitra A. Pancreatic cancer: Advances and challenges. Cell. 2023;186:1729–54.
Siegel RL, Miller KD, Fuchs HE, Jemal A. Cancer Statistics, 2021. CA Cancer J Clin. 2021;71:7–33.
Wood LD, Canto MI, Jaffee EM, Simeone DM. Pancreatic Cancer: Pathogenesis, Screening, Diagnosis, and Treatment. Gastroenterology. 2022;163:386–402.e1.
Duan H, Li L, He S. Advances and Prospects in the Treatment of Pancreatic Cancer. Int J Nanomed. 2023;18:3973–88.
He Z, Wang J, Zhu C, Xu J, Chen P, Jiang X, et al. Exosome-derived FGD5-AS1 promotes tumor-associated macrophage M2 polarization-mediated pancreatic cancer cell proliferation and metastasis. Cancer Lett. 2022;548:215751.
Zhang Y, Liu X, Wang Y, Lai S, Wang Z, Yang Y, et al. The m6A demethylase ALKBH5-mediated upregulation of DDIT4-AS1 maintains pancreatic cancer stemness and suppresses chemosensitivity by activating the mTOR pathway. Mol Cancer. 2022;21:174.
Cave DD, Buonaiuto S, Sainz B Jr, Fantuz M, Mangini M, Carrer A, et al. LAMC2 marks a tumor-initiating cell population with an aggressive signature in pancreatic cancer. J Exp Clin Cancer Res. 2022;41:315.
Fatica A, Bozzoni I. Long non-coding RNAs: new players in cell differentiation and development. Nat Rev Genet. 2014;15:7–21.
Wang J, He Z, Liu X, Xu J, Jiang X, Quan G, et al. LINC00941 promotes pancreatic cancer malignancy by interacting with ANXA2 and suppressing NEDD4L-mediated degradation of ANXA2. Cell Death Dis. 2022;13:718.
Zhou M, Pan S, Qin T, Zhao C, Yin T, Gao Y, et al. LncRNA FAM83H-AS1 promotes the malignant progression of pancreatic ductal adenocarcinoma by stabilizing FAM83H mRNA to protect β-catenin from degradation. J Exp Clin Cancer Res. 2022;41:288.
Jiang F, Li S, Wang X, Deng Y, Peng S. DPP10-AS1-mediated downregulation of MicroRNA-324-3p Is conducive to the malignancy of pancreatic cancer by enhancing CLDN3 Expression. Pancreas. 2022;51:1201–10.
Terashima M, Ishimura A, Wanna-Udom S, Suzuki T. MEG8 long noncoding RNA contributes to epigenetic progression of the epithelial-mesenchymal transition of lung and pancreatic cancer cells. J Biol Chem. 2018;293:18016–30.
Xu F, Huang M, Chen Q, Niu Y, Hu Y, Hu P, et al. LncRNA HIF1A-AS1 promotes gemcitabine resistance of pancreatic cancer by enhancing glycolysis through modulating the AKT/YB1/HIF1a pathway. Cancer Res. 2021;81:5678–91.
Liu Y, Shi M, He X, Cao Y, Liu P, Li F, et al. LncRNA-PACERR induces pro-tumour macrophages via interacting with miR-671-3p and m6A-reader IGF2BP2 in pancreatic ductal adenocarcinoma. J Hematol Oncol. 2022;15:52.
Qiu X, Shi Q, Zhang X, Shi X, Jiang H, Qin S. LncRNA A2M-AS1 promotes ferroptosis in pancreatic cancer via interacting with PCBP3. Mol Cancer Res. 2022;20:1636–45.
Li X, Deng SJ, Zhu S, Jin Y, Cui SP, Chen JY, et al. Hypoxia-induced lncRNA-NUTF2P3-001 contributes to tumorigenesis of pancreatic cancer by derepressing the miR-3923/KRAS pathway. Oncotarget. 2016;7:6000–14.
Jin Y, Zhang Z, Yu Q, Zeng Z, Song H, Huang X, et al. Positive reciprocal feedback of lncRNA ZEB1-AS1 and HIF-1α contributes to hypoxia-promoted tumorigenesis and metastasis of pancreatic cancer. Front Oncol. 2021;11:761979.
Hinshaw DC, Shevde LA. The tumor microenvironment innately modulates cancer progression. Cancer Res. 2019;79:4557–66.
Jung HY, Fattet L, Yang J. Molecular pathways: linking tumor microenvironment to epithelial-mesenchymal transition in metastasis. Clin Cancer Res. 2015;21:962–8.
Jing X, Yang F, Shao C, Wei K, Xie M, Shen H, et al. Role of hypoxia in cancer therapy by regulating the tumor microenvironment. Mol Cancer. 2019;18:157.
Tao J, Yang G, Zhou W, Qiu J, Chen G, Luo W, et al. Targeting hypoxic tumor microenvironment in pancreatic cancer. J Hematol Oncol. 2021;14:14.
Liu M, Zhong J, Zeng Z, Huang K, Ye Z, Deng S, et al. Hypoxia-induced feedback of HIF-1a and lncRNA-CF129 contributes to pancreatic cancer progression through stabilization of p53 protein. Theranostics. 2019;9:4795–810.
Niu Y, Jin Y, Deng SC, Deng SJ, Zhu S, Liu Y, et al. MiRNA-646-mediated reciprocal repression between HIF-1a and MIIP contributes to tumorigenesis of pancreatic cancer. Oncogene. 2018;37:1743–58.
Deng S, Li X, Niu Y, Zhu S, Jin Y, Deng S, et al. MiR-652 inhibits acidic microenvironment induced epithelial-mesenchymal transition of pancreatic cancer cells by targeting ZEB1. Oncotarget. 2015;6:39661–75.
Zhu S, Zhou HY, Deng SC, Deng SJ, He C, Li X, et al. ASIC1 and ASIC3 contribute to acidity-induced EMT of pancreatic cancer through activating Ca2+/RhoA pathway. Cell Death Dis. 2017;8: e2806 https://doi.org/10.1038/cddis.2017.189.
Pastushenko I, Blanpain C. EMT transition states during tumor progression and metastasis. Trends Cell Biol. 2019;29:212–26.
Brabletz S, Schuhwerk H, Brabletz T, Stemmler MP. Dynamic EMT: a multi-tool for tumor progression. EMBO J. 2021;40:e108647.
Choupani J, Mansoori Derakhshan S, Bayat S, Alivand MR, Shekari Khaniani M. Narrower insight to SIRT1 role in cancer: A potential therapeutic target to control epithelial-mesenchymal transition in cancer cells. J Cell Physiol. 2018;233:4443–57.
Palmirotta R, Cives M, Della-Morte D, Capuani B, Lauro D, Guadagni F, et al. Sirtuins and cancer: role in the epithelial-mesenchymal transition. Oxid Med Cell Longev. 2016;2016. 3031459.
Deng S, Zhu S, Wang B, Li X, Liu Y, Qin Q, et al. Chronic pancreatitis and pancreatic cancer demonstrate active epithelial-mesenchymal transition profile, regulated by miR-217-SIRT1 pathway. Cancer Lett. 2014;355:184–91.
Zhou L, Jiang J, Huang Z, Jin P, Peng L, Luo M, et al. Hypoxia-induced lncRNA STEAP3-AS1 activates Wnt/β-catenin signaling to promote colorectal cancer progression by preventing m6A-mediated degradation of STEAP3 mRNA. Mol Cancer. 2022;21:168.
Li X, Yang J, Ni R, Chen J, Zhou Y, Song H, et al. Hypoxia-induced lncRNA RBM5-AS1 promotes tumorigenesis via activating Wnt/β-catenin signaling in breast cancer. Cell Death Dis. 2022;13:95.
Huang J, Zheng W, Zhang P, Lin Q, Chen Z, Xuan J, et al. ChIPBase v3.0: the encyclopedia of transcriptional regulations of non-coding RNAs and protein-coding genes. Nucleic Acids Res. 2023;51:D46–D56.
Yao L, Yan J, Gan L, Huang S, Cheng F, Fang N. Upregulation of MANCR predicts poor survival in patients with gastric cancer. Oncol Lett. 2019;18:6801–6.
Nagasawa M, Tomimatsu K, Terada K, Kondo K, Miyazaki K, Miyazaki M, et al. Long non-coding RNA MANCR is a target of BET bromodomain protein BRD4 and plays a critical role in cellular migration and invasion abilities of prostate cancer. Biochem Biophys Res Commun. 2020;526:128–34.
Tahmouresi F, Razmara E, Pakravan K, Mossahebi-Mohammadi M, Rouhollah F, Montazeri M, et al. Upregulation of the long noncoding RNAs DSCAM-AS1 and MANCR is a potential diagnostic marker for breast carcinoma. Biotechnol Appl Biochem. 2021;68:1250–6.
Liu C, Li H, Li X, Zhao X, Zhang X. LncRNA MANCR positively affects the malignant progression of lung adenocarcinoma. BMC Pulm Med. 2021;21:272.
Jung JG, Le A. Metabolism of immune cells in the tumor microenvironment. Adv Exp Med Biol. 2021;1311:173–85.
Li J, Eu JQ, Kong LR, Wang L, Lim YC, Goh BC, et al. Targeting metabolism in cancer cells and the tumour microenvironment for cancer therapy. Molecules. 2020;25:4831.
Singleton DC, Macann A, Wilson WR. Therapeutic targeting of the hypoxic tumour microenvironment. Nat Rev Clin Oncol. 2021;18:751–72.
Wei X, Chen Y, Jiang X, Peng M, Liu Y, Mo Y, et al. Mechanisms of vasculogenic mimicry in hypoxic tumor microenvironments. Mol Cancer. 2021;20:7.
Tan Z, Xu J, Zhang B, Shi S, Yu X, Liang C. Hypoxia: a barricade to conquer the pancreatic cancer. Cell Mol Life Sci. 2020;77:3077–83.
Hua Q, Mi B, Xu F, Wen J, Zhao L, Liu J, et al. Hypoxia-induced lncRNA-AC020978 promotes proliferation and glycolytic metabolism of non-small cell lung cancer by regulating PKM2/HIF-1a axis. Theranostics. 2020;10:4762–78.
Carmeliet P, Dor Y, Herbert JM, Fukumura D, Brusselmans K, Dewerchin M, et al. Role of HIF-1alpha in hypoxia-mediated apoptosis, cell proliferation and tumour angiogenesis. Nature. 1998;394:485–90.
Sokkar P, Sathis V, Ramachandran M. Computational modeling on the recognition of the HRE motif by HIF-1: molecular docking and molecular dynamics studies. J Mol Model. 2012;18:1691–700.
Maxwell PH, Wiesener MS, Chang GW, Clifford SC, Vaux EC, Cockman ME, et al. The tumour suppressor protein VHL targets hypoxia-inducible factors for oxygen-dependent proteolysis. Nature. 1999;399:271–5.
Jeong JW, Bae MK, Ahn MY, Kim SH, Sohn TK, Bae MH, et al. Regulation and destabilization of HIF-1alpha by ARD1-mediated acetylation. Cell. 2002;111:709–20.
Kim SH, Jeong JW, Park JA, Lee JW, Seo JH, Jung BK, et al. Regulation of the HIF-1alpha stability by histone deacetylases. Oncol Rep. 2007;17:647–51.
Jiang Y, Luo Z, Gong Y, Fu Y, Luo Y. NAD+ supplementation limits triple-negative breast cancer metastasis via SIRT1-P66Shc signaling. Oncogene. 2023;42:808–24.
Xie M, Liu M, He CS. SIRT1 regulates endothelial Notch signaling in lung cancer. PLoS One. 2012;7:e45331.
Yang D, Zhang P, Yang Z, Hou G, Yang Z. miR-4461 inhibits liver cancer stem cells expansion and chemoresistance via regulating SIRT1. Carcinogenesis. 2024;45:463–74.
Yu L, Dong L, Li H, Liu Z, Luo Z, Duan G, et al. Ubiquitination-mediated degradation of SIRT1 by SMURF2 suppresses CRC cell proliferation and tumorigenesis. Oncogene. 2020;39:4450–64.
Huang J, Zhang F, Hu G, Pan Y, Sun W, Jiang L, et al. SIRT1 suppresses pituitary tumor progression by downregulating PTTG1 expression. Oncol Rep. 2022;48:143.
Dilmac S, Kuscu N, Caner A, Yildirim S, Yoldas B, Farooqi AA, et al. SIRT1/FOXO signaling pathway in breast cancer progression and metastasis. Int J Mol Sci. 2022;23:10227.
Leng S, Huang W, Chen Y, Yang Y, Feng D, Liu W, et al. SIRT1 coordinates with the CRL4B complex to regulate pancreatic cancer stem cells to promote tumorigenesis. Cell Death Differ. 2021;28:3329–43.
Lim JH, Lee YM, Chun YS, Chen J, Kim JE, Park JW. Sirtuin 1 modulates cellular responses to hypoxia by deacetylating hypoxia-inducible factor 1alpha. Mol Cell. 2010;38:864–78.
Joo HY, Yun M, Jeong J, Park ER, Shin HJ, Woo SR, et al. SIRT1 deacetylates and stabilizes hypoxia-inducible factor-1a (HIF-1a) via direct interactions during hypoxia. Biochem Biophys Res Commun. 2015;462:294–300.
Xue ST, Zheng B, Cao SQ, Ding JC, Hu GS, Liu W, et al. Long non-coding RNA LINC00680 functions as a ceRNA to promote esophageal squamous cell carcinoma progression through the miR-423-5p/PAK6 axis. Mol Cancer. 2022;21:69.
Song YX, Sun JX, Zhao JH, Yang YC, Shi JX, Wu ZH, et al. Non-coding RNAs participate in the regulatory network of CLDN4 via ceRNA-mediated miRNA evasion. Nat Commun. 2017;8:289.
Liu Y, Li X, Zhu S, Zhang JG, Yang M, Qin Q, et al. Ectopic expression of miR-494 inhibited the proliferation, invasion and chemoresistance of pancreatic cancer by regulating SIRT1 and c-Myc. Gene Ther. 2015;22:729–38.
Acknowledgements
Not applicable.
Funding
This study was funded by National Natural Science Foundation of China (NO. 82370651, NO. 82070657, NO. 81902515), University Natural Science Research Project of Anhui Province (NO. 2024AH051959 and NO. KJ2019A0412), AnHui Province Educational Natural Science Project (2023AH040254) and Key Science Foundation of Wannan medical college (WK2023ZZD26 and WK2023ZZD25).
Author information
Authors and Affiliations
Contributions
Yan Jin, Hao Hu, and Yitong Tian conceived and coordinated the study, drafted and revised the manuscript. Long Cheng and Xiaoyu Guo performed the cell culture and some cellular experiments. Xiaoxu Huang, Han Xu, and Qiao Yu performed the molecular experiments. Zongwei Wang, Xiaoming Wang and Gang Wang analyzed the data. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing financial interests or personal relationships that may have influenced the work reported in this study.
Ethics approval and consent to participate
The present study was conducted in accordance with the relevant guidelines and regulations that govern the ethical conduct of research in this field. The human patient consent forms and protocols for this study were approved by the Wannan Medical College Ethics Committee (No: Wannan Medical College IRB2020011), and written informed consent was obtained from all participants prior to their inclusion in the study. The ethical policies and procedures for animal experiments were approved by the Animal Ethics Committee of the Academic Medical Centre at Wannan Medical College (Wuhu, China) (No: LLSC-2020-081). All animal care and experimental procedures adhered to the guidelines outlined in the Guide for the Care and Use of Laboratory Animals (2011). It is also noteworthy that no identifiable images or other personal data of participants were used in this study.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Jin, Y., Hu, H., Tian, Y. et al. The role of LncRNA-MANCR induced by HIF-1α drive the malignant progression of pancreatic cancer by targeting miRNA-494/SIRT1 signaling axis under hypoxic conditions. Cancer Gene Ther 32, 633–648 (2025). https://doi.org/10.1038/s41417-025-00900-0
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41417-025-00900-0
This article is cited by
-
HypoxamiRs in pancreatic cancer: master regulators of the hypoxic tumor microenvironment
Cell Biology and Toxicology (2025)