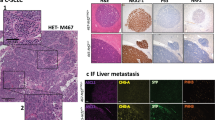
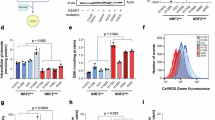
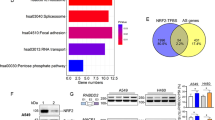
Abstract
The NRF2 signaling pathway promotes tumor initiation, progression and resistance to chemotherapy, radiation therapy and immune checkpoint inhibitors. The mechanisms underlying the biology of NRF2-active tumors are varied and include altered cellular metabolism, a reductive shift in redox state, and immunosuppression. Here we determined the molecular and phenotypic impact of NRF2 activation on two human non-small cell lung cancer (NSCLC) cell models. Inducible expression of NRF2E79Q, a common activating NRF2 mutation, in H358 lung adenocarcinoma (LUAD) cells altered cellular morphology and increased xenograft tumor growth in mice but not in 2D cell culture. In contrast, NRF2E79Q expression in H596 lung adeno-squamous cell carcinoma altered cellular morphology, increased neuroendocrine marker gene expression, but did not impact tumor growth in 2D or in xenografts. Gene expression profiling revealed shared and unique NRF2 transcriptional programs between these models, some of which were shared in primary lung tumors. Collectively, our findings reveal context-dependent effects of NRF2 activation on the growth and differentiation state of two human NSCLC models, supporting a role for NRF2 activation in altering the differentiation of human NSCLC during tumor progression.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The authors confirm that the data supporting the findings of this study are available within the article and its Supplementary Figures and Tables. The RNA-seq data have been deposited in the NCBI GEO database (GSE289043). Raw data that support the findings of this study are available from the corresponding author (SHH), upon reasonable request.
References
Siegel RL, Giaquinto AN, Jemal A. Cancer statistics, 2024. CA Cancer J Clin. 2024;74:12–49.
Thai AA, Solomon BJ, Sequist LV, Gainor JF, Heist RS. Lung cancer. Lancet. 2021;398:535–54.
Tang S, Qin C, Hu H, Liu T, He Y, Guo H, et al. Immune checkpoint inhibitors in non-small cell lung cancer: progress, challenges, and prospects. Cells. 2022;11:320.
Araki T, Kanda S, Horinouchi H, Ohe Y. Current treatment strategies for EGFR-mutated non-small cell lung cancer: from first line to beyond osimertinib resistance. Jpn J Clin Oncol. 2023;53:547–61.
Cerami E, Gao J, Dogrusoz U, Gross BE, Sumer SO, Aksoy BA, et al. The cBio cancer genomics portal: an open platform for exploring multidimensional cancer genomics data. Cancer Discov. 2012;2:401–4.
Cancer Genome Atlas Research N. Comprehensive molecular profiling of lung adenocarcinoma. Nature. 2014;511:543–50.
Hammerman PS, Lawrence MS, Voet D, Jing R, Cibulskis K, Sivachenko A, et al. Comprehensive genomic characterization of squamous cell lung cancers. Nature. 2012;489:519–25.
Zhang P, Singh A, Yegnasubramanian S, Esopi D, Kombairaju P, Bodas M, et al. Loss of Kelch-like ECH-associated protein 1 function in prostate cancer cells causes chemoresistance and radioresistance and promotes tumor growth. Mol Cancer Ther. 2010;9:336–46.
Guan L, Nambiar DK, Cao H, Viswanathan V, Kwok S, Hui AB, et al. NFE2L2 mutations enhance radioresistance in head and neck cancer by modulating intratumoral myeloid cells. Cancer Res. 2023;83:861–74.
Xu Q, Zhang P, Han X, Ren H, Yu W, Hao W, et al. Role of ionizing radiation activated NRF2 in lung cancer radioresistance. Int J Biol Macromol. 2023;241:124476.
Shao Y, Zhong DS, Guan SS. Histologic transformation of lung adenocarcinoma to squamous cell carcinoma after chemotherapy: two case reports. Transl Cancer Res. 2020;9:388–93.
Offin M, Chan JM, Tenet M, Rizvi HA, Shen R, Riely GJ, et al. Concurrent RB1 and TP53 alterations define a subset of egfr-mutant lung cancers at risk for histologic transformation and inferior clinical outcomes. J Thorac Oncol. 2019;14:1784–93.
Zou M, Toivanen R, Mitrofanova A, Floch N, Hayati S, Sun Y, et al. Transdifferentiation as a mechanism of treatment resistance in a mouse model of castration-resistant prostate cancer. Cancer Discov. 2017;7:736–49.
Bryan HK, Olayanju A, Goldring CE, Park BK. The Nrf2 cell defence pathway: Keap1-dependent and -independent mechanisms of regulation. Biochem Pharmacol. 2013;85:705–17.
Hammad A, Zheng ZH, Gao Y, Namani A, Shi HF, Tang X. Identification of novel Nrf2 target genes as prognostic biomarkers in colitis-associated colorectal cancer in Nrf2-deficient mice. Life Sci. 2019;238:116968.
Khor TO, Huang MT, Prawan A, Liu Y, Hao X, Yu S, et al. Increased susceptibility of Nrf2 knockout mice to colitis-associated colorectal cancer. Cancer Prev Res. 2008;1:187–91.
Blanpain C. Tracing the cellular origin of cancer. Nat Cell Biol. 2013;15:126–34.
Kobayashi A, Waku T. New addiction to the NRF2-related factor NRF3 in cancer cells: ubiquitin-independent proteolysis through the 20S proteasome. Cancer Sci. 2020;111:6–14.
Hamad SH, Sellers RS, Wamsley N, Zolkind P, Schrank TP, Major MB, et al. NRF2 activation in Trp53;p16-deficient mice drives oral squamous cell carcinoma. Cancer Res Commun. 2024;4:487–95.
Hamad SH, Montgomery SA, Simon JM, Bowman BM, Spainhower KB, Murphy RM, et al. TP53, CDKN2A/P16, and NFE2L2/NRF2 regulate the incidence of pure- and combined-small cell lung cancer in mice. Oncogene. 2022;41:3423–32.
Baird L, Yamamoto M. The molecular mechanisms regulating the KEAP1-NRF2 pathway. Mol Cell Biol. 2020;40:e00099-20
Gong M, Li Y, Ye X, Zhang L, Wang Z, Xu X, et al. Loss-of-function mutations in KEAP1 drive lung cancer progression via KEAP1/NRF2 pathway activation. Cell Commun Signal. 2020;18:98.
Kerins MJ, Ooi A. A catalogue of somatic NRF2 gain-of-function mutations in cancer. Sci Rep. 2018;8:12846.
Shibata T, Ohta T, Tong KI, Kokubu A, Odogawa R, Tsuta K, et al. Cancer related mutations in NRF2 impair its recognition by Keap1-Cul3 E3 ligase and promote malignancy. Proc Natl Acad Sci USA. 2008;105:13568–73.
Kansanen E, Kuosmanen SM, Leinonen H, Levonen AL. The Keap1-Nrf2 pathway: mechanisms of activation and dysregulation in cancer. Redox Biol. 2013;1:45–49.
Wei D, Goldfarb D, Song S, Cannon C, Yan F, Sakellariou-Thompson D, et al. SNF5/INI1 deficiency redefines chromatin remodeling complex composition during tumor development. Mol Cancer Res. 2014;12:1574–85.
Ahler E, Sullivan WJ, Cass A, Braas D, York AG, Bensinger SJ, et al. Doxycycline alters metabolism and proliferation of human cell lines. PLoS ONE. 2013;8:e64561.
Mulvaney KM, Matson JP, Siesser PF, Tamir TY, Goldfarb D, Jacobs TM, et al. Identification and characterization of MCM3 as a Kelch-like ECH-associated protein 1 (KEAP1) substrate. J Biol Chem. 2016;291:23719–33.
Reichard JF, Motz GT, Puga A. Heme oxygenase-1 induction by NRF2 requires inactivation of the transcriptional repressor BACH1. Nucleic Acids Res. 2007;35:7074–86.
Witherspoon JG, Hall JR, Jima D, Atkins HM, Wamsley NT, Major MB, et al. Mutant Nrf2(E79Q) enhances the promotion and progression of a subset of oncogenic Ras keratinocytes and skin tumors. Redox Biol. 2024;75:103261.
Takahashi N, Cho P, Selfors LM, Kuiken HJ, Kaul R, Fujiwara T, et al. 3D Culture models with crispr screens reveal hyperactive nrf2 as a prerequisite for spheroid formation via regulation of proliferation and ferroptosis. Mol Cell. 2020;80:828–44.
Badia-i-Mompel P, Vélez Santiago J, Braunger J, Geiss C, Dimitrov D, Müller-Dott S, et al. decoupleR: ensemble of computational methods to infer biological activities from omics data. Bioinform Adv. 2022;2:vbac016.
Singh A, Boldin-Adamsky S, Thimmulappa RK, Rath SK, Ashush H, Coulter J, et al. RNAi-mediated silencing of nuclear factor erythroid-2-related factor 2 gene expression in non-small cell lung cancer inhibits tumor growth and increases efficacy of chemotherapy. Cancer Res. 2008;68:7975–84.
Cescon DW, She D, Sakashita S, Zhu CQ, Pintilie M, Shepherd FA, et al. NRF2 pathway activation and adjuvant chemotherapy benefit in lung squamous cell carcinoma. Clin Cancer Res. 2015;21:2499–505.
Grossman RL, Heath AP, Ferretti V, Varmus HE, Lowy DR, Kibbe WA, et al. Toward a shared vision for cancer genomic data. N Engl J Med. 2016;375:1109–12.
Hou S, Zhou S, Qin Z, Yang L, Han X, Yao S, et al. Evidence, mechanism, and clinical relevance of the transdifferentiation from lung adenocarcinoma to squamous cell carcinoma. Am J Pathol. 2017;187:954–62.
Kang SM, Kang HJ, Shin JH, Kim H, Shin DH, Kim SK, et al. Identical epidermal growth factor receptor mutations in adenocarcinomatous and squamous cell carcinomatous components of adenosquamous carcinoma of the lung. Cancer. 2007;109:581–7.
Hofmann HS, Knolle J, Neef H. The adenosquamous lung carcinoma: clinical and pathological characteristics. J Cardiovasc Surg (Torino). 1994;35:543–7.
Haruki T, Nakanishi A, Matsui S, Kidokoro Y, Kubouchi Y, Takagi Y, et al. Transformation from adenocarcinoma to squamous cell carcinoma associated with long-term administration of EGFR-TKIs. Mol Clin Oncol. 2020;13:82.
Watanabe K, Kage H, Shinozaki-Ushiku A, Kohsaka S, Takai D, Nakajima J, et al. Spontaneous transdifferentiation from small cell lung carcinoma to squamous cell carcinoma. J Thorac Oncol. 2019;14:e31–34.
Handorf CR, Kulkarni A, Grenert JP, Weiss LM, Rogers WM, Kim OS, et al. A multicenter study directly comparing the diagnostic accuracy of gene expression profiling and immunohistochemistry for primary site identification in metastatic tumors. Am J Surg Pathol. 2013;37:1067–75.
Vassella E, Langsch S, Dettmer MS, Schlup C, Neuenschwander M, Frattini M, et al. Molecular profiling of lung adenosquamous carcinoma: hybrid or genuine type? Oncotarget. 2015;6:23905–16.
Collisson EA, Campbell JD, Brooks AN, Berger AH, Lee W, Chmielecki J, et al. Comprehensive molecular profiling of lung adenocarcinoma. Nature. 2014;511:543–50.
Szulzewsky F, Holland EC, Vasioukhin V. YAP1 and its fusion proteins in cancer initiation, progression and therapeutic resistance. Dev Biol. 2021;475:205–21.
Dhanasekaran R, Deutzmann A, Mahauad-Fernandez WD, Hansen AS, Gouw AM, Felsher DW. The MYC oncogene - the grand orchestrator of cancer growth and immune evasion. Nat Rev Clin Oncol. 2022;19:23–36.
Ciamporcero E, Daga M, Pizzimenti S, Roetto A, Dianzani C, Compagnone A, et al. Crosstalk between Nrf2 and YAP contributes to maintaining the antioxidant potential and chemoresistance in bladder cancer. Free Radic Biol Med. 2018;115:447–57.
Wallbillich NJ, Lu H. Role of c-Myc in lung cancer: progress, challenges, and prospects. Chin Med J Pulm Crit Care Med. 2023;1:129–38.
Eftekhari Kenzerki M, Ahmadi M, Mousavi P, Ghafouri-Fard S. MYC and non-small cell lung cancer: a comprehensive review. Hum Gene. 2023;37:201185.
von Eyss B, Jaenicke LA, Kortlever RM, Royla N, Wiese KE, Letschert S, et al. A MYC-driven change in mitochondrial dynamics limits YAP/TAZ function in mammary epithelial cells and breast cancer. Cancer Cell. 2015;28:743–57.
Li X, Tao J, Cigliano A, Sini M, Calderaro J, Azoulay D, et al. Co-activation of PIK3CA and Yap promotes development of hepatocellular and cholangiocellular tumors in mouse and human liver. Oncotarget. 2015;6:10102–15.
Uitdehaag JC, de Roos JA, van Doornmalen AM, Prinsen MB, Spijkers-Hagelstein JA, de Vetter JR, et al. Selective targeting of CTNBB1-, KRAS- or MYC-driven cell growth by combinations of existing drugs. PLoS ONE. 2015;10:e0125021.
Abduch RH, Carolina Bueno A, Leal LF, Cavalcanti MM, Gomes DC, Brandalise SR, et al. Unraveling the expression of the oncogene YAP1, a Wnt/beta-catenin target, in adrenocortical tumors and its association with poor outcome in pediatric patients. Oncotarget. 2016;7:84634–44.
George J, Lim JS, Jang SJ, Cun Y, Ozretić L, Kong G, et al. Comprehensive genomic profiles of small cell lung cancer. Nature. 2015;524:47–53.
Levings DC, Wang X, Kohlhase D, Bell DA, Slattery M. A distinct class of antioxidant response elements is consistently activated in tumors with NRF2 mutations. Redox Biol. 2018;19:235–49.
Takahashi J, Suzuki T, Sato M, Nitta S, Yaguchi N, Muta T, et al. Differential squamous cell fates elicited by NRF2 gain of function versus KEAP1 loss of function. Cell Rep. 2024;43:114104.
Sequist LV, Waltman BA, Dias-Santagata D, Digumarthy S, Turke AB, Fidias P, et al. Genotypic and histological evolution of lung cancers acquiring resistance to EGFR inhibitors. Sci Transl Med. 2011;3:75ra26.
Jiang M, Zhu X, Han X, Jing H, Han T, Li Q, et al. Histologic transformation of non-small-cell lung cancer in brain metastases. Int J Clin Oncol. 2019;24:375–84.
Scher KS, Saldivar JS, Fishbein M, Marchevsky A, Reckamp KL. EGFR-mutated lung cancer with T790M-acquired resistance in the brain and histologic transformation in the lung. J Natl Compr Canc Netw. 2013;11:1040–4.
Park S, Han J, Sun JM. Histologic transformation of ALK-rearranged adenocarcinoma to squamous cell carcinoma after treatment with ALK inhibitor. Lung Cancer. 2019;127:66–8.
Hsu CL, Chen KY, Kuo SW, Chang YL. Histologic transformation in a patient with lung cancer treated with chemotherapy and pembrolizumab. J Thorac Oncol. 2017;12:e75–6.
Xu K, Ma H, McCown TJ, Verma IM, Kafri T. Generation of a stable cell line producing high-titer self-inactivating lentiviral vectors. Mol Ther. 2001;3:97–104.
Schöchlin M, Weissinger SE, Brandes AR, Herrmann M, Möller P, Lennerz JK. A nuclear circularity-based classifier for diagnostic distinction of desmoplastic from spindle cell melanoma in digitized histological images. J Pathol Inf. 2014;5:40.
Iyer S, Zhang S, Yucel S, Horn H, Smith SG, Reinhardt F, et al. Genetically defined syngeneic mouse models of ovarian cancer as tools for the discovery of combination immunotherapy. Cancer Discov. 2021;11:384–407.
Lockworth CR, Kim SJ, Liu J, Palla SL, Craig SL. Effect of enrichment devices on aggression in manipulated nude mice. J Am Assoc Lab Anim Sci. 2015;54:731–6.
Yamano S, Gi M, Tago Y, Doi K, Okada S, Hirayama Y, et al. Role of deltaNp63(pos)CD44v(pos) cells in the development of N-nitroso-tris-chloroethylurea-induced peripheral-type mouse lung squamous cell carcinomas. Cancer Sci. 2016;107:123–32.
Mootha VK, Lindgren CM, Eriksson KF, Subramanian A, Sihag S, Lehar J, et al. PGC-1alpha-responsive genes involved in oxidative phosphorylation are coordinately downregulated in human diabetes. Nat Genet. 2003;34:267–73.
Rekhtman N, Pietanza MC, Hellmann MD, Naidoo J, Arora A, Won H, et al. Next-generation sequencing of pulmonary large cell neuroendocrine carcinoma reveals small cell carcinoma-like and non-small cell carcinoma-like subsets. Clin Cancer Res. 2016;22:3618–29.
Schwendenwein A, Megyesfalvi Z, Barany N, Valko Z, Bugyik E, Lang C, et al. Molecular profiles of small cell lung cancer subtypes: therapeutic implications. Mol Ther Oncolytics. 2021;20:470–83.
Rudin CM, Poirier JT, Byers LA, Dive C, Dowlati A, George J, et al. Molecular subtypes of small cell lung cancer: a synthesis of human and mouse model data. Nat Rev Cancer. 2019;19:289–97.
Garcia-Alonso L, Holland CH, Ibrahim MM, Turei D, Saez-Rodriguez J. Benchmark and integration of resources for the estimation of human transcription factor activities. Genome Res. 2019;29:1363–75.
Acknowledgements
The authors thank Charlene Santos and her staff at the UNC-LCCC Animal Studies Core for the monitoring, weighing, and isolation of tumors of the mice; Dawud Hilliard, Gabriela Cruz, and the staff at the UNC-LCCC Pathology Services Core Facility for tissue processing and staining for H&E and IHC, and Dr. Pablo Ariel and his staff at the UNC Microscopy Services Laboratory for their help with the microscopy studies. This study was supported by a grant from the National Institutes of Environmental and Health Sciences (T32ES007126) SHH, a Goldberg Postdoctoral Fellow (SHH), and a grant from the National Cancer Institute (CA216051) (BEW and MBM). These studies were also supported in part by the Surgery Department at Cooper University Health Care, Cooper Medical School of Rowan University, and MD Anderson Cancer Center at Cooper.
Author information
Authors and Affiliations
Contributions
SHH: Conception, study supervision, experimental design, methodology, data analysis, interpretation of data, and writing, review, and revision of the manuscript; HJ: Conducting experiments, and help in writing, review and revision of the manuscript; TH: Conducting experiments, and help in writing, review and revision of the manuscript; SRJ: Data analysis, bioinformatics, and help in writing, review and revision of the manuscript; ZS: Conducting experiments, and help in review and revision of the manuscript; NB: Statistical analysis, and help in writing, review and revision of the manuscript; RS: Methodology, data analysis and interpretation, and help in review and revision of the manuscript; GZ: Analysis and interpretation of data, and help in review and revision of the manuscript; TS: Interpretation of data, and help in review and revision of the manuscript; RTM: Conducting experiments, and help in review and revision of the manuscript; DC: Bioinformatics, and help in review and revision of the manuscript; JMS: Bioinformatics, and help in review and revision of the manuscript; FRS: Study supervision, interpretation of data, and help in review and revision of the manuscript; DS: Study supervision, interpretation of data, and help in review and revision of the manuscript; MBM: Study supervision, interpretation of data, and help in writing, review, and revision of the manuscript; BEW: Conception, study supervision, experimental design, methodology, data analysis, interpretation of data, and help in writing, review, and revision of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
Animal studies were conducted following an approved protocol (#19-242.0) established by Institutional Animal Care and Use Committee (IACUC) at the University of North Carolina at Chapel Hill, in accordance with the guidelines of the Animal Welfare Act and the Guide for the Care and Use of Laboratory Animals.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hamad, S.H., Joshi, H., Hess, T. et al. Activating NRF2E79Q mutation alters the differentiation of human non-small cell lung cancer. Cancer Gene Ther 32, 1428–1438 (2025). https://doi.org/10.1038/s41417-025-00966-w
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41417-025-00966-w