Abstract
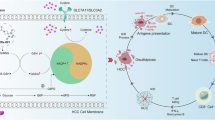
Disulfidptosis, a recently identified form of disulfide-dependent cell death, has emerged as a key regulator of cancer progression and therapy resistance. This review examines the molecular mechanisms connecting the redox balance within the tumor microenvironment (TME), outlines its crosstalk with traditional cell death pathways, and highlights its promise as a therapeutic strategy. Current evidence indicates that disulfidptosis is driven by glutathione (GSH) depletion and thiol-disulfide exchange imbalance, leading to endoplasmic reticulum stress and tumor cell vulnerability. Translational applications—including small-molecule activators and combination therapies—are discussed, alongside challenges in clinical translation and future research directions.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Zhang R, Kang R, Tang D. Reductive cell death: the other side of the coin. Cancer Gene Ther. 2023;30:929–31.
Liu X, Nie L, Zhang Y, Yan Y, Wang C, Colic M, et al. Actin cytoskeleton vulnerability to disulfide stress mediates disulfidptosis. Nat Cell Biol. 2023;25:404–14.
Liu X, Zhuang L, Gan B. Disulfidptosis: disulfide stress-induced cell death. Trends Cell Biol. 2024;34:327–37.
Xie J, Deng X, Xie Y, Zhu H, Liu P, Deng W, et al. Multi-omics analysis of disulfidptosis regulators and therapeutic potential reveals glycogen synthase 1 as a disulfidptosis triggering target for triple-negative breast cancer. MedComm. 2024;5:e502.
Wan J, Shi JH, Shi M, Huang H, Zhang Z, Li W, et al. Lactate dehydrogenase B facilitates disulfidptosis and exhaustion of tumour-infiltrating CD8(+) T cells. Nat Cell Biol. 2025;27:972–82.
Shi M, Li X, Guo Y, Zhang Y, Xu J, Yan L, et al. Gaudichaudione H enhances the sensitivity of hepatocellular carcinoma cells to disulfidptosis via regulating NRF2-SLC7A11 signaling pathway. Adv Sci. 2025;12:e2411131.
Wang J, Chen J, Fan K, Wang M, Gao M, Ren Y, et al. Inhibition of endoplasmic reticulum stress cooperates with SLC7A11 to promote disulfidptosis and suppress tumor growth upon glucose limitation. Adv Sci. 2025;12:e2408789.
Hu H, Hua S, Lu F, Zhang W, Zhang Z, Cui J, et al. Mucous permeable nanoparticle for inducing cuproptosis-like death in broad-spectrum bacteria for nebulized treatment of acute pneumonia. Adv Sci. 2025;12:e2408580.
Hwang SM, Chang S, Rodriguez PC, Cubillos-Ruiz JR. Endoplasmic reticulum stress responses in anticancer immunity. Nat Rev Cancer. 2025;25:684–702.
Pagliaro L, Marchesini M, Roti G. Targeting oncogenic Notch signaling with SERCA inhibitors. J Hematol Oncol. 2021;14:8.
Lehmann BD, Pietenpol JA. Targeting mutant p53 in human tumors. J Clin Oncol. 2012;30:3648–50.
Liang HY, Luo RZ, Deng R, Chen SL, Liu X, Yang X, et al. Glycogen stores mediated by the p53-GYS1 feedback circuit engenders platinum resistance in ovarian clear cell carcinoma. Cell Death Differ. 2025;32:1707–21.
Zhang M, Zheng H, Zhu X, Liu S, Jin H, Chen Y, et al. Synchronously evoking disulfidptosis and ferroptosis via systematical glucose deprivation targeting SLC7A11/GSH/GPX4 antioxidant axis. ACS Nano. 2025;19:14233–48.
Zhu Y, Wang X, Feng L, Zhao R, Yu C, Liu Y, et al. Intermetallics triggering pyroptosis and disulfidptosis in cancer cells promote anti-tumor immunity. Nat Commun. 2024;15:8696.
Zheng Z, Song Y. Integrated analysis of disulfidptosis-related genes SLC7A11, SLC3A2, RPN1 and NCKAP1 across cancers. Discov Oncol. 2024;15:724.
Wang B, Wang W, Wang Y, Wen X, Wang Z, Leng H, et al. Analysis and experimental validation of disulfidptosis related genes solute carrier family 3 member 2 (SLC3A2) in endometrial cancer. Cancer Cell Int. 2024;24:390.
Zhang X, Lin Y, Shi L, Zhai A, Wu C, Zhu QY. Disulfidptosis-related gene SLC3A2: a novel prognostic biomarker in nasopharyngeal carcinoma and head and neck squamous cell carcinoma. Front Oncol. 2025;15:1451034.
Zong Y, Zhu A, Liu P, Fu P, Li Y, Chen S, et al. Pan-cancer analysis of the disulfidptosis-related gene RPN1 and its potential biological function and prognostic significance in gliomas. Heliyon. 2024;10:e31875.
Zhou Q, Song B, He Y, Zhang Z, Chen S, Chen W, et al. Identification of a disulfidptosis-related genes signature for diagnostic and immune infiltration characteristics in cervical cancer. PLoS One. 2025;20:e0322387.
Wang Y, Tsukamoto Y, Hori M, Iha H. Disulfidptosis: a novel prognostic criterion and potential treatment strategy for diffuse large B-cell lymphoma (DLBCL). Int J Mol Sci. 2024;25:7156.
Che C, Song D, Xue P, Yin X. A novel disulfidptosis-related risk signature for prognostic prediction in patients with ewing sarcoma. J Orthop Res. 2025;43:790–802.
Han L, Li Y, Yu Y, Liu G, Gao X, Wang F, et al. Integrated analysis and experiments uncover the function of disulfidptosis in predicting immunotherapy effectiveness and delineating immune landscapes in uterine corpus endometrial carcinoma. Front Immunol. 2024;15:1454730.
Hu G, Yao H, Wei Z, Li L, Yu Z, Li J, et al. A bioinformatics approach to identify a disulfidptosis-related gene signature for prognostic implication in colon adenocarcinoma. Sci Rep. 2023;13:12403.
Zhang X, Hong B, Li H, Sun Z, Zhao J, Li M, et al. Disulfidptosis and ferroptosis related genes define the immune microenvironment and NUBPL serves as a potential biomarker for predicting prognosis and immunotherapy response in bladder cancer. Heliyon. 2024;10:e37638.
Liu L, Zhang F, Yu J, Dong P, Deng J, Liu W, et al. Multidisciplinary analysis of the prognosis and biological function of NUBPL in gastric cancer. Front Immunol. 2025;16:1603898.
Sun Y, Wang R, Li X, Zhou W, Huang H, Zhou Y, et al. The involvement of lncRNA EMSLR in the disulfidptosis and progression of endometrial carcinoma. Cancer Gene Ther. 2025;32:1107–19. https://doi.org/10.1038/s41417-025-00918-4.
Author information
Authors and Affiliations
Contributions
Writing-original draft: XD, CZ, JX, BT, XT and YZ; Writing-review & editing: XD and YZ. All authors contributed to the article and approved the submitted version.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Deng, X., Zhang, C., Xie, J. et al. Disulfidptosis in cancer: from redox stress to therapeutic strategy. Cancer Gene Ther 33, 22–25 (2026). https://doi.org/10.1038/s41417-025-00985-7
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41417-025-00985-7