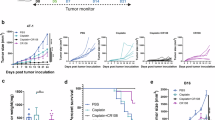
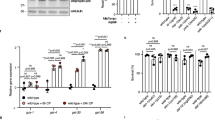
Abstract
Cisplatin resistance causes ineffectiveness of cisplatin-based treatment for cervical carcinoma. The combination of cisplatin and other chemotherapeutic drugs is an available strategy to overcome this problem. However, chemotherapeutic drugs combined with cisplatin may show tissue toxicity and systemic side effects. Thus, there is a great need of seeking effective substitutes for these chemotherapeutic drugs to improve combination therapy. Here, we found that inactivating IL-6/JAK2/STAT3 signaling pathway sensitized carcinoma cells to cisplatin toxicity by increasing cisplatin accumulation, impairing DNA damage repair, and inhibiting the initiation and development of autophagy, which subsequently caused the increases in DNA damage levels and apoptosis rates in cisplatin-treated cells. We predicted that TFF3 negatively regulated transduction in the IL-6/JAK2/STAT3 pathway based on in silico analysis of the differentially expressed genes (DEGs) between highly trefoil factor 3(TFF3)-encoding mRNA-expressing carcinoma tissues and low-expressing counterparts, and experimentally determined that both ectopic expression of TFF3-encoding gene and TFF3 administration inhibited IL-6-induced STAT3 activation in carcinoma cells. Mechanistically, upon binding to IGF2R, TFF3 stabilized IGF2R by inhibiting the ubiquitin-proteasome degradation pathway to inactivate Akt and thereby STAT3. Moreover, we discovered that TFF3 administration antagonized protective effects of IL-6 stimulation against tumor-killing capacity of cisplatin. Based on these findings, we consider that TFF3 may be employed as a cisplatin sensitizer and have advantages over traditional chemotherapeutic drugs in cisplatin-based combination therapy, since it is a naturally occurring protein in cervical tissue.

This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
All relevant data generated in this study are available within the article and supplementary files. The original data can be requested from the corresponding author for reasonable purposes. The bioinformatics analysis data were obtained from the TCGA (https://www.cancer.gov/) and GEO (https://www.ncbi.nlm.nih.gov/geo/).
Code availability
The “limma” package was employed to determine the differentially expressed genes. The “clusterProfiler” and “enrichplot” packages were used to perform gene enrichement analysis. The expression levels of TFF3 and XRCC4-encoding mRNAs were obtained from GSE7410, GSE39001 and GSE3514. And the “GEOquery” and “ggplot2” packages were separately exploited to group data and to conduct boxplot analysis that provides insightful visualization.
References
Vu M, Yu J, Awolude OA, Chuang L. Cervical cancer worldwide. Curr Probl Cancer. 2018;42:457–65.
Brooke G, Wendel S, Banerjee A, Wallace N. Opportunities to advance cervical cancer prevention and care. Tumour Virus Res. 2024;18:200292.
Włoszek E, Krupa K, Skrok E, Budzik MP, Deptała A, Badowska-Kozakiewicz A. HPV and cervical cancer-biology, prevention, and treatment updates. Curr Oncology 2025;32:122.
Ghosh S. Cisplatin: the first metal based anticancer drug. Bioorg Chem. 2019;88:102925.
Dasari S, Tchounwou PB. Cisplatin in cancer therapy: molecular mechanisms of action. Eur J Pharm. 2014;740:364–78.
Achkar IW, Abdulrahman N, Al-Sulaiti H, Joseph JM, Uddin S, Mraiche F. Cisplatin based therapy: the role of the mitogen activated protein kinase signaling pathway. J Transl Med. 2018;16:96.
Trimmer EE, Essigmann JM. Cisplatin. Essays Biochem. 1999;34:191–211.
Lorusso D, Petrelli F, Coinu A, Raspagliesi F, Barni S. A systematic review comparing cisplatin and carboplatin plus paclitaxel-based chemotherapy for recurrent or metastatic cervical cancer. Gynecol Oncol. 2014;133:117–23.
Rottenberg S, Disler C, Perego P. The rediscovery of platinum-based cancer therapy. Nat Rev Cancer. 2021;21:37–50.
Zhang J, Ye Z, Tew KD, Townsend DM. Cisplatin chemotherapy and renal function. Adv Cancer Res. 2021;152:305–27.
Wang X, Zhou Y, Wang D, Wang Y, Zhou Z, Ma X, et al. Cisplatin-induced ototoxicity: from signaling network to therapeutic targets. Biomed Pharmacother Biomed Pharmacother. 2023;157:114045.
Zhu H, Luo H, Zhang W, Shen Z, Hu X, Zhu X. Molecular mechanisms of cisplatin resistance in cervical cancer. Drug Des, Dev Ther. 2016;10:1885–95.
Galluzzi L, Senovilla L, Vitale I, Michels J, Martins I, Kepp O, et al. Molecular mechanisms of cisplatin resistance. Oncogene. 2012;31:1869–83.
Thim L, May FEB. Structure of mammalian trefoil factors and functional insights. Cellular Mol Life Sci CMLS. 2005;62:2956–73.
Wong WM, Poulsom R, Wright NA. Trefoil peptides. Gut. 1999;44:890–95.
Kjellev S. The trefoil factor family - small peptides with multiple functionalities. Cellular Mol Life Sci CMLS. 2009;66:1350–69.
Belovari T, Bijelić N, Tolušić Levak M, Baus Lončar M. Trefoil factor family peptides TFF1 and TFF3 in the nervous tissues of developing mouse embryo. Bosn J Basic Med Sci. 2015;15:33–37.
Braga Emidio N, Hoffmann W, Brierley SM, Muttenthaler M. Trefoil factor family: unresolved questions and clinical perspectives. Trends Biochem Sci. 2019;44:387–90.
Muskett FW, May FEB, Westley BR, Feeney J. Solution structure of the disulfide-linked dimer of human intestinal trefoil factor (TFF3): the intermolecular orientation and interactions are markedly different from those of other dimeric trefoil proteins. Biochemistry. 2003;42:15139–47.
Yang Y, Lin Z, Lin Q, Bei W, Guo J. Pathological and therapeutic roles of bioactive peptide trefoil factor 3 in diverse diseases: recent progress and perspective. Cell Death Dis. 2022;13:62.
Buda A, Jepson MA, Pignatelli M. Regulatory function of trefoil peptides (TFF) on intestinal cell junctional complexes. Cell Commun Adhes. 2012;19:63–68.
Aamann L, Vestergaard EM, Grønbæk H. Trefoil factors in inflammatory bowel disease. World J Gastroenterol. 2014;20:3223–30.
Cui H, Wang S, Song F, Cheng X, Nan G, Zhao Y, et al. CD147 receptor is essential for TFF3-mediated signaling, regulating colorectal cancer progression. Signal Transduct Target Ther. 2021;6:268.
Yang L, Zhang X, Zhang J, Liu Y, Ji T, Mou J, et al. Low expression of TFF3 in papillary thyroid carcinoma may correlate with poor prognosis but high immune cell infiltration. Future Oncol. 2022;18:333–48.
Belle NM, Ji Y, Herbine K, Wei Y, Park J, Zullo K, et al. TFF3 interacts with LINGO2 to regulate EGFR activation for protection against colitis and gastrointestinal helminths. Nat Commun. 2019;10:4408.
Yang T, Fu X, Tian R, Cui H, Li L, Han J, et al. TFF3 promotes clonogenic survival of colorectal cancer cells through upregulation of EP4 via activation of STAT3. Transl Cancer Res. 2023;12:1503–15.
You M, Chen Y, Chong Q, Wu M, Pandey V, Chen R, et al. Trefoil factor 3 mediation of oncogenicity and chemoresistance in hepatocellular. Oncotarget. 2017;8:39323–44.
Große-Kreul J, Busch M, Winter C, Pikos S, Stephan H, Dünker N. Forced trefoil factor family peptide 3 (TFF3) expression reduces growth, viability, and tumorigenicity of human retinoblastoma cell lines. PLoS ONE. 2016;11:e0163025.
You M, Chen Y, Chong Q, Wu M, Pandey V, Chen R, et al. Trefoil factor 3 mediation of oncogenicity and chemoresistance in hepatocellular carcinoma is AKT-BCL-2 dependent. Oncotarget. 2017;8:39323–44.
Takano T, Yamada H. Trefoil factor 3 (TFF3): a promising indicator for diagnosing thyroid follicular carcinoma. Endocr J. 2009;56:9–16.
Maděrka M, Dvořák V, Hambálek J, Stejskal D, Krejčí ŠM, Langová K, et al. Serum concentrations of S100-A11 and AIF-1 are elevated in cervical cancer patients with lymph node involvement. Ceska Gynekol. 2021;86:17–21.
Houben T, Harder S, Schlüter H, Kalbacher H, Hoffmann W. Different forms of TFF3 in the human saliva: heterodimerization with IgG fc binding protein (FCGBP). Int J Mol Sci. 2019;20:5000.
Karimi-Zarchi M, Abbasi H, Javaheri A, Hadadan A, Meibodi B, Tabatabaei RS, et al. Association of IL-12b rs3212227 and IL-6 rs1800795 polymorphisms with susceptibility to cervical cancer: a systematic review and meta-analysis. Asian Pac J Cancer Prev APJCP. 2020;21:1197–206.
Cai C, Peng X, Zhang Y. Serum IL-6 level predicts the prognosis and diagnosis in cervical cancer patients. Int J Womens Health. 2022;14:655–63.
Konishi H, Okamoto K, Ohmori Y, Yoshino H, Ohmori H, Ashihara M, et al. An orally available, small-molecule interferon inhibits viral replication. Sci Rep. 2012;2:259.
Ghandadi M, Sahebkar A. Interleukin-6: a critical cytokine in cancer multidrug resistance. Curr Pharm Des. 2016;22:518–26.
Galluzzi L, Vitale I, Michels J, Brenner C, Szabadkai G, Harel-Bellan A, et al. Systems biology of cisplatin resistance: past, present, and future. Cell Death Dis. 2014;5:e1257.
Jiang M, Jia K, Wang L, Li W, Chen B, Liu Y, et al. Alterations of DNA damage response pathway: biomarker and therapeutic strategy for cancer immunotherapy. Acta Pharm Sin B. 2021;11:2983–94.
Chang HHY, Pannunzio NR, Adachi N, Lieber MR. Non-homologous DNA end joining and alternative pathways to double-strand break repair. Nat Rev Mol Cell Biol. 2017;18:495–506.
Zhang Y, Guo R, Wang S, Jiang X, Cui H, Guo Y, et al. Autophagy-related proteins in genome stability: autophagy-dependent and independent actions. Int J Biol Sci. 2022;18:5329–44.
Cheng SM, Shieh M, Lin T, Cheung CHA. The “dark side” of autophagy on the maintenance of genome stability: does it really exist during excessive activation?. J Cell Physiol. 2022;237:178–88.
Miller S, Oleksy A, Perisic O, Williams RL. Finding a fitting shoe for Cinderella: searching for an autophagy inhibitor. Autophagy. 2010;6:805–07.
Lokau J, Garbers C. Activating mutations of the gp130/JAK/STAT pathway in human diseases. Adv Protein Chem Struct Biol. 2019;116:283–309.
Wang X, Lin L, Lan B, Wang Y, Du L, Chen X, et al. IGF2r-initiated proton rechanneling dictates an anti-inflammatory property in macrophages. Sci Adv. 2020;6:eabb7389.
Han YH, Moon HJ, You BR, Park WH. The effect of MG132, a proteasome inhibitor, on HeLa cells in relation to cell growth, reactive oxygen species, and GSH. Oncol Rep. 2009;22:215–21.
Schneider-Poetsch T, Ju J, Eyler DE, Dang Y, Bhat S, Merrick WC, et al. Inhibition of eukaryotic translation elongation by cycloheximide and lactimidomycin. Nat Chem Biol. 2010;6:209–17.
Nandi D, Tahiliani P, Kumar A, Chandu D. The ubiquitin-proteasome system. J Biosci. 2006;31:137–55.
Swatek KN, Komander D. Ubiquitin modifications. Cell Res. 2016;26:399–422.
Md Mokhtar AH, Malik IA, Abd Aziz NAA, Almabhouh FA, Durairajanayagam D, Singh HJ. LY294002, a PI3k pathway inhibitor, prevents leptin-induced adverse effects on spermatozoa in Sprague-Dawley rats. Andrologia. 2019;51:e13196.
Zhao Y, Tian Y, Zhang J, Xu F, Yang Y, Huang Y, et al. Effects of an oral allosteric AKT inhibitor (MK-2206) on human nasopharyngeal cancer in vitro and in vivo. Drug Des Dev Ther. 2014;8:1827–37.
Ferrajoli A, Faderl S, Van Q, Koch P, Harris D, Liu Z, et al. WP1066 disrupts Janus kinase-2 and induces caspase-dependent apoptosis in acute myelogenous leukemia cells. Cancer Res. 2007;67:11291–99.
McMurray JS. A new small-molecule STAT3 inhibitor. Chem Biol. 2006;13:1123–24.
Schindler S, Magalhães RA, Costa ELF, Brites C. Genital cytokines in HIV/human papillomavirus co-infection: a systematic review. AIDS Res Hum Retrovir. 2022;38:683–91.
Liu H, Lyu D, Zhang Y, Sheng L, Tang N. Association between the IL-6 rs1800795 polymorphism and the risk of cervical cancer: a meta-analysis of 1210 cases and 1525 controls. Technol Cancer Res Treat. 2017;16:662–67.
Hu F, Song D, Yan Y, Huang C, Shen C, Lan J, et al. IL-6 regulates autophagy and chemotherapy resistance by promoting BECN1 phosphorylation. Nat Commun. 2021;12:3651.
Kumari N, Dwarakanath BS, Das A, Bhatt AN. Role of interleukin-6 in cancer progression and therapeutic resistance. Tumour Biol J Int Soc Oncodev Biol Med. 2016;37:11553–72.
Wei LH, Kuo ML, Chen CA, Chou CH, Cheng WF, Chang MC, et al. The anti-apoptotic role of interleukin-6 in human cervical cancer is mediated by up-regulation of MCL-1 through a PI 3-k/akt pathway. Oncogene. 2001;20:5799–809.
Das R, Bhattacharya K, Samanta SK, Pal BC, Mandal C. Improved chemosensitivity in cervical cancer to cisplatin: synergistic activity of mahanine through STAT3 inhibition. Cancer Lett. 2014;351:81–90.
Vázquez-Mellado MJ, Cortés-Ballinas LG, Blanco-Flores IC, Aguilar C, Vázquez-Gómez G, Rocha-Zavaleta L. Erythropoietin promotes expression of survivin via STAT3 activation and reduces sensitivity to cisplatin in cervical cancer cells. Oncol Rep. 2019;41:1333–41.
Belotte J, Fletcher NM, Awonuga AO, Alexis M, Abu-Soud HM, Saed MG, et al. The role of oxidative stress in the development of cisplatin resistance in epithelial ovarian cancer. Reprod Sci. 2014;21:503–08.
Bravo-Cuellar A, Ortiz-Lazareno PC, Sierra-Díaz E, Solorzano-Ibarra F, Méndez-Clemente AS, Aguilar-Lemarroy A, et al. Pentoxifylline sensitizes cisplatin-resistant human cervical cancer cells to cisplatin treatment: involvement of mitochondrial and NF-kappa b pathways. Front Oncol. 2020;10:592706.
Goto S, Yoshida K, Morikawa T, Urata Y, Suzuki K, Kondo T. Augmentation of transport for cisplatin-glutathione adduct in cisplatin-resistant cancer cells. Cancer Res. 1995;55:4297–301.
Ceccaldi R, Rondinelli B, D’Andrea AD. Repair pathway choices and consequences at the double-strand break. Trends Cell Biol. 2016;26:52–64.
Symington LS, Gautier J. Double-strand break end resection and repair pathway choice. Annu Rev Genet. 2011;45:247–71.
Yang S, Wang XQ. XLF-mediated NHEJ activity in hepatocellular carcinoma therapy resistance. BMC Cancer. 2017;17:344.
Zheng S, Tian Q, Yuan Y, Sun S, Li T, Xia R, et al. Extracellular vesicle-packaged circBIRC6 from cancer-associated fibroblasts induce platinum resistance via SUMOylation modulation in pancreatic cancer. J Exp Clin Cancer Res CR. 2023;42:324.
Mi L, Cai Y, Qi J, Chen L, Li Y, Zhang S, et al. Elevated nonhomologous end-joining by AATF enables efficient DNA damage repair and therapeutic resistance in glioblastoma. Nat Commun. 2025;16:4941.
Yang J, Chatterjee-Kishore M, Staugaitis SM, Nguyen H, Schlessinger K, Levy DE, et al. Novel roles of unphosphorylated STAT3 in oncogenesis and transcriptional regulation. Cancer Res. 2005;65:939–47.
Shi S, Ma H, Zhang Z. Clinicopathological and prognostic value of STAT3/p-STAT3 in cervical cancer: a meta and bioinformatics analysis. Pathol Res Pract. 2021;227:153624.
Wu L, Shen B, Li J, Zhang H, Zhang K, Yang Y, et al. STAT3 exerts pro-tumor and anti-autophagy roles in cervical cancer. Diagn Pathol. 2022;17:13.
Rolén U, Kobzeva V, Gasparjan N, Ovaa H, Winberg G, Kisseljov F, et al. Activity profiling of deubiquitinating enzymes in cervical carcinoma biopsies and cell lines. Mol Carcinog. 2006;45:260–69.
Meng Y, Hong C, Yang S, Qin Z, Yang L, Huang Y. Roles of USP9x in cellular functions and tumorigenesis (review). Oncol Lett. 2023;26:506.
Yuan Z, Chen D, Chen X, Yang H, Wei Y. Overexpression of trefoil factor 3 (TFF3) contributes to the malignant progression in cervical cancer cells. Cancer Cell Int. 2017;17:7.
Ara T, Nakata R, Sheard MA, Shimada H, Buettner R, Groshen SG, et al. Critical role of STAT3 in IL-6-mediated drug resistance in human neuroblastoma. Cancer Res. 2013;73:3852–64.
Liang Q, Gong M, Zou J, Luo M, Jiang L, Wang C, et al. A phosphoglycerate mutase 1 allosteric inhibitor overcomes drug resistance to EGFR-targeted therapy via disrupting IL-6/JAK2/STAT3 signaling pathway in lung adenocarcinoma. Drug Resist Updates Rev Comment Antimicrob Anticancer Chemother. 2023;68:100957.
Bubner B, Baldwin IT. Use of real-time PCR for determining copy number and zygosity in transgenic plants. Plant Cell Rep. 2004;23:263–71.
Chou TC, Talalay P. Quantitative analysis of dose-effect relationships: the combined effects of multiple drugs or enzyme inhibitors. Adv Enzyme Regul. 1984:22:27–55.
Olive PL, Banáth JP. The comet assay: a method to measure DNA damage in individual cells. Nat Protoc. 2006;1:23–29.
Chaubey RC. Computerized image analysis software for the comet assay. Methods Mol Biol. 2005;291:97–106.5.
Acknowledgements
We thank Professor Tie Wang (Institute of Intelligent Detection for Life and Health, Tianjin University of Technology) for providing the experimental platform and instruments for this research. This work was supported by the National Natural Science Foundation of China (grant number 81602403) and Tianjin Natural Science Foundation (No.23JCQNJC00790).
Author information
Authors and Affiliations
Contributions
YXZ and LY designed the study; YXZ performed the bioinformatics experiments; YXZ and LPY performed all the experiments; NA and BHF performed the preparation of the recombinant protein TFF3; WZ and LSJ cultured the human cervical cancer cell line; YXZ and LPY performed the data processing and visualization; YXZ and LY wrote and revised the manuscript; and JYG completed the construction of the plasmid.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, Y., Yan, L., An, N. et al. TFF3 sensitizes cervical carcinoma cells to cisplatin toxicity by binding to IGF2R. Cancer Gene Ther 33, 155–168 (2026). https://doi.org/10.1038/s41417-025-00990-w
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41417-025-00990-w