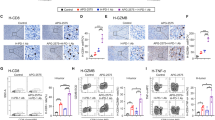
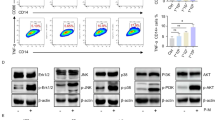
Abstract
Immune checkpoint inhibitors (ICIs) targeting the PD-1/PD-L1 axis have revolutionized cancer therapy, yet primary and acquired resistance remain major clinical obstacles. Dysregulated angiogenesis fuels the development of an immunosuppressive tumor microenvironment, while crosstalk between immunity and angiogenesis further propels tumor immune evasion and treatment resistance. The present study aimed to establish a penpulimab-resistant model, delineate anti-PD-1 resistance traits via single-cell RNA sequencing, and unravel the precise mechanisms through which anlotinib—an anti-angiogenic agent—mitigates penpulimab resistance. These findings offer insights to guide clinical management of immune-pretreated patients. Single-cell sequencing analyses demonstrated that anlotinib reverses penpulimab resistance by reprogramming the tumor immune microenvironment, thereby boosting PD-1 blockade efficacy via modulation of immune infiltration and tumor signaling pathways. Identifying Apoe⁺ M2 macrophages, Srgn⁺ M1 macrophages, and Cxcl2⁺ T cells provides key cellular and molecular targets for developing clinically actionable immunotherapies. Taken together, this work validates the preclinical potential of anlotinib combined with immunotherapy for immunotherapy-resistant tumors.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
All data are presented within the article and supplementary online data.
References
Bray F, Laversanne M, Sung H, Ferlay J, Siegel RL, Soerjomataram I, et al. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2024;74:229–63.
Surovtsova I, Herth FJF, Kokh DB, Morakis P. Outcomes of Pembrolizumab plus chemotherapy for patients with metastatic non-squamous NSCLC: real-world evidence. Pulmonology. 2025;31:2457856.
Planchard D, Popat S, Kerr K, Novello S, Smit EF, Faivre-Finn C, et al. Metastatic non-small cell lung cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2018;29:iv192–iv237.
Reck M, Remon J, Hellmann MD. First-line immunotherapy for non-small-cell lung cancer. J Clin Oncol. 2022;40:586–97.
Xiong W, Zhao Y, Du H, Guo X. Current status of immune checkpoint inhibitor immunotherapy for lung cancer. Front Oncol. 2021;11:704336.
Yi M, Zheng X, Niu M, Zhu S, Ge H, Wu K. Combination strategies with PD-1/PD-L1 blockade: current advances and future directions. Mol Cancer. 2022;21:28.
Petrovic N. Targeting angiogenesis in cancer treatments: where do we stand? J Pharm Pharm Sci. 2016;19:226–38.
Yi M, Jiao D, Qin S, Chu Q, Wu K, Li A. Synergistic effect of immune checkpoint blockade and anti-angiogenesis in cancer treatment. Mol Cancer. 2019;18:60.
Liang L, Hui K, Hu C, Wen Y, Yang S, Zhu P, et al. Autophagy inhibition potentiates the anti-angiogenic property of multikinase inhibitor anlotinib through JAK2/STAT3/VEGFA signaling in non-small cell lung cancer cells. J Exp Clin Cancer Res. 2019;38:71.
Lei T, Xu T, Zhang N, Zou X, Kong Z, Wei C, et al. Anlotinib combined with osimertinib reverses acquired osimertinib resistance in NSCLC by targeting the c-MET/MYC/AXL axis. Pharmacol Res. 2023;188:106668.
Han B, Chen J, Wang Z, Li X, WANG L, Wu L, et al. Penpulimab in combination with anlotinib as first-line treatment in advanced nonsquamous non-small-cell lung cancer. J Clin Oncol. 2021;39:e21072.
Song F, Hu B, Liang XL, Cheng JW, Wang CG, Wang PX, et al. Anlotinib potentiates anti-PD1 immunotherapy via transferrin receptor-dependent CD8(+) T-cell infiltration in hepatocellular carcinoma. Clin Transl Med. 2024;14:e1738.
Hao Y, Stuart T, Kowalski MH, Choudhary S, Hoffman P, Hartman A, et al. Dictionary learning for integrative, multimodal and scalable single-cell analysis. Nat Biotechnol. 2024;42:293–304.
Korsunsky I, Millard N, Fan J, Slowikowski K, Zhang F, Wei K, et al. Fast, sensitive and accurate integration of single-cell data with Harmony. Nat Methods. 2019;16:1289–96.
Schubert M, Klinger B, Klünemann M, Sieber A, Uhlitz F, Sauer S, et al. Perturbation-response genes reveal signaling footprints in cancer gene expression. Nat Commun. 2018;9:20.
Zheng SC, Stein-O’Brien G, Augustin JJ, Slosberg J, Carosso GA, Winer B, et al. Universal prediction of cell-cycle position using transfer learning. Genome Biol. 2022;23:41.
Qiu X, Mao Q, Tang Y, Wang L, Chawla R, Pliner HA, et al. Reversed graph embedding resolves complex single-cell trajectories. Nat Methods. 2017;14:979–982.
Wu Y, Yang S, Ma J, Chen Z, Song G, Rao D, et al. Spatiotemporal immune landscape of colorectal cancer liver metastasis at single-cell level. Cancer Discov. 2022;12:134–53.
Jin S, Guerrero-Juarez CF, Zhang L, Chang I, Ramos R, Kuan C-H, et al. Inference and analysis of cell-cell communication using CellChat. Nat Commun. 2021;12:1088.
Liberzon A, Birger C, Thorvaldsdóttir H, Ghandi M, Mesirov JP, Tamayo P. The Molecular Signatures Database (MSigDB) hallmark gene set collection. Cell Syst. 2015;1:417–25.
Liberzon A, Subramanian A, Pinchback R, Thorvaldsdóttir H, Tamayo P, Mesirov JP. Molecular signatures database (MSigDB) 3.0. Bioinformatics. 2011;27:1739–40.
Nicol PB, Paulson D, Qian G, Liu XS, Irizarry R, Sahu AD. Robust identification of perturbed cell types in single-cell RNA-seq data. Nat Commun. 2024;15:7610.
Gulati GS, Sikandar SS, Wesche DJ, Manjunath A, Bharadwaj A, Berger MJ, et al. Single-cell transcriptional diversity is a hallmark of developmental potential. Science. 2020;367:405–11.
Lu J, Luo Y, Rao D, Wang T, Lei Z, Chen X, et al. Myeloid-derived suppressor cells in cancer: therapeutic targets to overcome tumor immune evasion. Exp Hematol Oncol. 2024;13:39.
Laatsch A, Panteli M, Sornsakrin M, Hoffzimmer B, Grewal T, Heeren J. Low density lipoprotein receptor-related protein 1 dependent endosomal trapping and recycling of apolipoprotein E. PLoS One. 2012;7:e29385.
Bonacina F, Coe D, Wang G, Longhi MP, Baragetti A, Moregola A, et al. Myeloid apolipoprotein E controls dendritic cell antigen presentation and T cell activation. Nat Commun. 2018;9:3083.
Cedeno-Laurent F, Dimitroff CJ. Galectin-1 research in T cell immunity: past, present and future. Clin Immunol. 2012;142:107–16.
Su Y, Luo B, Lu Y, Wang D, Yan J, Zheng J, et al. Anlotinib induces a T cell-inflamed tumor microenvironment by facilitating vessel normalization and enhances the efficacy of PD-1 checkpoint blockade in neuroblastoma. Clin Cancer Res. 2022;28:793–809.
Han X, Liu J, Zhang Y, Tse E, Yu Q, Lu Y, et al. Increasing the tumour targeting of antitumour drugs through anlotinib-mediated modulation of the extracellular matrix and the RhoA/ROCK signalling pathway. J Pharm Anal. 2024;14:100984.
Imodoye SO, Adedokun KA. EMT-induced immune evasion: connecting the dots from mechanisms to therapy. Clin Exp Med. 2023;23:4265–87.
Gialeli C, Fountas P, Shami A, Sun J, Bocci M, Nitulescu M et al. The extracellular matrix proteoglycan serglycin is associated with human atherosclerotic plaque inflammation. Cardiovasc Res. 2024;120:cvae088.181.
Huo R, Zhao R, Li Z, Li M, Bin Y, Wang D, et al. APOE expression in papillary thyroid carcinoma: influencing tumor progression and macrophage polarization. Immunobiology. 2024;229:152821.
Lv Y, Chen C, Han M, Tian C, Song F, Feng S, et al. CXCL2: a key player in the tumor microenvironment and inflammatory diseases. Cancer Cell Int. 2025;25:133.
Liu D, Gao S, Zhai Y, Yang X, Zhai G. Research progress of tumor targeted drug delivery based on PD-1/PD-L1. Int J Pharm. 2022;616:121527.
Gide TN, Wilmott JS, Scolyer RA, Long GV. Primary and acquired resistance to immune checkpoint inhibitors in metastatic melanoma. Clin Cancer Res. 2018;24:1260–70.
Jiang Y, Zhan H. Communication between EMT and PD-L1 signaling: new insights into tumor immune evasion. Cancer Lett. 2020;468:72–81.
Pouysségur J, Marchiq I, Parks SK, Durivault J, Ždralević M, Vucetic M. ‘Warburg effect’ controls tumor growth, bacterial, viral infections and immunity - Genetic deconstruction and therapeutic perspectives. Semin Cancer Biol. 2022;86:334–46.
Yoshimura A, Muto G. TGF-β function in immune suppression. Curr Top Microbiol Immunol. 2011;350:127–47.
Wang S, Wang J, Chen Z, Luo J, Guo W, Sun L, et al. Targeting M2-like tumor-associated macrophages is a potential therapeutic approach to overcome antitumor drug resistance. NPJ Precis Oncol. 2024;8:31.
Pang K, Shi ZD, Wei LY, Dong Y, Ma YY, Wang W, et al. Research progress of therapeutic effects and drug resistance of immunotherapy based on PD-1/PD-L1 blockade. Drug Resist Updat. 2023;66:100907.
Huang Z, Li Y, Liu Q, Chen X, Lin W, Wu W, et al. SPP1-mediated M2 macrophage polarization shapes the tumor microenvironment and enhances prognosis and immunotherapy guidance in nasopharyngeal carcinoma. Int Immunopharmacol. 2025;147:113944.
Xue Z, Lui VWY, Li Y, Jia L, You C, Li X, et al. Therapeutic evaluation of palbociclib and its compatibility with other chemotherapies for primary and recurrent nasopharyngeal carcinoma. J Exp Clin Cancer Res. 2020;39:262.
Lu Y, Han X, Zhang H, Zheng L, Li X. Multi-omics study on the molecular mechanism of anlotinib in regulating tumor metabolism. Eur J Pharmacol. 2024;975:176639.
Liu S, Qin T, Liu Z, Wang J, Jia Y, Feng Y, et al. anlotinib alters tumor immune microenvironment by downregulating PD-L1 expression on vascular endothelial cells. Cell Death Dis. 2020;11:309.
Zavacky E. Investigating the heterogeneity of tumour-associated macrophages in renal cell carcinoma milieu. Montreal, Quebec, Canada; McGill University: 2023.
Zhang Y, Li Z, Chen C, Wei W, Li Z, Zhou H, et al. SRGN promotes macrophage recruitment through CCL3 in osteoarthritis. Connect Tissue Res. 2024;65:330–42.
Wang X, Xiong H, Liang D, Chen Z, Li X, Zhang K. The role of SRGN in the survival and immune infiltrates of skin cutaneous melanoma (SKCM) and SKCM-metastasis patients. BMC Cancer. 2020;20:378.
Gao J, Liang Y, Wang L. Shaping polarization of tumor-associated macrophages in cancer immunotherapy. Front Immunol. 2022;13:888713.
Ruytinx P, Proost P, Van Damme J, Struyf S. Chemokine-induced macrophage polarization in inflammatory conditions. Front Immunol. 2018;9:1930.
Zhang Y, Guan XY, Jiang P. Cytokine and chemokine signals of T-cell exclusion in tumors. Front Immunol. 2020;11:594609.
Zhang M, Wei Z, Wei B, Lai C, Zong G, Tao E, et al. Microbiota-derived urocanic acid triggered by tyrosine kinase inhibitors potentiates cancer immunotherapy efficacy. Cell Host Microbe. 2025;33:915–31.e9.
Lu C, Gao Z, Wu D, Zheng J, Hu C, Huang D, et al. Understanding the dynamics of TKI-induced changes in the tumor immune microenvironment for improved therapeutic effect. J Immunother Cancer. 2024;12:e009165.
Fang Y, Wang Y, Zeng D, Zhi S, Shu T, Huang N, et al. Comprehensive analyses reveal TKI-induced remodeling of the tumor immune microenvironment in EGFR/ALK-positive non-small-cell lung cancer. Oncoimmunology. 2021;10:1951019.
Allen E, Jabouille A, Rivera LB, Lodewijckx I, Missiaen R, Steri V, et al. Combined antiangiogenic and anti-PD-L1 therapy stimulates tumor immunity through HEV formation. Sci Transl Med. 2017;9:eaak9679.
Acknowledgements
We thank Chia Tai Tianqing Pharmaceutical Group Co., Ltd. for providing anlotinib and penpulimab.
Funding
This work was supported by the National Natural Science Foundation of China (Grant No. 82574740, 82473955, 82173842), the Fundamental Research Funds for the Central Universities (2632025TD04), and the Priority Academic Program Development (PAPD) of Jiangsu Higher Education Institutions.
Author information
Authors and Affiliations
Contributions
Lufeng Zheng and Xiaoman Li designed the research. Wanjin Shi and Yidong Zhang analyzed the data. Wanjin Shi, Yidong Zhang, Qiyi Yu, Huilong Li, Miaomiao Niu, Shengtao Xu and Yin Jun performed the research. Wanjin Shi and Yidong Zhang wrote the paper. Lufeng Zheng, and Xiaoman Li reviewed this paper. All data were generated in-house, and no paper mill was used. All authors agree to be accountable for all aspects of work ensuring integrity and accuracy.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
Animal studies were conducted in strict compliance with guidelines and protocols submitted to and approved by the Animal Welfare and Ethics Committee (AWEC) of China Pharmaceutical University (approval number: 2024-08-003), in adherence to the National Institutes of Health (NIH) Guide for the Care and Use of Laboratory Animals.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Shi, W., Zhang, Y., Yu, Q. et al. Exploration of the mechanism of anlotinib in reversing PD-1 immunotherapy resistance: insights from single-cell sequencing. Cancer Gene Ther 33, 248–260 (2026). https://doi.org/10.1038/s41417-026-01000-3
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41417-026-01000-3