Abstract
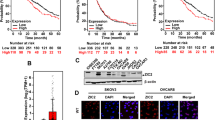
Oral squamous cell carcinoma (OSCC) is a common malignant tumor of the head and neck. Early-stage OSCC is primarily treated using surgery; advanced-stage OSCC is managed using a multidisciplinary approach, including surgery combined with adjuvant radiotherapy and chemotherapy. However, tumor recurrence and metastasis remain major challenges, with a 5-year survival rate of ~50%. Dysregulation of transcription factors is associated with the pathogenesis of various cancers. This study focused on the role of ZIC2, a member of the zinc finger protein family, in OSCC. ZIC2 was identified as a prognostically relevant transcription factor in OSCC through bioinformatic analysis, showing high expression in OSCC and association with poor prognosis in patients. In vitro and in vivo, ZIC2 knockdown inhibited the proliferation, migration, invasion, and spheroid formation ability of OSCC cells and restored their sensitivity to chemotherapeutic drugs; overexpression of ZIC2 showed the opposite effect. RNA-seq and targeted metabolomics analyses revealed that in OSCC cells with zic2 knockdown, the expression of glycerophosphocholine (GPC) and the key rate-limiting enzyme LYPLA2 was decreased. LYPLA2 overexpression rescued the effects of ZIC2 knockdown on the proliferation, migration, and invasion of OSCC cells. GPC increased the stemness of OSCC tumor cells; ZIC2-regulated GPC metabolism through LYPLA2, inducing changes in the expression of the cancer stem cell markers Nanog and OCT4. In conclusion, we identified ZIC2 as an OSCC stemness-related gene, a potential therapeutic target for OSCC, providing new insights for treating OSCC.
Similar content being viewed by others
Data availability
The RNA-seq data have been deposited in the GEO database (GSE304366). The results of targeted metabolomics are provided in the supplementary materials 5, and detailed information can be obtained by contacting the corresponding author.
References
Ma X, Zhang X, Li H, Mei S, Wang B, Guan S, et al. Knockdown of HOXD13 in oral squamous cell carcinoma inhibited its proliferation, migration, and influenced fatty acid metabolism. J Cancer. 2025;16:214–26.
Ng JH, Iyer NG, Tan MH, Edgren G. Changing epidemiology of oral squamous cell carcinoma of the tongue: a global study. Head Neck. 2017;39:297–304.
Mody MD, Rocco JW, Yom SS, Haddad RI, Saba NF. Head and neck cancer. Lancet. 2021;398:2289–99.
Kinane DF, Gabert J, Xynopoulos G, Guzeldemir-Akcakanat E. Strategic approaches in oral squamous cell carcinoma diagnostics using liquid biopsy. Periodontol 2000. 2024;96:316–28.
Olmedo I, Martínez D, Carrasco-Rojas J, Jara JA. Mitochondria in oral cancer stem cells: unraveling the potential drug targets for new and old drugs. Life Sci. 2023;331:122065.
Magnes T, Wagner S, Kiem D, Weiss L, Rinnerthaler G, Greil R, et al. Prognostic and predictive factors in advanced head and neck squamous cell carcinoma. Int J Mol Sci 2021;22:4981.
Jiang C, Liu F, Xiao S, He L, Wu W, Zhao Q. miR-29a-3p enhances the radiosensitivity of oral squamous cell carcinoma cells by inhibiting ADAM12. Eur J Histochem 2021;65:3295.
Gan M, Liu N, Li W, Chen M, Bai Z, Liu D, et al. Metabolic targeting of regulatory T cells in oral squamous cell carcinoma: new horizons in immunotherapy. Mol Cancer. 2024;23:273.
Morgan MP, Finnegan E, Das S. The role of transcription factors in the acquisition of the four latest proposed hallmarks of cancer and corresponding enabling characteristics. Semin Cancer Biol. 2022;86:1203–15.
Marchini S, Poynor E, Barakat RR, Clivio L, Cinquini M, Fruscio R, et al. The zinc finger gene ZIC2 has features of an oncogene and its overexpression correlates strongly with the clinical course of epithelial ovarian cancer. Clin Cancer Res. 2012;18:4313–24.
Lu SX, Zhang CZ, Luo RZ, Wang CH, Liu LL, Fu J, et al. Zic2 promotes tumor growth and metastasis via PAK4 in hepatocellular carcinoma. Cancer Lett. 2017;402:71–80.
Lv Z, Wang M, Hou H, Tang G, Xu H, Wang X, et al. FOXM1-regulated ZIC2 promotes the malignant phenotype of renal clear cell carcinoma by activating UBE2C/mTOR signaling pathway. Int J Biol Sci. 2023;19:3293–306.
Liu Q, Yang T, Zhang Y, Hu ZD, Liu YM, Luo YL, et al. ZIC2 induces pro-tumor macrophage polarization in nasopharyngeal carcinoma by activating the JUNB/MCSF axis. Cell Death Dis. 2023;14:455.
Liu ZH, Chen ML, Zhang Q, Zhang Y, An X, Luo YL, et al. ZIC2 is downregulated and represses tumor growth via the regulation of STAT3 in breast cancer. Int J Cancer. 2020;147:505–18.
Prager BC, Xie Q, Bao S, Rich JN. Cancer stem cells: the architects of the tumor ecosystem. Cell Stem Cell. 2019;24:41–53.
Praharaj PP, Panigrahi DP, Bhol CS, Patra S, Mishra SR, Mahapatra KK, et al. Mitochondrial rewiring through mitophagy and mitochondrial biogenesis in cancer stem cells: A potential target for anti-CSC cancer therapy. Cancer Lett. 2021;498:217–28.
Rodrigues M, Xavier FCA, Andrade NP, Lopes C, Miguita Luiz L, Sedassari BT, et al. Prognostic implications of CD44, NANOG, OCT4, and BMI1 expression in tongue squamous cell carcinoma. Head Neck. 2018;40:1759–73.
Chen H, Lee LJ, Vincent KM, Xu Z, Liu J, Zhang G, et al. Transcription factor ZIC2 regulates the tumorigenic phenotypes associated with both bulk and cancer stem cells in epithelial ovarian cancer. Oncogene. 2024;43:1688–1700.
Zhu P, Wang Y, He L, Huang G, Du Y, Zhang G, et al. ZIC2-dependent OCT4 activation drives self-renewal of human liver cancer stem cells. J Clin Invest. 2015;125:3795–808.
Martin-Perez M, Urdiroz-Urricelqui U, Bigas C, Benitah SA. The role of lipids in cancer progression and metastasis. Cell Metab. 2022;34:1675–99.
Anaokar S, Kodali R, Jonik B, Renne MF, Brouwers J, Lager I, et al. The glycerophosphocholine acyltransferase Gpc1 is part of a phosphatidylcholine (PC)-remodeling pathway that alters PC species in yeast. J Biol Chem. 2019;294:1189–201.
Sonkar K, Ayyappan V, Tressler CM, Adelaja O, Cai R, Cheng M, et al. Focus on the glycerophosphocholine pathway in choline phospholipid metabolism of cancer. NMR Biomed. 2019;32:e4112.
Sakuma K, Kasamatsu A, Yamatoji M, Yamano Y, Fushimi K, Iyoda M, et al. Expression status of Zic family member 2 as a prognostic marker for oral squamous cell carcinoma. J Cancer Res Clin Oncol. 2010;136:553–9.
Meng X, Lou QY, Yang WY, Wang YR, Chen R, Wang L, et al. The role of non-coding RNAs in drug resistance of oral squamous cell carcinoma and therapeutic potential. Cancer Commun. 2021;41:981–1006.
Celià-Terrassa T, Jolly MK. Cancer stem cells and epithelial-to-mesenchymal transition in cancer metastasis. Cold Spring Harb Perspect Med 2020;10:a036905.
Mitrofanova A, Merscher S, Fornoni A. Kidney lipid dysmetabolism and lipid droplet accumulation in chronic kidney disease. Nat Rev Nephrol. 2023;19:629–45.
Li H, Feng Z, He ML. Lipid metabolism alteration contributes to and maintains the properties of cancer stem cells. Theranostics. 2020;10:7053–69.
Daniel Y, Lelou E, Aninat C, Corlu A, Cabillic F. Interplay between metabolism reprogramming and epithelial-to-mesenchymal transition in cancer stem cells. Cancers 2021;13:1973.
Tressler CM, Sonkar K, Cheng M, Ayyappan V, Cai R, Glunde K. Molecular effects of clinically relevant chemotherapeutic agents on choline phospholipid metabolism in triple negative breast cancer cells. Transl Oncol. 2025;53:102311.
Li Z, Tan Y, Li X, Quan J, Bode AM, Cao Y, et al. DHRS2 inhibits cell growth and metastasis in ovarian cancer by downregulation of CHKα to disrupt choline metabolism. Cell Death Dis. 2022;13:845.
Wei-Hua W, Ning Z, Qian C, Dao-Wen J. ZIC2 promotes cancer stem cell traits via up-regulating OCT4 expression in lung adenocarcinoma cells. J Cancer. 2020;11:6070–80.
Tayebati SK, Marucci G, Santinelli C, Buccioni M, Amenta F. Choline-containing phospholipids: structure-activity relationships versus therapeutic applications. Curr Med Chem. 2015;22:4328–40.
Keller M, Rohlf K, Glotzbach A, Leonhardt G, Lüke S, Derksen K, et al. Inhibiting the glycerophosphodiesterase EDI3 in ER-HER2+ breast cancer cells resistant to HER2-targeted therapy reduces viability and tumour growth. J Exp Clin Cancer Res. 2023;42:25.
Wang G, Xu J, Zhao J, Yin W, Liu D, Chen W, et al. Arf1-mediated lipid metabolism sustains cancer cells and its ablation induces anti-tumor immune responses in mice. Nat Commun. 2020;11:220.
Zang W, Geng F, Liu J, Wang Z, Zhang S, Li Y, et al. Porphyromonas gingivalis potentiates stem-like properties of oral squamous cell carcinoma by modulating SCD1-dependent lipid synthesis via NOD1/KLF5 axis. Int J Oral Sci. 2025;17:15.
Wepy JA, Galligan JJ, Kingsley PJ, Xu S, Goodman MC, Tallman KA, et al. Lysophospholipases cooperate to mediate lipid homeostasis and lysophospholipid signaling. J Lipid Res. 2019;60:360–74.
Acknowledgements
We would like to express our gratitude to Dr. Jingcun Shi, Dr. Ruiyu Guo, Dr. Huasheng Li, Dr. Lu Zhao, Dr. Mei Zhang and Dr. Hexin Ma from the Shanghai Key Laboratory of Stomatology for their assistance in this study. We also thank the staff of the TCGA project team and the GEO project team for providing public data.
Funding
This study was supported by the Project of Central Government Guiding Local Scientific and Technological Development Funds in Hebei Province (Project no.: 246Z7728G), the Key Project of the Joint Fund for Precision Medicine Innovation and Development (Project no.: H2025206023), the Excellent Clinical Medicine Talents Training Project Funded by the Government in 2024 (Project no.: ZF2024148), and the Excellent Clinical Medicine Talents Training Project Funded by the Government in 2025 (Project no.: ZF2025241).
Author information
Authors and Affiliations
Contributions
Siyi Li and Xingyue Ma designed the experiments, completed most of the experimental work, and wrote the manuscript. Ming Yan, Longwei Hu, and Ran Li provided experimental guidance. Xingyue Ma and Yuantao Li conducted the animal experiments. Haiyang Li and Bowen Wang assisted in completing the molecular biology experiments. Jianping Liu and Xiaoyan Zhang carried out the data analysis. Shuang Mei and Xiangjun Li revised the manuscript and supervised the research.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics statement
This study has been approved by the Ethics Committee of the Stomatological Hospital of Hebei Medical University, and written informed consent has been collected from each patient with OSCC. The animal experiment has been approved by the Laboratory Animal Welfare and Ethics Committee of Hebei Medical University (Laboratory Animal Use License no.: SYXK(Ji)2020-002), and all experiments have been carried out in accordance with the guiding principles and relevant laws.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Edited by Professor Mauro Piacentini
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Li, S., Ma, X., Li, Y. et al. ZIC2 affects oral squamous cell carcinoma stemness by regulating glycerophosphocholine metabolism via LYPLA2. Cell Death Dis (2026). https://doi.org/10.1038/s41419-026-08483-w
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41419-026-08483-w