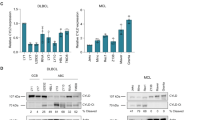
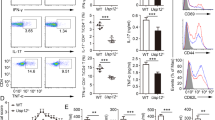
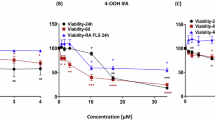
Abstract
CYLD is a functional deubiquitinase, involved in the regulation of significant cellular functions, including survival and apoptosis. To elucidate the role of CYLD in B cell differentiation, we generated transgenic animals with targeted deletion of the catalytically active form of the protein in B cells, starting from early differentiation stages. Our results indicate that catalytic inactivation of CYLD leads to a severe reduction of mature B cells, associated with blockade of differentiation at the Pro B cell stage, altered distribution of B cell populations in the spleen and bone marrow, culminating in impaired immune responses to model antigens. Single cell RNA sequencing of bone marrow B cells confirmed the severe perturbation of lymphopoiesis. Mechanistically, we found impaired expression of the IL-7 receptor alpha chain (IL-7Ra) and its upstream transcriptional activator FOXO1, leading to defective IL-7 signaling that is vital for early B cell development. However, the substrate(s) deubiquitinated by CYLD that regulates the FOXO1-IL-7R pathway remains unclear. Overall, our data imply a crucial role for the deubiquitinase activity of CYLD in B cell lymphopoiesis.
Similar content being viewed by others
Data availability
Single cell RNA-seq data are deposited at Zenodo.org (https://zenodo.org/records/10124303?token=eyJhbGciOiJIUzUxMiJ9.eyJpZCI6IjUyZmIyNWFjLTBlOWItNDNlMi05NWFjLWVkYTI2YTIzNWQ2MyIsImRhdGEiOnt9LCJyYW5kb20iOiIwMjQ0NTcwOTJlOTUwYzljNjAzNGYxMjNlNTVmOTQ2YyJ9.QCtDq1LN61nqsNZ94dTGcXXDN1icwO-_iDOO_1_oVv5VGuG3-rktO4SYqDo). All relevant scripts, code and information about the bioinformatic analysis and computational models generated can be found at https://github.com/BiodataAnalysisGroup/Cyld-regulation-of-bcell-maturation. All other data are available from the corresponding authors upon request. Values for all data points in graphs are reported in the Supporting Data Values file.
References
Komander D, Reyes-Turcu F, Licchesi JDF, Odenwaelder P, Wilkinson KD, Barford D. Molecular discrimination of structurally equivalent Lys 63-linked and linear polyubiquitin chains. EMBO Rep. 2009;10:466–73.
Bignell GR, Warren W, Seal S, Takahashi M, Rapley E, Barfoot R, et al. Identification of the familial cylindromatosis tumour-suppressor gene. Nat Genet. 2000;25:160–5.
Pannem RR, Dorn C, Ahlqvist K, Bosserhoff AK, Hellerbrand C, Massoumi R. CYLD controls c-MYC expression through the JNK-dependent signaling pathway in hepatocellular carcinoma. Carcinogenesis. 2014;35:461–8.
Massoumi R, Kuphal S, Hellerbrand C, Haas B, Wild P, Spruss T, et al. Down-regulation of CYLD expression by Snail promotes tumor progression in malignant melanoma. J Exp Med. 2009;206:221–32.
Li YY, Chung GTY, Lui VWY, To K-F, Ma BBY, Chow C, et al. Exome and genome sequencing of nasopharynx cancer identifies NF-κB pathway activating mutations. Nat Commun. 2017;8:14121.
Zhang J, Stirling B, Temmerman ST, Ma CA, Fuss IJ, Derry JMJ, et al. Impaired regulation of NF-κB and increased susceptibility to colitis-associated tumorigenesis in CYLD-deficient mice. J Clin Investig. 2006;116:3042–9.
Karatzas DN, Xanthopoulos K, Kotantaki P, Pseftogas A, Teliousis K, Hatzivassiliou EG, et al. Inactivation of CYLD in intestinal epithelial cells exacerbates colitis-associated colorectal carcinogenesis - a short report. Cell Oncol. 2016;39:287–93.
Nikolaou K, Tsagaratou A, Eftychi C, Kollias G, Mosialos G, Talianidis I. Inactivation of the deubiquitinase CYLD in hepatocytes causes apoptosis, inflammation, fibrosis, and cancer. Cancer Cell. 2012;21:738–50.
Ling T, Yu F, Cao H. miR-182 controls cell growth in gastrointestinal stromal tumors by negatively regulating CYLD expression. Oncol Rep. 2018. https://doi.org/10.3892/or.2018.6765.
Xu D, Zhou P, Wang Y, Zhang L, Fu W, Ruan B, et al. Reciprocal activation between STAT3 and miR-181b regulates the proliferation of esophageal cancer stem-like cells via the CYLD pathway. Mol Cancer. 2016;15:40.
Hajek M, Sewell A, Kaech S, Burtness B, Yarbrough WG, Issaeva N. TRAF3/CYLD mutations identify a distinct subset of human papillomavirus-associated head and neck squamous cell carcinoma. Cancer. 2017;123:1778–90.
Jiang K, Xie L-F, Xiao T-Z, Qiu M-Y, Wang W-L. MiR-181d inhibits cell proliferation and metastasis through PI3K/AKT pathway in gastric cancer. Eur Rev Med Pharm Sci. 2019;23:8861–9.
Mo Z, Wood S, Namiranian S, Mizukoshi R, Weng S, Jang IS, et al. Deciphering the mechanisms of CC-122 resistance in DLBCL via a genome-wide CRISPR screen. Blood Adv. 2021;5:2027–39.
Xu X, Wei T, Zhong W, Ang R, Lei Y, Zhang H, et al. Down-regulation of cylindromatosis protein phosphorylation by BTK inhibitor promotes apoptosis of non-GCB-diffuse large B-cell lymphoma. Cancer Cell Int. 2021;21:195.
Annunziata CM, Davis RE, Demchenko Y, Bellamy W, Gabrea A, Zhan F, et al. Frequent engagement of the classical and alternative NF-κB pathways by diverse genetic abnormalities in multiple myeloma. Cancer Cell. 2007;12:115–30.
Jenner MW, Leone PE, Walker BA, Ross FM, Johnson DC, Gonzalez D, et al. Gene mapping and expression analysis of 16q loss of heterozygosity identifies WWOX and CYLD as being important in determining clinical outcome in multiple myeloma. Blood. 2007;110:3291–3300.
Keats JJ, Fonseca R, Chesi M, Schop R, Baker A, Chng W-J, et al. Promiscuous mutations activate the noncanonical NF-κB pathway in multiple myeloma. Cancer Cell. 2007;12:131–44.
Brummelkamp TR, Nijman SMB, Dirac AMG, Bernards R. Loss of the cylindromatosis tumour suppressor inhibits apoptosis by activating NF-κB. Nature. 2003;424:797–801.
Kovalenko A, Chable-Bessia C, Cantarella G, Israël A, Wallach D, Courtois G. The tumour suppressor CYLD negatively regulates NF-kappaB signalling by deubiquitination. Nature. 2003;424:801–5.
Trompouki E, Hatzivassillou E, Tsichritzis T, Farmer H, Ashworth A, Mosialos G. CYLD is a deubiquitinating enzyme that negatively regulates NF-kappaB activation by TNFR family members. Nature. 2003;424:793–6.
Tauriello DVF, Haegebarth A, Kuper I, Edelmann MJ, Henraat M, Canninga-van Dijk MR, et al. Loss of the tumor suppressor CYLD enhances Wnt/beta-catenin signaling through K63-linked ubiquitination of Dvl. Mol Cell. 2010;37:607–19.
Rajan N, Elliott RJR, Smith A, Sinclair N, Swift S, Lord CJ, et al. The cylindromatosis gene product, CYLD, interacts with MIB2 to regulate notch signalling. Oncotarget. 2014;5:12126–40.
Yang Y, Zhou J. CYLD – a deubiquitylase that acts to fine-tune microtubule properties and functions. J Cell Sci 2016. https://doi.org/10.1242/jcs.183319.
Sun S-C. CYLD: a tumor suppressor deubiquitinase regulating NF-κB activation and diverse biological processes. Cell Death Differ. 2010;17:25–34.
Trompouki E, Tsagaratou A, Kosmidis SK, Dollá P, Qian J, Kontoyiannis DL, et al. Truncation of the catalytic domain of the cylindromatosis tumor suppressor impairs lung maturation. Neoplasia. 2009;11:469–76.
Eguether T, Ermolaeva MA, Zhao Y, Bonnet MC, Jain A, Pasparakis M, et al. The deubiquitinating enzyme CYLD controls apical docking of basal bodies in ciliated epithelial cells. Nat Commun. 2014;5:1–14.
Massoumi R, Chmielarska K, Hennecke K, Pfeifer A, Fässler R. Cyld inhibits tumor cell proliferation by blocking Bcl-3-dependent NF-κB Signaling. Cell. 2006;125:665–77.
Reiley W, Zhang M, Jin W, Losiewicz M, Donohue KB, Norbury CC, et al. Regulation of T cell development by the deubiquitinating enzyme CYLD. Nat Immunol. 2006;7:411–7.
Massoumi R. Ubiquitin chain cleavage: CYLD at work. Trends Biochem Sci. 2010;35:392–9.
Jin W, Reiley WR, Lee AJ, Wright A, Wu X, Zhang M, et al. Deubiquitinating enzyme CYLD regulates the peripheral development and naive phenotype maintenance of B Cells *♦. J Biol Chem. 2007;282:15884–93.
Hövelmeyer N, Wunderlich FT, Massoumi R, Jakobsen CG, Song J, Wörns MA, et al. Regulation of B cell homeostasis and activation by the tumor suppressor gene CYLD. J Exp Med. 2007;204:2615–27.
Hahn M, Bürckert JP, Luttenberger CA, Klebow S, Hess M, Al-Maarri M, et al. Aberrant splicing of the tumor suppressor CYLD promotes the development of chronic lymphocytic leukemia via sustained NF-κB signaling. Leukemia. 2018;32:72–82.
Hobeika E, Thiemann S, Storch B, Jumaa H, Nielsen PJ, Pelanda R, et al. Testing gene function early in the B cell lineage in mb1-cre mice. Proc Natl Acad Sci USA. 2006;103:13789–94.
Holl TM, Haynes BF, Kelsoe G. Stromal cell independent B cell development in vitro: generation and recovery of autoreactive clones. J Immunol Methods. 2010;354:53–67.
Hao Y, Hao S, Andersen-Nissen E, Mauck WM, Zheng S, Butler A, et al. Integrated analysis of multimodal single-cell data. Cell. 2021;184:3573–3587.e29.
Hao Y, Stuart T, Kowalski M, Choudhary S, Hoffman P, Hartman A et al. Dictionary learning for integrative, multimodal, and scalable single-cell analysis. bioRxiv 2022: 2022.02.24.481684.
Hafemeister C, Satija R. Normalization and variance stabilization of single-cell RNA-seq data using regularized negative binomial regression. Genome Biol. 2019;20:1–15.
Aran D, Looney AP, Liu L, Wu E, Fong V, Hsu A, et al. Reference-based analysis of lung single-cell sequencing reveals a transitional profibrotic macrophage. Nat Immunol. 2019;20:163–72.
Cannoodt R, Saelens W, Sichien D, Tavernier S, Janssens S, Guilliams M et al. SCORPIUS improves trajectory inference and identifies novel modules in dendritic cell development. bioRxiv 2016; 079509.
Szklarczyk D, Gable AL, Lyon D, Junge A, Wyder S, Huerta-Cepas J, et al. STRING v11: protein–protein association networks with increased coverage, supporting functional discovery in genome-wide experimental datasets. Nucleic Acids Res. 2019;47:D607–D613.
Szklarczyk D, Kirsch R, Koutrouli M, Nastou K, Mehryary F, Hachilif R, et al. The STRING database in 2023: protein–protein association networks and functional enrichment analyses for any sequenced genome of interest. Nucleic Acids Res. 2023;51:D638–D646.
Shannon P, Markiel A, Ozier O, Baliga NS, Wang JT, Ramage D, et al. Cytoscape: a software environment for integrated models of biomolecular interaction networks. Genome Res. 2003;13:2498–504.
Yu J, Ding L, Shi H, Qian C, Burdyshaw C, Veloso J et al. scMINER: a mutual information-based framework for identifying hidden drivers from single-cell omics data. 2023. https://doi.org/10.21203/rs.3.rs-2476875/v1.
Clarke DJB, Kuleshov MV, Schilder BM, Torre D, Duffy ME, Keenan AB, et al. eXpression2Kinases (X2K) web: linking expression signatures to upstream cell signaling networks. Nucleic Acids Res. 2018;46:W171–W179.
Marín-Rubio JL, Raote I, Inns J, Dobson-Stone C, Rajan N. CYLD in health and disease. Dis Model Mech. 2023;16:050093. https://doi.org/10.1242/dmm.050093.
Zhang XK, Moussa O, LaRue A, Bradshaw S, Molano I, Spyropoulos DD, et al. The transcription factor Fli-1 modulates marginal zone and follicular B cell development in mice. J Immunol. 2008;181:1644–54.
Awaji K, Shibata S, Koyama A, Yamamoto T, Fukui Y, Toyama S, et al. Impact of Fli1 deletion on B cell populations: A focus on age-associated B cells and transcriptional dynamics. J Dermatol Sci. 2025;117:19–29.
Clarke DJB, Evangelista JE, Xie Z, Marino GB, Maurya MR, Srinivasan S et al. Playbook workflow builder: interactive construction of bioinformatics workflows from a network of microservices. 2024. https://doi.org/10.1101/2024.06.08.598037.
Marino GB, Evangelista JE, Clarke DJB, Ma’ayan A. L2S2: chemical perturbation and CRISPR KO LINCS L1000 signature search engine. Nucleic Acids Res. 2025;53:W338–W350.
Park J-H, Yu Q, Erman B, Appelbaum JS, Montoya-Durango D, Grimes HL, et al. Suppression of IL7Rα transcription by IL-7 and other prosurvival cytokines. Immunity. 2004;21:289–302.
Dengler HS, Baracho GV, Omori SA, Bruckner S, Arden KC, Castrillon DH, et al. Distinct functions for the transcription factor Foxo1 at various stages of B cell differentiation. Nat Immunol. 2008;9:1388–98.
Srivastava B, Quinn WJ, Hazard K, Erikson J, Allman D. Characterization of marginal zone B cell precursors. J Exp Med. 2005;202:1225–34.
Gavriilidis GI, Vasileiou V, Dimitsaki S, Karakatsoulis G, Giannakakis A, Pavlopoulos GA et al. APNet, an explainable sparse deep learning model to discover differentially active drivers of severe COVID-19. Bioinformatics 2025. https://doi.org/10.1093/bioinformatics/btaf063.
Cox E-M, El-Behi M, Ries S, Vogt JF, Kohlhaas V, Michna T, et al. AKT activity orchestrates marginal zone B cell development in mice and humans. Cell Rep. 2023;42:112378.
Ochiai K, Maienschein-Cline M, Mandal M, Triggs JR, Bertolino E, Sciammas R, et al. A self-reinforcing regulatory network triggered by limiting IL-7 activates pre-BCR signaling and differentiation. Nat Immunol. 2012;13:300–7.
Peschon JJ, Morrissey PJ, Grabstein KH, Ramsdell FJ, Maraskovsky E, Gliniak BC, et al. Early lymphocyte expansion is severely impaired in interleukin 7 receptor-deficient mice. J Exp Med. 1994;180:1955–60.
Von Freeden-Jeffry U, Vieira P, Lucian LA, McNeil T, Burdach SEG, Murray R. Lymphopenia in interleukin (IL)-7 gene-deleted mice identifies IL-7 as a nonredundant cytokine. J Exp Med. 1995;181:1519–26.
Corfe SA, Paige CJ. The many roles of IL-7 in B cell development; Mediator of survival, proliferation and differentiation. Semin Immunol. 2012;24:198–208.
Clark MR, Mandal M, Ochiai K, Singh H. Orchestrating B cell lymphopoiesis through interplay of IL-7 receptor and pre-B cell receptor signalling. Nat Rev Immunol. 2013;14:69–80.
Kaiser FMP, Janowska I, Menafra R, de Gier M, Korzhenevich J, Pico-Knijnenburg I, et al. IL-7 receptor signaling drives human B-cell progenitor differentiation and expansion. Blood. 2023;142:1113–30.
Acknowledgements
This study was funded by AIRC (5 per Mille 2018, Project ID 21198, G.P.; iCare-2 fellowship, Project ID 22466, P.A.; AIRC fellowship, Project ID 28100, P.A.), European Commission (H2020-MSCA-IF-2015, Project ID 708041, X.Κ. H2020-MSCA, Project ID 800924, P.A.).
Author information
Authors and Affiliations
Contributions
Conceptualization, XK, GP, and MoG; methodology, PA, BJ, FM, CA, RA, MoG, GM, FS-C, and VV; investigation, PA, BJ, GG, FM, CA, MG, TE, ST, DD, MoG, TC, PF, WTH, SK, GP, and KX; visualization, PA, BJ, and GG; funding acquisition, KX, PG, and KS; writing – original draft, PA, BJ, and GG; writing – review & editing KX, PG, and KS.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Edited by Professor Hans-Uwe Simon
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Pseftogas, A., Bordini, J., Gavriilidis, G. et al. The deubiquitinase activity of CYLD is required for B cell differentiation. Cell Death Dis (2026). https://doi.org/10.1038/s41419-026-08555-x
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41419-026-08555-x