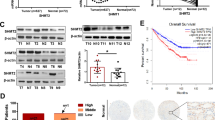
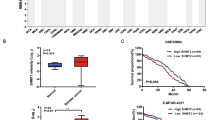
Abstract
Serine hydroxymethyltransferase (SHMT) is a key enzyme in one-carbon (1 C) metabolism, essential for nucleotide synthesis and epigenetic maintenance. In mammals, there are two distinct SHMT isozymes: the cytosolic SHMT1 and the mitochondrial SHMT2. Several studies report that high SHMT levels in cancer contribute to metabolic reprogramming. Conversely, a limited number of studies have linked decreased SHMT1 expression to the progression and poor prognosis of hepatocellular carcinoma and renal cell carcinoma, suggesting that SHMT may play dual roles as an oncogene or tumor suppressor, depending on the cellular context. However, the molecular mechanisms underlying SHMT tumor suppressor role remain unknown. In this work, we used the Drosophila RasV12DlgRNAi cancer model to investigate the effects of SHMT depletion on cancer progression and the associated mechanisms. We found that RNAi-mediated SHMT silencing promotes the progression of RasV12DlgRNAi cancers by impairing thymidylate biosynthesis in the folate pathway. SHMT depletion in RasV12DlgRNAi cells causes DNA and chromosome damage and renders these cells sensitive to genotoxic stressors such as X-rays or hydroxyurea. Genome instability is correlated with cancer progression, and it is largely due to the generation of reactive oxygen species (ROS) and, to a lesser degree, to replicative stress and compromised DNA repair mechanisms, all arising from SHMT depletion. Antioxidant treatment with N-acetyl cysteine (NAC) significantly reduces both DNA damage and tumor progression. Intriguingly, the combined depletion of SHMT and its cofactor pyridoxal 5’-phosphate (PLP) further increases oxidative stress, leading to extensive DNA damage that induces apoptosis in RasV12DlgRNAi cells, thereby limiting the tumor growth. Taken together, our data suggest that a diminished SHMT activity may drive the progression of RasV12DlgRNAi cancers through ROS-induced genome instability. Additionally, our study points to a novel gene-nutrient interaction, SHMT-PLP, that impacts cancer growth with potential therapeutic implications.
Similar content being viewed by others
Data availability
All data reported in this paper will be shared upon request.
References
Lyon P, Strippoli V, Fang B, Cimmino L. B Vitamins and One-Carbon Metabolism: Implications in Human Health and Disease. Nutrients. 2020;12:2867.
Fox JT, Stover PJ. Folate-mediated one-carbon metabolism. Vitam Horm. 2008;79:1–44.
Anderson DD, Stover PJ. SHMT1 and SHMT2 are functionally redundant in nuclear de novo thymidylate biosynthesis. PLoS One. 2009;4:e5839.
Liu X, Reig B, Nasrallah IM, Stover PJ. Human cytoplasmic serine hydroxymethyltransferase is an mRNA binding protein. Biochemistry. 2000;39:11523–31.
Guiducci G, Paone A, Tramonti A, Giardina G, Rinaldo S, Bouzidi A, et al. The moonlighting RNA-binding activity of cytosolic serine hydroxymethyltransferase contributes to control compartmentalization of serine metabolism. Nucleic Acids Res. 2019;47:4240–54.
Spizzichino S, Di Fonzo F, Marabelli C, Tramonti A, Chaves-Sanjuan A, Parroni A, et al. Structure-based mechanism of riboregulation of the metabolic enzyme SHMT1. Mol Cell. 2024;84:2682–2697.e6.
Fu R, Sun F, Wang W, Wang R, Zhang H, He X, et al. SHMT proteins: An emerging set of serine hydroxymethyltransferase in cancer. Cell Signal. 2025;135:111977.
Qi C, Qin X, Zhou Z, Wang Y, Yang Q, Liao T. Circ_0072995 Promotes Cell Carcinogenesis via Up-Regulating miR-149-5p-Mediated SHMT2 in Breast Cancer. Cancer Manag Res. 2020;12:11169–81.
Zhang H, Che Y, Xuan B, Wu X, Li H. Serine hydroxymethyltransferase 2 (SHMT2) potentiates the aggressive process of oral squamous cell carcinoma by binding to interleukin enhancer-binding factor 2 (ILF2). Bioengineered. 2022;13:8785–97.
Kim D, Fiske BP, Birsoy K, Freinkman E, Kami K, Possemato RL, et al. SHMT2 drives glioma cell survival in ischaemia but imposes a dependence on glycine clearance. Nature. 2015;520:363–7.
Wilke AC, Doebele C, Zindel A, Lee KS, Rieke SA, Ceribelli M, et al. SHMT2 inhibition disrupts the TCF3 transcriptional survival program in Burkitt lymphoma. Blood. 2022;139:538–53.
Zhang P, Yang Q. Overexpression of SHMT2 Predicts a Poor Prognosis and Promotes Tumor Cell Growth in Bladder Cancer. Front Genet. 2021;12:682856.
Su SW, Chen X, Wang G, Li P, Yang TX, Fang KW, et al. A study on the significance of serine hydroxymethyl transferase expression and its role in bladder cancer. Sci Rep. 2024;14:8324.
Shan Y, Liu D, Li Y, Wu C, Ye Y. The expression and clinical significance of serine hydroxymethyltransferase2 in gastric cancer. PeerJ. 2024;12:e16594.
Sun M, Zhao M, Li R, Zhang Y, Shi X, Ding C, et al. SHMT2 promotes papillary thyroid cancer metastasis through epigenetic activation of AKT signaling. Cell Death Dis. 2024;15:87.
Gao Y, Jing N, Teng X, Wang Y. Serine hydroxymethyltransferase 1 promotes low-grade glioma progression by activating mTORC1 signaling. Neurol Res. 2023;45:415–22.
Paone A, Marani M, Fiascarelli A, Rinaldo S, Giardina G, Contestabile R, et al. SHMT1 knockdown induces apoptosis in lung cancer cells by causing uracil misincorporation. Cell Death Dis. 2014;5:e1525.
Gupta R, Yang Q, Dogra SK, Wajapeyee N. Serine hydroxymethyl transferase 1 stimulates pro-oncogenic cytokine expression through sialic acid to promote ovarian cancer tumor growth and progression. Oncogene. 2017;36:4014–24.
Dou C, Xu Q, Liu J, Wang Y, Zhou Z, Yao W, et al. SHMT1 inhibits the metastasis of HCC by repressing NOX1-mediated ROS production. J Exp Clin Cancer Res. 2019;38:70.
Yang Y, Zhang M, Zhao Y, Deng T, Zhou X, Qian H, et al. HOXD8 suppresses renal cell carcinoma growth by upregulating SHMT1 expression. Cancer Sci. 2023;114:4583–95.
Macfarlane AJ, Perry CA, McEntee MF, Lin DM, Stover PJ. Shmt1 heterozygosity impairs folate-dependent thymidylate synthesis capacity and modifies risk of Apc(min)-mediated intestinal cancer risk. Cancer Res. 2011;71:2098–107.
Wu X, Xu W, Zhou T, Cao N, Ni J, Zou T, et al. The Role of Genetic Polymorphisms as Related to One-Carbon Metabolism, Vitamin B6, and Gene-Nutrient Interactions in Maintaining Genomic Stability and Cell Viability in Chinese Breast Cancer Patients. Int J Mol Sci. 2016;17:2003.
Wang L, Lu J, An J, Shi Q, Spitz MR, Wei Q. Polymorphisms of cytosolic serine hydroxymethyltransferase and risk of lung cancer: a case-control analysis. Lung Cancer. 2007;57:143–51.
Komlósi V, Hitre E, Pap E, Adleff V, Réti A, Székely E, et al. SHMT1 1420 and MTHFR 677 variants are associated with rectal but not colon cancer. BMC Cancer. 2010;10:525.
Skibola CF, Smith MT, Hubbard A, Shane B, Roberts AC, Law GR, et al. Polymorphisms in the thymidylate synthase and serine hydroxymethyltransferase genes and risk of adult acute lymphocytic leukemia. Blood. 2002;99:3786–91.
Hishida A, Matsuo K, Hamajima N, Ito H, Ogura M, Kagami Y, et al. Associations between polymorphisms in the thymidylate synthase and serine hydroxymethyltransferase genes and susceptibility to malignant lymphoma. Haematologica. 2003;88:159–66.
Winkler F, Kriebel M, Clever M, Gröning S, Großhans J. Essential Function of the Serine Hydroxymethyl Transferase (SHMT) Gene During Rapid Syncytial Cell Cycles in Drosophila. G3 (Bethesda). 2017;7:2305–14.
Pilesi E, Angioli C, Graziani C, Parroni A, Contestabile R, Tramonti A, et al. A gene-nutrient interaction between vitamin B6 and serine hydroxymethyltransferase (SHMT) affects genome integrity in Drosophila. J Cell Physiol. 2023;238:1558–66.
Karim FD, Rubin GM. Ectopic expression of activated Ras1 induces hyperplastic growth and increased cell death in Drosophila imaginal tissues. Development. 1998;125:1–9.
Woodhouse E, Hersperger E, Shearn A. Growth, metastasis, and invasiveness of Drosophila tumors caused by mutations in specific tumor suppressor genes. Dev Genes Evol. 1998;207:542–50.
Willecke M, Toggweiler J, Basler K. Loss of PI3K blocks cell-cycle progression in a Drosophila tumor model. Oncogene. 2011;30:4067–74.
Manent J, Banerjee S, de Matos Simoes R, Zoranovic T, Mitsiades C, Penninger JM, et al. Autophagy suppresses Ras-driven epithelial tumourigenesis by limiting the accumulation of reactive oxygen species. Oncogene. 2017;36:5576–92.
Pilesi E, Tesoriere G, Ferriero A, Mascolo E, Liguori F, Argirò L, et al. Vitamin B6 deficiency cooperates with oncogenic Ras to induce malignant tumors in Drosophila. Cell Death Dis. 2024;15:388.
Pagliarini RA, Xu T. A genetic screen in Drosophila for metastatic behavior. Science. 2003;302:1227–31.
Tramonti A, Cuyàs E, Encinar JA, Pietzke M, Paone A, Verdura S, et al. Metformin is a pyridoxal-5′-phosphate (PLP)-competitive inhibitor of SHMT2. Cancers. 2021;13:4009.
Hirabayashi S, Baranski TJ, Cagan RL. Transformed Drosophila Cells Evade Diet-Mediated Insulin Resistance Through Wingless Signaling. Cell. 2013;154:664.
Natarajan AT, Obe G. Molecular mechanisms involved in the production of chromosomal aberrations. III. Restriction endonucleases. Chromosoma. 1984;90:120–7.
Bonassi S, Znaor A, Norppa H, Hagmar L. Chromosomal aberrations and risk of cancer in humans: an epidemiologic perspective. Cytogenet Genome Res. 2004;104:376–82.
Yagüe-Capilla M, Rudd SG. Understanding the interplay between dNTP metabolism and genome stability in cancer. DMM Disease Models and Mechanisms. 2024;17:dmm050775.
Ozer U, Barbour KW, Clinton SA, Berger FG. Oxidative Stress and Response to Thymidylate Synthase-Targeted Antimetabolites. Mol Pharmacol. 2015;88:970–81.
Merigliano C, Marzio A, Renda F, Somma MP, Gatti M, Vernì F. A Role for the Twins Protein Phosphatase (PP2A-B55) in the Maintenance of Drosophila Genome Integrity. Genetics. 2017;205:1151–67.
YOUNG CW, HODAS S. Hydroxyurea: Inhibitory Effect On Dna Metabolism. Science. 1964;146:1172–4.
Ezeriņa D, Takano Y, Hanaoka K, Urano Y, Dick TP. N-Acetyl Cysteine Functions as a Fast-Acting Antioxidant by Triggering Intracellular H 2 S and Sulfane Sulfur Production. Cell Chem Biol. 2018;25:447–459.e4.
Marzio A, Merigliano C, Gatti M, Vernì F. Sugar and chromosome stability: clastogenic effects of sugars in vitamin B6-deficient cells. PLoS Genet. 2014;10:e1004199.
Tesoriere G, Pilesi E, De Rosa M, Giampaoli O, Patriarca A, Spagnoli M, et al. Vitamin B6 deficiency produces metabolic alterations in Drosophila. Metabolomics. 2025;21:42.
Sonoshita M, Cagan RL. Modeling Human Cancers in Drosophila. Curr Top Dev Biol. 2017;121:287–309.
Bilder D. Epithelial polarity and proliferation control: links from the Drosophila neoplastic tumor suppressors. Genes Dev. 2004;18:1909–25.
Herbig K, Chiang EP, Lee LR, Hills J, Shane B, Stover PJ. Cytoplasmic serine hydroxymethyltransferase mediates competition between folate-dependent deoxyribonucleotide and S-adenosylmethionine biosyntheses. J Biol Chem. 2002;277:38381–9.
Mognato M, Burdak-Rothkamm S, Rothkamm K. Interplay between DNA replication stress, chromatin dynamics and DNA-damage response for the maintenance of genome stability. Mutat Res Rev Mutat Res. 2021;787:108346.
Ma N, Wang Y, Li X, Xu M, Tan D. Reactive oxygen species in cancer: Mechanistic insights and therapeutic innovations. Cell Stress Chaperones. 2025;30:100108.
Hosea R, Hillary S, Naqvi S, Wu S, Kasim V. The two sides of chromosomal instability: drivers and brakes in cancer. Signal Transduct Target Ther. 2024;9:75.
Yang X, Wu H. RAS signaling in carcinogenesis, cancer therapy and resistance mechanisms. J Hematol Oncol. 2024;17:108.
Prior IA, Hood FE, Hartley JL. The Frequency of Ras Mutations in Cancer. Cancer Res [Internet]. 2020;80:2669–974.
Chen S, Xu H, Li N, Yang Y, Pang R, Zhang S, et al. The scaffold protein DLG4 facilitates RNF63-mediated ubiquitination and degradation of STAT3 in non-small cell lung cancer. Cell Commun Signal. 2025;23:325.
Lin HT, Steller MA, Aish L, Hanada T, Chishti AH. Differential expression of human Dlg in cervical intraepithelial neoplasias. Gynecol Oncol. 2004;93:422–8.
Song Xqiu, Li Q, Zhang J. A double-edged sword: DLG5 in diseases. Biomedicine and Pharmacotherapy. 2023;162:114611.
Perry C, Yu S, Chen J, Matharu KS, Stover PJ. Effect of vitamin B6 availability on serine hydroxymethyltransferase in MCF-7 cells. Arch Biochem Biophys. 2007;462:21–7.
Niraula P, Kim MS. extends lifespan of Drosophila via modulating ROS scavenger gene expression. Biogerontology. 2019;20:533–43.
Slack C, Foley A, Partridge L. Activation of AMPK by the putative dietary restriction mimetic metformin is insufficient to extend lifespan in Drosophila. PLoS One. 2012;7:e47699.
Zhao H, Kalivendi S, Zhang H, Joseph J, Nithipatikom K, Vásquez-Vivar J, et al. Superoxide reacts with hydroethidine but forms a fluorescent product that is distinctly different from ethidium: potential implications in intracellular fluorescence detection of superoxide. Free Radic Biol Med. 2003;34:1359–68.
Acknowledgements
We are grateful to Prof. K. Basler, Prof. S. Hirabayashi, and Prof. T. Xu for generous sharing of fly stocks. We acknowledge BDSC and VDRC for fly stocks.
Funding
This research was supported by grants from Sapienza University of Rome: RD12318A998C70B8 (BBCD Department project); AR1231889041EBDD to EP; AR1241905DD95C63 to GT; RP12419058A651D4 to FV.
Author information
Authors and Affiliations
Contributions
CA, AF, EP, GT, and BA performed the experiments. AT performed the experiments, analyzed, and interpreted the data. RC analyzed and interpreted the data. FV designed the project, analyzed the data, and wrote the paper. All authors read and approved the final paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Edited by Dr. Kim McCall
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Angioli, C., Ferriero, A., Eleonora, P. et al. Tumor suppressor function of SHMT in a Drosophila RasV12DlgRNAi model: DNA damage and synergistic gene-nutrient interaction with PLP. Cell Death Dis (2026). https://doi.org/10.1038/s41419-026-08602-7
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41419-026-08602-7