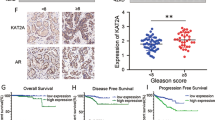
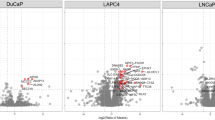
Abstract
In the treatment of locally advanced prostate cancer (PCa), abiraterone acetate (AA) serves both as a commonly used therapeutic agent and a radiosensitizer when combined with radiation therapy (RT). However, this combination therapy is not effective for all patients, and prolonged treatment may lead to decreased therapeutic sensitivity. Our study found that the combination of abiraterone (Abi, the active component of abiraterone acetate in vivo) and RT increases the expression of CtBP-interacting protein (CtIP) in prostate cancer cells, and elevated CtIP levels in PCa are associated with poor prognosis. CtIP has been demonstrated to be a key protein in the homologous recombination repair (HR) pathway of DNA damage repair (DDR). Furthermore, we observed that both Abi and RT enhance the transcriptional activity of CREB1 via phosphorylation, thereby modulating CtIP expression. Additionally, during the transition from normal prostate cells to prostate cancer cells, DNA demethylases (TETs) reduce DNA methylation levels in the promoter region of CtIP, facilitating the binding of CREB1 to the CtIP promoter. Finally, our in vitro and in vivo experiments indicate that the CREB1 phosphorylation inhibitor 666-15 significantly enhances the therapeutic efficacy of Abi-RT combination therapy. In summary, our study reveals that inhibition of the CREB1-CtIP axis effectively improves the therapeutic outcomes of Abi-RT combination therapy, which may offer a novel clinical strategy for the treatment of prostate cancer.
Similar content being viewed by others
Data availability
The raw data generated in this study are available upon request from the corresponding author.
References
Chang AJ, Autio KA, Roach M 3rd, Scher HI. High-risk prostate cancer-classification and therapy. Nat Rev Clin Oncol. 2014;11:308–23.
Diao X, Guo C, Jin Y, Li B, Gao X, Du X, et al. Cancer situation in China: an analysis based on the global epidemiological data released in 2024. Cancer Commun. 2025;45:178–97.
Schatten H. Brief overview of prostate cancer statistics, grading, diagnosis and treatment strategies. Adv Exp Med Biol. 2018;1095:1–14.
Singer EA, Golijanin DJ, Miyamoto H, Messing EM. Androgen deprivation therapy for prostate cancer. Expert Opin Pharmacother. 2008;9:211–28.
Voog JC, Smith MR, Efstathiou JA. Radiation with or without androgen deprivation therapy for localized prostate cancer. JAMA. 2016;315:1054–5.
Thakur A, Roy A, Ghosh A, Chhabra M, Banerjee S. Abiraterone acetate in the treatment of prostate cancer. Biomed Pharmacother. 2018;101:211–8.
Allen C, Her S, Jaffray DA. Radiotherapy for cancer: present and future. Adv Drug Deliv Rev. 2017;109:1–2.
Ryan ST, Sammon JD. Prostate cancer: radiotherapy might improve survival, even in the oldest men. Nat Rev Urol. 2016;13:444–5.
Wallis CJD, Saskin R, Choo R, Herschorn S, Kodama RT, Satkunasivam R, et al. Surgery versus radiotherapy for clinically-localized prostate cancer: a systematic review and meta-analysis. Eur Urol. 2016;70:21–30.
Huang RX, Zhou PK. DNA damage response signaling pathways and targets for radiotherapy sensitization in cancer. Signal Transduct Target Ther. 2020;5:60.
Santivasi WL, Xia F. Ionizing radiation-induced DNA damage, response, and repair. Antioxid Redox Signal. 2014;21:251–9.
Polkinghorn WR, Parker JS, Lee MX, Kass EM, Spratt DE, Iaquinta PJ, et al. Androgen receptor signaling regulates DNA repair in prostate cancers. Cancer Discov. 2013;3:1245–53.
Chou FJ, Chen Y, Chen D, Niu Y, Li G, Keng P, et al. Preclinical study using androgen receptor (AR) degradation enhancer to increase radiotherapy efficacy via targeting radiation-increased AR to better suppress prostate cancer progression. eBioMedicine. 2019;40:504–16.
Chatterjee N, Walker GC. Mechanisms of DNA damage, repair, and mutagenesis. Environ Mol Mutagen. 2017;58:235–63.
Cornford P, van den Bergh RCN, Briers E, Van den Broeck T, Brunckhorst O, Darraugh J, et al. EAU-EANM-ESTRO-ESUR-ISUP-SIOG guidelines on prostate cancer-2024 update. Part I: screening, diagnosis, and local treatment with curative intent. Eur Urol. 2024;86:148–63.
Kanesvaran R, Castro E, Wong A, Fizazi K, Chua MLK, Zhu Y, et al. Pan-Asian adapted ESMO Clinical Practice Guidelines for the diagnosis, treatment and follow-up of patients with prostate cancer. ESMO Open. 2022;7:100518.
Schaeffer EM, Srinivas S, Adra N, An Y, Bitting R, Chapin B, et al. NCCN Guidelines(R) insights: prostate cancer, version 3.2024. J Natl Compr Cancer Netw. 2024;22:140–50.
Tilki D, van den Bergh RCN, Briers E, Van den Broeck T, Brunckhorst O, Darraugh J, et al. EAU-EANM-ESTRO-ESUR-ISUP-SIOG guidelines on prostate cancer. Part II-2024 update: treatment of relapsing and metastatic prostate cancer. Eur Urol. 2024;86:164–82.
Francolini G, Allegra AG, Detti B, Di Cataldo V, Caini S, Bruni A, et al. Stereotactic body radiation therapy and abiraterone acetate for patients affected by oligometastatic castrate-resistant prostate cancer: a randomized phase II trial (ARTO). J Clin Oncol. 2023;41:5561–8.
Boeve LMS, Hulshof M, Vis AN, Zwinderman AH, Twisk JWR, Witjes WPJ, et al. Effect on survival of androgen deprivation therapy alone compared to androgen deprivation therapy combined with concurrent radiation therapy to the prostate in patients with primary bone metastatic prostate cancer in a prospective randomised clinical trial: data from the HORRAD trial. Eur Urol. 2019;75:410–8.
Smith M, Parker C, Saad F, Miller K, Tombal B, Ng QS, et al. Addition of radium-223 to abiraterone acetate and prednisone or prednisolone in patients with castration-resistant prostate cancer and bone metastases (ERA 223): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol. 2019;20:408–19.
Fizazi K, Foulon S, Carles J, Roubaud G, McDermott R, Flechon A, et al. Abiraterone plus prednisone added to androgen deprivation therapy and docetaxel in de novo metastatic castration-sensitive prostate cancer (PEACE-1): a multicentre, open-label, randomised, phase 3 study with a 2 x 2 factorial design. Lancet. 2022;399:1695–707.
Liu Y, Long W, Zhang Z, Mai L, Huang S, Liu B, et al. Cytoreductive radiotherapy combined with abiraterone in metastatic castration-resistance prostate cancer: a single center experience. Radiat Oncol. 2021;16:5.
Shaywitz AJ, Greenberg ME. CREB: a stimulus-induced transcription factor activated by a diverse array of extracellular signals. Annu Rev Biochem. 1999;68:821–61.
Lonze BE, Ginty DD. Function and regulation of CREB family transcription factors in the nervous system. Neuron. 2002;35:605–23.
Mayr B, Montminy M. Transcriptional regulation by the phosphorylation-dependent factor CREB. Nat Rev Mol Cell Biol. 2001;2:599–609.
Wu D, Zhau HE, Huang WC, Iqbal S, Habib FK, Sartor O, et al. cAMP-responsive element-binding protein regulates vascular endothelial growth factor expression: implication in human prostate cancer bone metastasis. Oncogene. 2007;26:5070–7.
Deng X, Liu H, Huang J, Cheng L, Keller ET, Parsons SJ, et al. Ionizing radiation induces prostate cancer neuroendocrine differentiation through interplay of CREB and ATF2: implications for disease progression. Cancer Res. 2008;68:9663–70.
Pan W, Zhang Z, Kimball H, Qu F, Berlind K, Stopsack KH, et al. Abiraterone acetate induces CREB1 phosphorylation and enhances the function of the CBP-p300 complex, leading to resistance in prostate cancer cells. Clin Cancer Res. 2021;27:2087–99.
Makharashvili N, Paull TT. CtIP: a DNA damage response protein at the intersection of DNA metabolism. DNA Repair. 2015;32:75–81.
Andres SN, Williams RS. CtIP/Ctp1/Sae2, molecular form fit for function. DNA Repair. 2017;56:109–17.
Deshpande RA, Myler LR, Soniat MM, Makharashvili N, Lee L, Lees-Miller SP, et al. DNA-dependent protein kinase promotes DNA end processing by MRN and CtIP. Sci Adv. 2020;6:eaay0922.
Barrett T, Wilhite SE, Ledoux P, Evangelista C, Kim IF, Tomashevsky M, et al. NCBI GEO: archive for functional genomics data sets-update. Nucleic Acids Res. 2013;41:D991–5.
Shen WK, Chen SY, Gan ZQ, Zhang YZ, Yue T, Chen MM, et al. AnimalTFDB 4.0: a comprehensive animal transcription factor database updated with variation and expression annotations. Nucleic Acids Res. 2023;51:D39–D45.
Cancer Genome Atlas Research N. The molecular taxonomy of primary prostate cancer. Cell. 2015;163:1011–25.
Mei S, Qin Q, Wu Q, Sun H, Zheng R, Zang C, et al. Cistrome Data Browser: a data portal for ChIP-Seq and chromatin accessibility data in human and mouse. Nucleic Acids Res. 2017;45:D658–D62.
Zheng R, Wan C, Mei S, Qin Q, Wu Q, Sun H, et al. Cistrome Data Browser: expanded datasets and new tools for gene regulatory analysis. Nucleic Acids Res. 2019;47:D729–D35.
Lee BT, Barber GP, Benet-Pages A, Casper J, Clawson H, Diekhans M, et al. The UCSC Genome Browser database: 2022 update. Nucleic Acids Res. 2022;50:D1115–D22.
Tang Z, Kang B, Li C, Chen T, Zhang Z. GEPIA2: an enhanced web server for large-scale expression profiling and interactive analysis. Nucleic Acids Res. 2019;47:W556–W60.
Subramanian A, Tamayo P, Mootha VK, Mukherjee S, Ebert BL, Gillette MA, et al. Gene set enrichment analysis: a knowledge-based approach for interpreting genome-wide expression profiles. Proc Natl Acad Sci USA. 2005;102:15545–50.
Gene Ontology C. Gene ontology consortium: going forward. Nucleic Acids Res. 2015;43:D1049–56.
Chen EY, Tan CM, Kou Y, Duan Q, Wang Z, Meirelles GV, et al. Enrichr: interactive and collaborative HTML5 gene list enrichment analysis tool. BMC Bioinform. 2013;14:128.
Kuleshov MV, Jones MR, Rouillard AD, Fernandez NF, Duan Q, Wang Z, et al. Enrichr: a comprehensive gene set enrichment analysis web server 2016 update. Nucleic Acids Res. 2016;44:W90–7.
Xie Z, Bailey A, Kuleshov MV, Clarke DJB, Evangelista JE, Jenkins SL, et al. Gene set knowledge discovery with enrichr. Curr Protoc. 2021;1:e90.
Agrawal A, Balci H, Hanspers K, Coort SL, Martens M, Slenter DN, et al. WikiPathways 2024: next generation pathway database. Nucleic Acids Res. 2024;52:D679–D89.
Uhlen M, Zhang C, Lee S, Sjostedt E, Fagerberg L, Bidkhori G, et al. A pathology atlas of the human cancer transcriptome. Science. 2017;357:eaan2507.
Dyer M, Lin QXX, Shapoval S, Thieffry D, Benoukraf T. MethMotif.Org 2024: a database integrating context-specific transcription factor-binding motifs with DNA methylation patterns. Nucleic Acids Res. 2024;52:D222–D8.
Ovek Baydar D, Rauluseviciute I, Aronsen DR, Blanc-Mathieu R, Bonthuis I, de Beukelaer H, et al. JASPAR 2026: expansion of transcription factor binding profiles and integration of deep learning models. Nucleic Acids Res. 2026, 54:D184-D193.
Chandrashekar DS, Bashel B, Balasubramanya SAH, Creighton CJ, Ponce-Rodriguez I, Chakravarthi B, et al. UALCAN: a portal for facilitating tumor subgroup gene expression and survival analyses. Neoplasia. 2017;19:649–58.
Chandrashekar DS, Karthikeyan SK, Korla PK, Patel H, Shovon AR, Athar M, et al. UALCAN: An update to the integrated cancer data analysis platform. Neoplasia. 2022;25:18–27.
Ianevski A, Giri AK, Aittokallio T. SynergyFinder 3.0: an interactive analysis and consensus interpretation of multi-drug synergies across multiple samples. Nucleic Acids Res. 2022;50:W739–W43.
Arnoult N, Correia A, Ma J, Merlo A, Garcia-Gomez S, Maric M, et al. Regulation of DNA repair pathway choice in S and G2 phases by the NHEJ inhibitor CYREN. Nature. 2017;549:548–52.
Qin C, Wang YL, Zhou JY, Shi J, Zhao WW, Zhu YX, et al. RAP80 phase separation at DNA double-strand break promotes BRCA1 recruitment. Nucleic Acids Res. 2023;51:9733–47.
You Z, Bailis JM. DNA damage and decisions: CtIP coordinates DNA repair and cell cycle checkpoints. Trends Cell Biol. 2010;20:402–9.
Zhang H, Liu Y, Liu J, Chen J, Wang J, Hua H, et al. cAMP-PKA/EPAC signaling and cancer: the interplay in tumor microenvironment. J Hematol Oncol. 2024;17:5.
Devaiah BN, Singh AK, Mu J, Chen Q, Meerzaman D, Singer DS. Phosphorylation by JNK switches BRD4 functions. Mol Cell. 2024;84:4282–96 e7.
Mao C, Ray-Gallet D, Tavitian A, Moreau-Gachelin F. Differential phosphorylations of Spi-B and Spi-1 transcription factors. Oncogene. 1996;12:863–73.
De Cesare D, Fimia GM, Sassone-Corsi P. Signaling routes to CREM and CREB: plasticity in transcriptional activation. Trends Biochem Sci. 1999;24:281–5.
Wang Y, Hu Z, Liu Z, Chen R, Peng H, Guo J, et al. MTOR inhibition attenuates DNA damage and apoptosis through autophagy-mediated suppression of CREB1. Autophagy. 2013;9:2069–86.
Zhang X, Odom DT, Koo SH, Conkright MD, Canettieri G, Best J, et al. Genome-wide analysis of cAMP-response element binding protein occupancy, phosphorylation, and target gene activation in human tissues. Proc Natl Acad Sci USA. 2005;102:4459–64.
Kim SH, Trinh AT, Larsen MC, Mastrocola AS, Jefcoate CR, Bushel PR, et al. Tunable regulation of CREB DNA binding activity couples genotoxic stress response and metabolism. Nucleic Acids Res. 2016;44:9667–80.
Kaluscha S, Domcke S, Wirbelauer C, Stadler MB, Durdu S, Burger L, et al. Evidence that direct inhibition of transcription factor binding is the prevailing mode of gene and repeat repression by DNA methylation. Nat Genet. 2022;54:1895–906.
Liu D, Li G, Zuo Y. Function determinants of TET proteins: the arrangements of sequence motifs with specific codes. Brief Bioinform. 2019;20:1826–35.
Liu W, Wu G, Xiong F, Chen Y. Advances in the DNA methylation hydroxylase TET1. Biomark Res. 2021;9:76.
Nickerson ML, Das S, Im KM, Turan S, Berndt SI, Li H, et al. TET2 binds the androgen receptor and loss is associated with prostate cancer. Oncogene. 2017;36:2172–83.
Yu G, Bao J, Zhan M, Wang J, Li X, Gu X, et al. Comprehensive analysis of m5C methylation regulatory genes and tumor microenvironment in prostate cancer. Front Immunol. 2022;13:914577.
Mozaffari NL, Pagliarulo F, Sartori AA. Human CtIP: A ‘double agent’ in DNA repair and tumorigenesis. Semin Cell Dev Biol. 2021;113:47–56.
Soria-Bretones I, Saez C, Ruiz-Borrego M, Japon MA, Huertas P. Prognostic value of CtIP/RBBP8 expression in breast cancer. Cancer Med. 2013;2:774–83.
Wu M, Soler DR, Abba MC, Nunez MI, Baer R, Hatzis C, et al. CtIP silencing as a novel mechanism of tamoxifen resistance in breast cancer. Mol Cancer Res. 2007;5:1285–95.
Mijnes J, Veeck J, Gaisa NT, Burghardt E, de Ruijter TC, Gostek S, et al. Promoter methylation of DNA damage repair (DDR) genes in human tumor entities: RBBP8/CtIP is almost exclusively methylated in bladder cancer. Clin Epigenetics. 2018;10:15.
Eke I, Bylicky MA, Sandfort V, Chopra S, Martello S, Graves EE, et al. The lncRNAs LINC00261 and LINC00665 are upregulated in long-term prostate cancer adaptation after radiotherapy. Mol Ther Nucleic Acids. 2021;24:175–87.
Han J, Wan L, Jiang G, Cao L, Xia F, Tian T, et al. ATM controls the extent of DNA end resection by eliciting sequential posttranslational modifications of CtIP. Proc Natl Acad Sci USA. 2021;118:e2022600118.
You Z, Shi LZ, Zhu Q, Wu P, Zhang YW, Basilio A, et al. CtIP links DNA double-strand break sensing to resection. Mol Cell. 2009;36:954–69.
Dibitetto D, Liptay M, Vivalda F, Dogan H, Gogola E, Gonzalez Fernandez M, et al. H2AX promotes replication fork degradation and chemosensitivity in BRCA-deficient tumours. Nat Commun. 2024;15:4430.
Jeong SY, Hariharasudhan G, Kim MJ, Lim JY, Jung SM, Choi EJ, et al. SIAH2 regulates DNA end resection and replication fork recovery by promoting CtIP ubiquitination. Nucleic Acids Res. 2022;50:10469–86.
Yin Y, Lin J, Cai X, Nie R, Lin J, Li S, et al. Phase separation of ERCC6L2-CtIP regulates the extent of DNA end resection. Nat Cell Biol. 2025;27:1771–84.
Gao M, Guo G, Huang J, Kloeber JA, Zhao F, Deng M, et al. USP52 regulates DNA end resection and chemosensitivity through removing inhibitory ubiquitination from CtIP. Nat Commun. 2020;11:5362.
Ding D, Zheng R, Tian Y, Jimenez R, Hou X, Weroha SJ, et al. Retinoblastoma protein as an intrinsic BRD4 inhibitor modulates small molecule BET inhibitor sensitivity in cancer. Nat Commun. 2022;13:6311.
Acknowledgements
The authors thank public data of TCGA, JASPAR, WikiPathways, Enrichr, Human TFDB, GEPIA, UCSC and Cistromedb. We thank Dr. Wan (wanxbo@zzu.edu.cn) from The First Affiliated Hospital of Zhengzhou University for gifting the plasmids and Hela cell.
Funding
This work was supported by National Natural Science Foundation of China (grant number: 882172564, 82303032, 82573338), Henan Science and Technology Research Program (grant number: 2018020142) and the Key Laboratory of Urinary Tumors, Henan Provincial Health Commission, Zhengzhou, Henan, China.
Author information
Authors and Affiliations
Contributions
XH and ZLH conceived and designed the study. XH, LS, and YKF performed experiments and drafted the manuscript. XH, ZHW, YL, and FBL performed experiments and analyzed the data. LS, LNW, and YL performed the statistical analysis. RYL, NYL, and SYM provided technical and methodological support. JJY supervised the study. ZKJ and ZLH reviewed and revised the manuscript. All authors read and approved the final version.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
Approval was obtained for all animal studies under the guidelines of the First Affiliated Hospital of Zhengzhou University. All clinical study was approved by the Ethics Committee of the First Affiliated Hospital of Zhengzhou University and written informed consent was obtained from all subjects. Ethics approval number: 2024-KY-1207-001.
Consent for publication
All co-authors have consented to the version of the manuscript for publication.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Edited by Professor Boris Zhivotovsky
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Han, X., Song, L., Feng, Y. et al. Targeted inhibition of the CREB1-CtIP axis enhances the efficacy of abiraterone combined with radiotherapy in prostate cancer. Cell Death Dis (2026). https://doi.org/10.1038/s41419-026-08633-0
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41419-026-08633-0