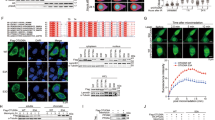
Abstract
Breast cancer (BC) remains the leading cause of global female cancer-related mortality, with poor survival in advanced stages driven largely by metastasis. Ubiquitination, a key post-translational modification, critically regulates the stability and function of various proteins, including oncoproteins and tumor suppressors, and deubiquitinases (DUBs) reversing this process are emerging therapeutic targets. In this study, we report that haploid germ cell-specific nuclear protein kinase (HASPIN) is highly expressed in BC and is closely associated with poor prognosis. We identify the DUB Otubain-2 (OTUB2) as a critical regulator of the oncogenic kinase HASPIN in BC. We demonstrate that OTUB2 binds to and deubiquitylates HASPIN, specifically counteracting its K48-linked polyubiquitination and subsequent proteasomal degradation. Acetylation of HASPIN at lysine 751 by acetyltransferase lysine acetyltransferase 5 (KAT5) enhances its affinity for OTUB2, promoting HASPIN stability. Functionally, OTUB2 depletion reduces HASPIN protein levels, while OTUB2 overexpression-induced HASPIN upregulation drives BC cell proliferation and invasion both in vivo and in vitro. These findings establish OTUB2 as a novel DUB for HASPIN and reveal a previously unknown regulatory axis involving KAT5, acetylation, OTUB2, ubiquitination, and HASPIN, which is crucial for BC progression. Consequently, HASPIN acts as an oncogene in BC and represents a promising new therapeutic target for intervention.
Similar content being viewed by others
Data availability
The data of LC-MS/MS are provided in the paper and its Supplementary Information.
References
Bray F, Laversanne M, Sung H, Ferlay J, Siegel RL, Soerjomataram I, et al. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2024;74:229–63.
Xia C, Dong X, Li H, Cao M, Sun D, He S, et al. Cancer statistics in China and United States, 2022: profiles, trends, and determinants. Chin Med J. 2022;135:584–90.
Jazieh K, Bell R, Agarwal N, Abraham J. Novel targeted therapies for metastatic breast cancer. Ann Transl Med. 2020;8:907.
Street W. Cancer facts & figures 2019. Atlanta, GA, USA: American Cancer Society; 2019.
Ganesh K, Massagué J. Targeting metastatic cancer. Nat Med. 2021;27:34–44.
Zhang B, Whiteaker JR, Hoofnagle AN, Baird GS, Rodland KD, Paulovich AG. Clinical potential of mass spectrometry-based proteogenomics. Nat Rev Clin Oncol. 2019;16:256–68.
Cockram PE, Kist M, Prakash S, Chen SH, Wertz IE, Vucic D. Ubiquitination in the regulation of inflammatory cell death and cancer. Cell Death Differ. 2021;28:591–605.
Loix M, Zelcer N, Bogie JFJ, Hendriks JJA. The ubiquitous role of ubiquitination in lipid metabolism. Trends Cell Biol. 2024;34:416–29.
Mevissen TET, Komander D. Mechanisms of deubiquitinase specificity and regulation. Annu Rev Biochem. 2017;86:159–92.
Clague MJ, Barsukov I, Coulson JM, Liu H, Rigden DJ, Urbé S. Deubiquitylases from genes to organism. Physiol Rev. 2013;93:1289–315.
D’Andrea A, Pellman D. Deubiquitinating enzymes: a new class of biological regulators. Crit Rev Biochem Mol Biol. 1998;33:337–52.
Komander D, Clague MJ, Urbé S. Breaking the chains: structure and function of the deubiquitinases. Nat Rev Mol Cell Biol. 2009;10:550–63.
Samy MA, Abd El Fatah NM, Yahia SE, Arafa RK. Friend or foe: UCHL3 mediated carcinogenesis and current approaches in small molecule inhibitors’ development. Curr Med Chem. 2021;28:8782–99.
Zheng LL, Wang LT, Pang YW, Sun LP, Shi L. Recent advances in the development of deubiquitinases inhibitors as antitumor agents. Eur J Med Chem. 2024;266:116161.
D’Arcy P, Wang X, Linder S. Deubiquitinase inhibition as a cancer therapeutic strategy. Pharm Ther. 2015;147:32–54.
Mevissen TE, Hospenthal MK, Geurink PP, Elliott PR, Akutsu M, Arnaudo N, et al. OTU deubiquitinases reveal mechanisms of linkage specificity and enable ubiquitin chain restriction analysis. Cell. 2013;154:169–84.
Taylor SS, Kornev AP. Protein kinases: evolution of dynamic regulatory proteins. Trends Biochem Sci. 2011;36:65–77.
Villa F, Capasso P, Tortorici M, Forneris F, de Marco A, Mattevi A, et al. Crystal structure of the catalytic domain of Haspin, an atypical kinase implicated in chromatin organization. Proc Natl Acad Sci USA. 2009;106:20204–9.
Amoussou NG, Bigot A, Roussakis C, Robert J-MH. Haspin: a promising target for the design of inhibitors as potent anticancer drugs. Drug Discov Today. 2018;23:409–15.
Han X, Kuang T, Ren Y, Lu Z, Liao Q, Chen W. Haspin knockdown can inhibit progression and development of pancreatic cancer in vitro and vivo. Exp Cell Res. 2019;385:111605.
Chen Y, Fu D, Zhao H, Cheng W, Xu F. GSG2 (Haspin) promotes development and progression of bladder cancer through targeting KIF15 (Kinase-12). Aging. 2020;12:8858–79.
Zhou J, Nie W, Yuan J, Zhang Z, Mi L, Wang C, et al. GSG2 knockdown suppresses cholangiocarcinoma progression by regulating cell proliferation, apoptosis and migration. Oncol Rep. 2021;45:91.
Hadders MA, Hindriksen S, Truong MA, Mhaskar AN, Wopken JP, Vromans MJM, et al. Untangling the contribution of Haspin and Bub1 to Aurora B function during mitosis. J Cell Biol. 2020;219:e201907087.
Wang P, Hua X, Bryner YH, Liu S, Gitter CB, Dai J. Haspin inhibition delays cell cycle progression through interphase in cancer cells. J Cell Physiol. 2020;235:4508–19.
Tang Y, Dai G, Yang Y, Liu H. GSG2 facilitates the progression of human breast cancer through MDM2-mediated ubiquitination of E2F1. J Transl Med. 2023;21:523.
Quadri R, Sertic S, Muzi-Falconi M. Roles and regulation of Haspin kinase and its impact on carcinogenesis. Cell Signal. 2022;93:110303.
Zhu D, Gu X, Lin Z, Yu D, Wang J, Li L. HASPIN is involved in the progression of gallbladder carcinoma. Exp Cell Res. 2020;390:111863.
Huang Y, Liu Y, Zhu K, Ma X, Lu R, Zhang M. GSG2 Promotes development and predicts poor prognosis of ovarian cancer. Cancer Manag Res. 2021;13:499–508.
Dai J, Sullivan BA, Higgins JM. Regulation of mitotic chromosome cohesion by Haspin and Aurora B. Dev Cell. 2006;11:741–50.
Wang F, Ulyanova NP, van der Waal MS, Patnaik D, Lens SM, Higgins JM. A positive feedback loop involving Haspin and Aurora B promotes CPC accumulation at centromeres in mitosis. Curr Biol. 2011;21:1061–9.
Wang F, Dai J, Daum JR, Niedzialkowska E, Banerjee B, Stukenberg PT, et al. Histone H3 Thr-3 phosphorylation by Haspin positions Aurora B at centromeres in mitosis. Science. 2010;330:231–5.
Quadri R, Sertic S, Ghilardi A, Rondelli D, Gallo GR, Del Giacco L, et al. Phosphorylation of H3-Thr3 by Haspin is required for primary cilia regulation. Int J Mol Sci. 2021;22:7753.
Panigada D, Grianti P, Nespoli A, Rotondo G, Castro DG, Quadri R, et al. Yeast haspin kinase regulates polarity cues necessary for mitotic spindle positioning and is required to tolerate mitotic arrest. Dev Cell. 2013;26:483–95.
Quadri R, Galli M, Galati E, Rotondo G, Gallo GR, Panigada D, et al. Haspin regulates Ras localization to promote Cdc24-driven mitotic depolarization. Cell Discov. 2020;6:42.
Nanao MH, Tcherniuk SO, Chroboczek J, Dideberg O, Dessen A, Balakirev MY. Crystal structure of human otubain 2. EMBO Rep. 2004;5:783–8.
Li J, Zhang N, Li M, Hong T, Meng W, Ouyang T. The emerging role of OTUB2 in diseases: from cell signaling pathway to physiological function. Front Cell Dev Biol. 2022;10:820781.
Zhang Z, Du J, Wang S, Shao L, Jin K, Li F, et al. OTUB2 promotes cancer metastasis via hippo-independent activation of YAP and TAZ. Mol Cell. 2019;73:7–21.e7.
Ma Y, Sun Y. miR-29a-3p inhibits growth, proliferation, and invasion of papillary thyroid carcinoma by suppressing NF-κB signaling via direct targeting of OTUB2. Cancer Manag Res. 2019;11:13–23.
Gu ZL, Huang J, Zhen LL. Knockdown of otubain 2 inhibits liver cancer cell growth by suppressing NF-κB signaling. Kaohsiung J Med Sci. 2020;36:399–404.
Li J, Cheng D, Zhu M, Yu H, Pan Z, Liu L, et al. OTUB2 stabilizes U2AF2 to promote the Warburg effect and tumorigenesis via the AKT/mTOR signaling pathway in non-small cell lung cancer. Theranostics. 2019;9:179–95.
Xu X, Wu G, Han K, Cui X, Feng Y, Mei X, et al. Inhibition of OTUB2 suppresses colorectal cancer cell growth by regulating β-Catenin signaling. Am J Cancer Res. 2023;13:5382–93.
Hu G, Yang J, Zhang H, Huang Z, Yang H. OTUB2 promotes proliferation and migration of hepatocellular carcinoma cells by PJA1 deubiquitylation. Cell Mol Bioeng. 2022;15:281–92.
Liu L, Cheng H, Ji M, Su L, Lu Z, Hu X, et al. OTUB2 regulates YAP1/TAZ to promotes the progression of esophageal squamous cell carcinoma. Biol Proced Online. 2022;24:10.
Yu S, Zang W, Qiu Y, Liao L, Zheng X. Deubiquitinase OTUB2 exacerbates the progression of colorectal cancer by promoting PKM2 activity and glycolysis. Oncogene. 2022;41:46–56.
Zhu W, Wu C, Liu Z, Zhao S, Huang J. OTU deubiquitinase, ubiquitin aldehyde binding 2 (OTUB2) modulates the stemness feature, chemoresistance, and epithelial-mesenchymal transition of colon cancer via regulating GINS complex subunit 1 (GINS1) expression. Cell Commun Signal. 2024;22:420.
Nan Y, Wu X, Luo Q, Chang W, Zhao P, Zhang L, et al. OTUB2 silencing promotes ovarian cancer via mitochondrial metabolic reprogramming and can be synthetically targeted by CA9 inhibition. Proc Natl Acad Sci USA. 2024;121:e2315348121.
Chang W, Luo Q, Wu X, Nan Y, Zhao P, Zhang L, et al. OTUB2 exerts tumor-suppressive roles via STAT1-mediated CALML3 activation and increased phosphatidylserine synthesis. Cell Rep. 2022;41:111561.
Ren W, Xu Z, Chang Y, Ju F, Wu H, Liang Z, et al. Pharmaceutical targeting of OTUB2 sensitizes tumors to cytotoxic T cells via degradation of PD-L1. Nat Commun. 2024;15:9.
Liu P, Cong X, Liao S, Jia X, Wang X, Dai W, et al. Global identification of phospho-dependent SCF substrates reveals a FBXO22 phosphodegron and an ERK-FBXO22-BAG3 axis in tumorigenesis. Cell Death Differ. 2022;29:1–13.
Valencia-Sánchez MI, De Ioannes P, Wang M, Truong DM, Lee R, Armache JP, et al. Regulation of the Dot1 histone H3K79 methyltransferase by histone H4K16 acetylation. Science. 2021;371:eabc6663.
Liu C, Zhao H, Xiao S, Han T, Chen Y, Wang T, et al. Slx5p-Slx8p promotes accurate chromosome segregation by mediating the degradation of synaptonemal complex components during meiosis. Adv Sci (Weinh). 2020;7:1900739.
Ghosh A, Chakraborty P, Biswas D. Fine tuning of the transcription juggernaut: a sweet and sour saga of acetylation and ubiquitination. Biochim Biophys Acta Gene Regul Mech. 2023;1866:194944.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (No. 82203745 to JG), The Clinical Research Fund of Collaborative Innovation Center for Individualized Cancer Medicine jointly built by the Ministry and the Province (to MH).
Author information
Authors and Affiliations
Contributions
MH and WS conceived and designed the research; JG and KK conducted the experiment and drafted the paper; SW and ZJ interpreted the data and revised the paper. HL and YZ conducted data analysis.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Edited by George Calin
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Guo, J., Kang, K., Wang, S. et al. KAT5-mediated acetylation enhances the deubiquitination of HASPIN by OTUB2 and promotes breast cancer progression. Cell Death Dis (2026). https://doi.org/10.1038/s41419-026-08658-5
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41419-026-08658-5