Abstract
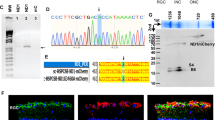
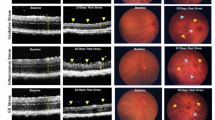
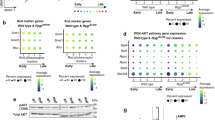
Early-onset inherited retinal degenerations (IRDs), such as Leber congenital amaurosis (LCA) caused by pathogenic variants in the NMNAT1 gene, lead to severe vision loss in children. Despite its ubiquitous expression, reduced NMNAT1 function primarily affects photoreceptor cells (PRs) of the retina, yet the mechanisms underlying their vulnerability remain incompletely understood. Here, we demonstrate that reduced NMNAT1 enzyme function due to the p.V9M mutation leads to DNA damage in PRs, characterized by the progressive accumulation of the oxidative DNA adduct 8-oxo-dG in Nmnat1V9M/V9M mutant mice. Cells with oxidative DNA damage also demonstrate DNA double-strand breaks, as evidenced by co-staining with antibodies to phosphorylated H2AX (γH2A.X). This DNA damage correlates with apoptosis-driven PR degeneration, as evidenced by caspase-9 activation and TUNEL staining in the PRs of the Nmnat1V9M/V9M mutant mice, while alternative cell death pathways such as necroptosis and parthanatos were not significantly activated. Treatment with the antioxidant N-acetylcysteine (NAC) reduced oxidative DNA damage and retinal immune responses, mitigated apoptosis, and preserved cone PRs. Longitudinal assessment via optical coherence tomography (OCT) and electroretinography (ERG) revealed sustained structural and functional protection of the retina in NAC-treated mice. These findings establish oxidative DNA damage as a key driver of PR degeneration in the Nmnat1V9M/V9M model and highlight NAC’s potential as a therapeutic strategy for NMNAT1-associated IRD and potentially other IRDs in which oxidative DNA damage contributes to disease pathogenesis.
Similar content being viewed by others
Data availability
All data relevant to the study are included in the article or uploaded as supplementary information. Source data are available upon reasonable request.
References
Sahel JA, Marazova K, Audo I. Clinical characteristics and current therapies for inherited retinal degenerations. Cold Spring Harb Perspect Med. 2015;5:a017111.
Bourne RRA, Flaxman SR, Braithwaite T, Cicinelli MV, Das A, Jonas JB, et al. Magnitude, temporal trends, and projections of the global prevalence of blindness and distance and near vision impairment: a systematic review and meta-analysis. Lancet Glob Health. 2017;5:e888–e897.
Falk MJ, Zhang Q, Nakamaru-Ogiso E, Kannabiran C, Fonseca-Kelly Z, Chakarova C, et al. NMNAT1 mutations cause Leber congenital amaurosis. Nat Genet. 2012;44:1040–5.
Perrault I, Hanein S, Zanlonghi X, Serre V, Nicouleau M, Defoort-Delhemmes S, et al. Mutations in NMNAT1 cause Leber congenital amaurosis with early-onset severe macular and optic atrophy. Nat Genet. 2012;44:975–7.
Chiang PW, Wang J, Chen Y, Fu Q, Zhong J, Chen Y, et al. Exome sequencing identifies NMNAT1 mutations as a cause of Leber congenital amaurosis. Nat Genet. 2012;44:972–4.
Koenekoop RK, Wang H, Majewski J, Wang X, Lopez I, Ren H, et al. Mutations in NMNAT1 cause Leber congenital amaurosis and identify a new disease pathway for retinal degeneration. Nat Genet. 2012;44:1035–9.
Sasaki Y, Margolin Z, Borgo B, Havranek JJ, Milbrandt J. Characterization of Leber Congenital Amaurosis-associated NMNAT1 mutants. J Biol Chem. 2015;290:17228–38.
Greenwald SH, Charette JR, Staniszewska M, Shi LY, Brown SDM, Stone L, et al. Mouse models of NMNAT1-Leber Congenital Amaurosis (LCA9) recapitulate key features of the human disease. Am J Pathol. 2016;186:1925.
Greenwald SH, Brown EE, Scandura MJ, Hennessey E, Farmer R, Du J, et al. Mutant Nmnat1 leads to a retina-specific decrease of NAD+ accompanied by increased poly(ADP-ribose) in a mouse model of NMNAT1-associated retinal degeneration. Hum Mol Genet. 2021;30:644–57.
Brown EE, Scandura MJ, Mehrotra S, Wang Y, Du J, Pierce EA. Reduced nuclear NAD+ drives DNA damage and subsequent immune activation in the retina. Hum Mol Genet. 2022;31:1370–88.
Lindahl T. Instability and decay of the primary structure of DNA. Nature. 1993;362:709–15.
Lindahl T, Barnes DE. Repair of endogenous DNA damage. Cold Spring Harb Symp Quant Biol. 2000;65:127–33.
Tubbs A, Nussenzweig A, Endogenous DNA. Damage as a source of genomic instability in cancer. Cell. 2017;168:644.
Chatterjee N, Walker GC. Mechanisms of DNA damage, repair and mutagenesis. Environ Mol Mutagen. 2017;58:235.
Hakem R. DNA-damage repair; the good, the bad, and the ugly. EMBO J. 2008;27:589.
Pan WW, Wubben TJ, Besirli CG. Photoreceptor metabolic reprogramming: current understanding and therapeutic implications. Commun Biol. 2021;4:245.
Okawa H, Sampath AP, Laughlin SB, Fain GL. ATP consumption by mammalian rod photoreceptors in darkness and in light. Curr Biol. 2008;18:1917–21.
Birol G, Wang S, Budzynski E, Wangsa-Wirawan ND, Linsenmeier RA. Oxygen distribution and consumption in the macaque retina. Am J Physiol Heart Circ Physiol. 2007;293:H1696–704.
Domènech EB, Marfany G. The relevance of oxidative stress in the pathogenesis and therapy of retinal dystrophies. Antioxidants. 2020;9:347.
Hagins WA, Penn RD, Yoshikami S. Dark current and photocurrent in retinal rods. Biophys J. 1970;10:380.
Gordon WC, Casey DM, Lukiw WJ, Bazan NG. DNA damage and repair in light-induced photoreceptor degeneration. Invest Opthalmol Vis Sci. 2002;43:3511–21.
Greenwald SH, Pierce EA. Parthanatos as a cell death pathway underlying retinal disease. Adv Exp Med Biol. 2019;1185:323–7.
Li M, Xu J, Wang Y, Du X, Zhang T, Chen Y, et al. Protects against photoreceptor degeneration in part through suppressing oxidative stress and DNA damage-induced necroptosis and inflammation in the retina. J Inflamm Res. 2022;15:2995–3020.
Dong K, Yan Y, Lu L, Wang Y, Li J, Zhang M, et al. PJ34 protects photoreceptors from cell death by inhibiting PARP-1 induced parthanatos after experimental retinal detachment. Curr Eye Res. 2021;46:115–21.
Yu Z, Correa VSMC, Efstathiou NE, Albertos-Arranz H, Chen X, Ishihara K, et al. UVA induces retinal photoreceptor cell death via receptor interacting protein 3 kinase mediated necroptosis. Cell Death Discov 2022 8:1. 2022;8:1–11.
Shi Y. Apoptosome: the cellular engine for the activation of caspase-9. Structure. 2002;10:285–8.
Andrabi SA, Dawson TM, Dawson VL. Mitochondrial and nuclear cross talk in cell death: parthanatos. Ann N Y Acad Sci. 2008;1147:233–41.
Wang Y, An R, Umanah GK, Park H, Nambiar K, Eacker SM, et al. A nuclease that mediates cell death induced by DNA damage and poly(ADP-ribose) polymerase-1. Science. 2016;354:aad6872.
Kaczmarek A, Vandenabeele P, Krysko DV. Necroptosis: the release of damage-associated molecular patterns and its physiological relevance. Immunity. 2013;38:209–23.
Biton S, Ashkenazi ANEMO. and RIP1 control cell fate in response to extensive DNA damage via TNF-α feedforward signaling. Cell. 2011;145:92–103.
Chen D, Tong J, Yang L, Wei L, Stolz DB, Yu J, et al. PUMA amplifies necroptosis signaling by activating cytosolic DNA sensors. Proc Natl Acad Sci USA. 2018;115:3930–5.
Hahm JY, Park J, Jang ES, Chi SW. 8-Oxoguanine: from oxidative damage to epigenetic and epitranscriptional modification. Exp Mol Med. 2022;54:1626–42.
Firsanov DV, Solovjeva LV, Svetlova MP. H2AX phosphorylation at the sites of DNA double-strand breaks in cultivated mammalian cells and tissues. Clin Epigenet. 2011;2:283.
Sano H, Namekata K, Kimura A, Shitara H, Guo X, Harada C, et al. Differential effects of N-acetylcysteine on retinal degeneration in two mouse models of normal tension glaucoma. Cell Death Dis. 2019;10:75.
Lee SY, Usui S, Zafar AB, Oveson BC, Jo YJ, Lu L, et al. N-Acetylcysteine promotes long-term survival of cones in a model of retinitis pigmentosa. J Cell Physiol. 2011;226:1843–9.
Greenwald SH, Charette JR, Staniszewska M, Shi LY, Brown SDM, Stone L, et al. Mouse models of NMNAT1-Leber congenital amaurosis recapitulate key features of the human disease. Invest Ophthalmol Vis Sci. 2016;57:2255–2255.
Brown EE, Scandura MJ, Pierce EA. Expression of NMNAT1 in the photoreceptors is sufficient to prevent NMNAT1-associated retinal degeneration. Mol Ther Methods Clin Dev. 2023;29:319–28.
Verhasselt V, Vanden Berghe W, Vanderheyde N, Willems F, Haegeman G, Goldman M. N-Acetyl-l-Cysteine inhibits primary human T cell responses at the dendritic cell level: association with NF-κB inhibition. J Immunol. 1999;162:2569–74.
Yoshida N, Ikeda Y, Notomi S, Ishikawa K, Murakami Y, Hisatomi T, et al. Laboratory evidence of sustained chronic inflammatory reaction in retinitis pigmentosa. Ophthalmology. 2013;120:e5–12.
Giordani L, Quaranta MG, Malorni W, Boccanera M, Giacomini E, Viora M. N-acetylcysteine inhibits the induction of an antigen-specific antibody response down-regulating CD40 and CD27 co-stimulatory molecules. Clin Exp Immunol. 2002;129:254–64.
Bonnaure G, Néron S. N-acetyl cysteine regulates the phosphorylation of JAK proteins following CD40-activation of human memory B cells. Mol Immunol. 2014;62:209–18.
Sakai M, Yu Z, Taniguchi M, Picotin R, Oyama N, Stellwagen D, et al. N-acetylcysteine suppresses microglial inflammation and induces mortality dose-dependently via tumor necrosis factor-α signaling. Int J Mol Sci. 2023;24:3798.
Chen H, Weber AJ. Expression of glial fibrillary acidic protein and glutamine synthetase by Müller cells after optic nerve damage and intravitreal application of brain-derived neurotrophic factor. Glia. 2002;38:115–25.
Imai Y, Ibata I, Ito D, Ohsawa K, Kohsaka S. A novel gene iba1 in the major histocompatibility complex class III region encoding an EF hand protein expressed in a monocytic lineage. Biochem Biophys Res Commun. 1996;224:855–62.
Ito D, Imai Y, Ohsawa K, Nakajima K, Fukuuchi Y, Kohsaka S. Microglia-specific localisation of a novel calcium binding protein, Iba1. Mol Brain Res. 1998;57:1–9.
Ibrahim AS, El-Remessy AB, Matragoon S, Zhang W, Patel Y, Khan S, et al. Retinal microglial activation and inflammation induced by amadori-glycated albumin in a rat model of diabetes. Diabetes. 2011;60:1122–33.
Li T, Chen ZJ. The cGAS–cGAMP–STING pathway connects DNA damage to inflammation, senescence, and cancer. J Exp Med. 2018;215:1287.
Huang Y, Xu Z, Xiong S, Qin G, Sun F, Yang J, et al. Dual extra-retinal origins of microglia in the model of retinal microglia repopulation. Cell Discov 2018 4:1. 2018;4:1–16.
Okunuki Y, Mukai R, Nakao T, Tabor SJ, Butovsky O, Dana R, et al. Retinal microglia initiate neuroinflammation in ocular autoimmunity. Proc Natl Acad Sci USA. 2019;116:9989–98.
Okunuki Y, Mukai R, Pearsall EA, Klokman G, Husain D, Park DH, et al. Microglia inhibit photoreceptor cell death and regulate immune cell infiltration in response to retinal detachment. Proc Natl Acad Sci USA. 2018;115:E6264–73.
Burrows CJ, Muller JG. Oxidative nucleobase modifications leading to strand scission. Chem Rev. 1998;98:1109–51.
Cadet J, Davies KJA, Medeiros MH, Di Mascio P, Wagner JR. Formation and repair of oxidatively generated damage in cellular DNA. Free Radic Biol Med. 2017;107:13–34.
Ruszkiewicz JA, Bürkle A, Mangerich A. Fueling genome maintenance: on the versatile roles of NAD+ in preserving DNA integrity. J Biol Chem. 2022;298:102037.
Fang EF, Scheibye-Knudsen M, Chua KF, Mattson MP, Croteau DL, Bohr VA. Nuclear DNA damage signalling to mitochondria in ageing. Nat Rev Mol Cell Biol. 2016;17:308–21.
Lautrup S, Sinclair DA, Mattson MP, Fang EF. NAD+ in brain aging and neurodegenerative disorders. Cell Metab. 2019;30:630.
Wang J, Li M, Geng Z, Khattak S, Ji X, Wu D, et al. Role of oxidative stress in retinal disease and the early intervention strategies: a review. Oxid Med Cell Longev. 2022;2022:7836828.
Fu Z, Kern TS, Hellström A, Smith LEH. Fatty acid oxidation and photoreceptor metabolic needs. J Lipid Res. 2021;62:10035.
Murakami Y, Nakabeppu Y, Sonoda KH. Oxidative stress and microglial response in retinitis pigmentosa. Int J Mol Sci. 2020;21:7170.
Xu L, Kong L, Wang J, Ash JD. Stimulation of AMPK prevents degeneration of photoreceptors and the retinal pigment epithelium. Proc Natl Acad Sci. 2018;115:10475–80.
Nakamura M, Kuse Y, Tsuruma K, Shimazawa M, Hara H. The involvement of the oxidative stress in murine blue LED light-induced retinal damage model. Biol Pharm Bull. 2017;40:1219–25.
Sanz MM, Johnson LE, Ahuja S, Ekström PAR, Romero J, van Veen T. Significant photoreceptor rescue by treatment with a combination of antioxidants in an animal model for retinal degeneration. Neuroscience. 2007;145:1120–9.
Nair AB, Jacob S. A simple practice guide for dose conversion between animals and human. J Basic Clin Pharm. 2016;7:27.
Kong X, Ibukun F, Khan MJ, Hafiz G, Wehling D, Dreger K, et al. Protocol for NAC Attack, a phase-3, multicenter randomized, parallel, double masked, placebo controlled trial evaluating the efficacy and safety of oral N-acetylcysteine (NAC) in patients with retinitis pigmentosa. medRxiv. 2025. https://www.medrxiv.org/content/10.1101/2025.11.05.25339486v1.
Vitale I, Pietrocola F, Guilbaud E, Aaronson SA, Abrams JM, Adam D, et al. Apoptotic cell death in disease—Current understanding of the NCCD 2023. Cell Death Differ. 2023;30:1097–154.
Wozniak J, DiSalvo M, Farrell A, Vaudreuil C, Uchida M, Ceranoglu TA, et al. Findings from a pilot open-label trial of N-acetylcysteine for the treatment of pediatric mania and hypomania. BMC Psychiatry. 2022;22:314.
Hardan AY, Fung LK, Libove RA, Obukhanych TV, Nair S, Herzenberg LA, et al. A randomized controlled pilot trial of oral N-acetylcysteine in children with autism. Biol Psychiatry. 2012;71:956–61.
Li F, Welling MC, Johnson JA, Coughlin C, Mulqueen J, Jakubovski E, et al. N-acetylcysteine for pediatric obsessive-compulsive disorder: a small pilot study. J Child Adolesc Psychopharmacol. 2020;30:32–7.
Chen Y, Zizmare L, Calbiague V, Wang L, Yu S, Herberg FW, et al. Retinal metabolism displays evidence for uncoupling of glycolysis and oxidative phosphorylation via Cori-, Cahill-, and mini-Krebs-cycle. Elife. 2023;12:RP91141.
Karademir D, Todorova V, Ebner LJA, Samardzija M, Grimm C. Single-cell RNA sequencing of the retina in a model of retinitis pigmentosa reveals early responses to degeneration in rods and cones. BMC Biol. 2022;20:1–19.
Stepp MA, Menko AS. Immune responses to injury and their links to eye disease. Transl Res. 2021;236:52.
O’Koren EG, Yu C, Klingeborn M, Wong AYW, Prigge CL, Mathew R, et al. Microglial function is distinct in different anatomical locations during retinal homeostasis and degeneration. Immunity. 2019;50:723.
Orihuela R, McPherson CA, Harry GJ. Microglial M1/M2 polarization and metabolic states. Br J Pharm. 2016;173:649–65.
Cherry JD, Olschowka JA, O’Banion MK. Neuroinflammation and M2 microglia: the good, the bad, and the inflamed. J Neuroinflamm. 2014;11:1–15.
Zhao L, Zabel MK, Wang X, Ma W, Shah P, Fariss RN, et al. Microglial phagocytosis of living photoreceptors contributes to inherited retinal degeneration. EMBO Mol Med. 2015;7:1179–97.
Chen Y, Jiang M, Li L, Yang S, Liu Z, Lin S, et al. Absent in melanoma 2: a potent suppressor of retinal pigment epithelial-mesenchymal transition and experimental proliferative vitreoretinopathy. Cell Death Dis. 2025;16:1–13.
Viringipurampeer IA, Metcalfe AL, Bashar AE, Sivak O, Yanai A, Mohammadi Z, et al. NLRP3 inflammasome activation drives bystander cone photoreceptor cell death in a P23H rhodopsin model of retinal degeneration. Hum Mol Genet. 2016;25:1501.
Osada H, Okamoto T, Kawashima H, Toda E, Miyake S, Nagai N, et al. Neuroprotective effect of bilberry extract in a murine model of photo-stressed retina. PLoS One. 2017;12:e0178627.
Yu N, Wu X, Zhang C, Qin Q, Gu Y, Ke W, et al. NADPH and NAC synergistically inhibits chronic ocular hypertension-induced neurodegeneration and neuroinflammation through regulating p38/MAPK pathway and peroxidation. Biomed Pharmacother. 2024;175:116711.
Zhang H, Sajdak BS, Merriman DK, McCall MA, Carroll J, Lipinski DM. Electroretinogram of the cone-dominant thirteen-lined ground squirrel during Euthermia and Hibernation in comparison with the rod-dominant Brown Norway rat. Invest Ophthalmol Vis Sci. 2020;61:6–6.
Zhang H, Sajdak BS, Merriman DK, Carroll J, Lipinski DM. Pre-retinal delivery of recombinant adeno-associated virus vector significantly improves retinal transduction efficiency. Mol Ther Methods Clin Dev. 2021;22:96–106.
Acknowledgements
The authors thank the Animal Facility staff for their help with animal care, and gratefully acknowledge Caitlin Keiper and Miele Macmillan for their technical assistance.
Funding
This work was supported by grants from the National Eye Institute (EY012910 to EAP), the Knights Templar Eye Foundation (2024-40 to HZ), and the Gavin R. Stevens Foundation.
Author information
Authors and Affiliations
Contributions
EAP and HZ designed the study and prepared the manuscript. HZ performed the experiments and analyzed the data. KV contributed to mouse genotyping and OCT image analysis. RMB assisted with NAC and PBS intraperitoneal injections in mice.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
All experimental animal procedures were approved by the Animal Care and Use Committee of Massachusetts Eye and Ear (2021N000059) and were conducted in accordance with its guidelines and regulations.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Edited by Professor Massimiliano Agostini
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zhang, H., Valestil, K., Butcher, R.M. et al. Oxidative DNA damage drives apoptotic photoreceptor loss in NMNAT1-associated inherited retinal degeneration: a therapeutic opportunity. Cell Death Dis (2026). https://doi.org/10.1038/s41419-026-08680-7
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41419-026-08680-7