Abstract
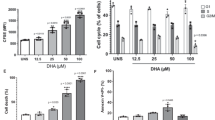
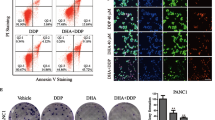
Several epidemiological and preclinical studies suggest that omega-3 (n-3) polyunsaturated fatty acids (PUFAs) exert anticancer activity at multiple stages of colorectal cancer (CRC) progression. However, inconsistent clinical evidence and the lack of a clearly defined molecular mechanism underlying the antitumor effects of n-3 PUFAs have raised doubts about their efficacy as anticancer therapies. To address these issues, we investigated the effects of the n-3 PUFA docosahexaenoic acid (DHA) in a collection of CRC patient-derived tumor organoids (PDTOs), a powerful platform for functional analysis of patient-specific tumors. DHA treatment markedly reduced CRC cell viability in a time- and concentration-dependent manner without inducing apoptosis. CRC-derived PDTOs exhibited pronounced sensitivity to DHA, irrespective of KRAS or TP53 mutational status, whereas organoids from normal colon tissue were less affected. Mechanistically, DHA induced ferroptosis in both CRC cells and PDTOs, as evidenced by lipid peroxide accumulation and partial rescue by ferroptosis inhibitors. Fluorescently labeled DHA localized predominantly to the endoplasmic reticulum and mitochondria, where it promoted oxidative stress. Moreover, DHA impaired the regrowth of oxaliplatin-tolerant persister cells and enhanced oxaliplatin efficacy in sequential treatment models. Together, these findings indicate that exploiting the intrinsic oxidative vulnerability of cancer cells with DHA may represent a promising, low-toxicity strategy to enhance chemotherapy efficacy and target drug-tolerant persister cells in colorectal cancer.
Similar content being viewed by others
Data availability
Requests for further information and resources should be directed to and will be fulfilled by the lead contact Luca Primo (luca.primo@unito.it).
References
Stockwell BR, Friedmann Angeli JP, Bayir H, Bush AI, Conrad M, Dixon SJ, et al. Ferroptosis: a regulated cell death nexus linking metabolism, redox biology, and disease. Cell. 2017;171:273–85.
Dixon SJ, Lemberg KM, Lamprecht MR, Skouta R, Zaitsev EM, Gleason CE, et al. Ferroptosis: an iron-dependent form of nonapoptotic cell death. Cell. 2012;149:1060–72.
Yang WS, SriRamaratnam R, Welsch ME, Shimada K, Skouta R, Viswanathan VS, et al. Regulation of ferroptotic cancer cell death by GPX4. Cell. 2014;156:317–31.
Yang WS, Kim KJ, Gaschler MM, Patel M, Shchepinov MS, Stockwell BR. Peroxidation of polyunsaturated fatty acids by lipoxygenases drives ferroptosis. Proc Natl Acad Sci USA. 2016;113:E4966–75.
Doll S, Proneth B, Tyurina YY, Panzilius E, Kobayashi S, Ingold I, et al. ACSL4 dictates ferroptosis sensitivity by shaping cellular lipid composition. Nat Chem Biol. 2017;13:91–98.
Fuentes NR, Mlih M, Barhoumi R, Fan YY, Hardin P, Steele TJ, et al. Long-chain n-3 fatty acids attenuate oncogenic KRas-driven proliferation by altering plasma membrane nanoscale proteolipid composition. Cancer Res. 2018;78:3899–912.
Calder PC. Marine omega-3 fatty acids and inflammatory processes: effects, mechanisms and clinical relevance. Biochim Biophys Acta. 2015;1851:469–84.
Aldoori J, Cockbain AJ, Toogood GJ, Hull MA. Omega-3 polyunsaturated fatty acids: moving towards precision use for prevention and treatment of colorectal cancer. Gut. 2022;71:822–37.
Siegel RL, Miller KD, Fuchs HE, Jemal A. Cancer statistics, 2022. CA Cancer J Clin. 2022;72:7–33.
Fearon ER, Vogelstein B. A genetic model for colorectal tumorigenesis. Cell. 1990;61:759–67.
Cunningham D, Humblet Y, Siena S, Khayat D, Bleiberg H, Santoro A, et al. Cetuximab monotherapy and cetuximab plus irinotecan in irinotecan-refractory metastatic colorectal cancer. N Engl J Med. 2004;351:337–45.
Pu Y, Li L, Peng H, Liu L, Heymann D, Robert C, et al. Drug-tolerant persister cells in cancer: the cutting edges and future directions. Nat Rev Clin Oncol. 2023;20:799–813.
MacLean CH, Newberry SJ, Mojica WA, Khanna P, Issa AM, Suttorp MJ, et al. Effects of omega-3 fatty acids on cancer risk: a systematic review. JAMA. 2006;295:403–15.
Volpato M, Hull MA. Omega-3 polyunsaturated fatty acids as adjuvant therapy of colorectal cancer. Cancer Metastasis Rev. 2018;37:545–55.
Sun G, Fuller H, Fenton H, Race AD, Downing A, Rees CJ, et al. The relationship between dietary and supplemental omega-3 highly unsaturated fatty acid intake, blood and tissue omega-3 highly unsaturated fatty acid concentrations, and colorectal polyp recurrence: a secondary analysis of the seAFOod polyp prevention trial. J Nutr. 2025;155:549–58.
Song M, Ou FS, Zemla TJ, Hull MA, Shi Q, Limburg PJ, et al. Marine omega-3 fatty acid intake and survival of stage III colon cancer according to tumor molecular markers in NCCTG Phase III trial N0147 (Alliance). Int J Cancer. 2019;145:380–9.
Cockbain AJ, Volpato M, Race AD, Munarini A, Fazio C, Belluzzi A, et al. Anticolorectal cancer activity of the omega-3 polyunsaturated fatty acid eicosapentaenoic acid. Gut. 2014;63:1760–8.
Liu H, Chen J, Shao W, Yan S, Ding S. Efficacy and safety of Omega-3 polyunsaturated fatty acids in adjuvant treatments for colorectal cancer: a meta-analysis of randomized controlled trials. Front Pharmacol. 2023;14:1004465.
Hull MA, Ow PL, Ruddock S, Brend T, Smith AF, Marshall H, et al. Randomised, placebo-controlled, phase 3 trial of the effect of the omega-3 polyunsaturated fatty acid eicosapentaenoic acid (EPA) on colorectal cancer recurrence and survival after surgery for resectable liver metastases: EPA for metastasis trial 2 (EMT2) study protocol. BMJ Open. 2023;13:e077427.
van de Wetering M, Francies HE, Francis JM, Bounova G, Iorio F, Pronk A, et al. Prospective derivation of a living organoid biobank of colorectal cancer patients. Cell. 2015;161:933–45.
Sachs N, Clevers H. Organoid cultures for the analysis of cancer phenotypes. Curr Opin Genet Dev. 2014;24:68–73.
Schutgens F, Clevers H. Human organoids: tools for understanding biology and treating diseases. Annu Rev Pathol. 2020;15:211–34.
Leto SM, Grassi E, Avolio M, Vurchio V, Cottino F, Ferri M, et al. XENTURION is a population-level multidimensional resource of xenografts and tumoroids from metastatic colorectal cancer patients. Nat Commun. 2024;15:7495.
Jayathilake AG, Luwor RB, Nurgali K, Su XQ. Molecular mechanisms associated with the inhibitory role of long chain n-3 PUFA in colorectal cancer. Integr Cancer Ther. 2024;23:15347354241243024.
Qiu B, Zandkarimi F, Bezjian CT, Reznik E, Soni RK, Gu W, et al. Phospholipids with two polyunsaturated fatty acyl tails promote ferroptosis. Cell. 2024;187:1177–90.e1118.
Dierge E, Debock E, Guilbaud C, Corbet C, Mignolet E, Mignard L, et al. Peroxidation of n-3 and n-6 polyunsaturated fatty acids in the acidic tumor environment leads to ferroptosis-mediated anticancer effects. Cell Metab. 2021;33:1701–15.e1705.
Kerk SA, Papagiannakopoulos T, Shah YM, Lyssiotis CA. Metabolic networks in mutant KRAS-driven tumours: tissue specificities and the microenvironment. Nat Rev Cancer. 2021;21:510–25.
Friedmann Angeli JP, Schneider M, Proneth B, Tyurina YY, Tyurin VA, Hammond VJ, et al. Inactivation of the ferroptosis regulator Gpx4 triggers acute renal failure in mice. Nat Cell Biol. 2014;16:1180–91.
Rodriguez R, Schreiber SL, Conrad M. Persister cancer cells: Iron addiction and vulnerability to ferroptosis. Mol Cell. 2022;82:728–40.
Hangauer MJ, Viswanathan VS, Ryan MJ, Bole D, Eaton JK, Matov A, et al. Drug-tolerant persister cancer cells are vulnerable to GPX4 inhibition. Nature. 2017;551:247–50.
Jiang X, Stockwell BR, Conrad M. Ferroptosis: mechanisms, biology and role in disease. Nat Rev Mol Cell Biol. 2021;22:266–82.
Murray MB, Leak LB, Lee WC, Dixon SJ. Protocol for detection of ferroptosis in cultured cells. STAR Protoc. 2023;4:102457.
Sokol KH, Lee CJ, Rogers TJ, Waldhart A, Ellis AE, Madireddy S, et al. Lipid availability influences ferroptosis sensitivity in cancer cells by regulating polyunsaturated fatty acid trafficking. Cell Chem Biol. 2025;32:408–22.e406.
Perez MA, Magtanong L, Dixon SJ, Watts JL. Dietary lipids induce ferroptosis in Caenorhabditis elegans and human cancer cells. Dev Cell. 2020;54:447–54.e444.
Ubellacker JM, Dixon SJ. Prospects for ferroptosis therapies in cancer. Nat Cancer. 2025;6:1326–36.
Viale A, Pettazzoni P, Lyssiotis CA, Ying H, Sánchez N, Marchesini M, et al. Oncogene ablation-resistant pancreatic cancer cells depend on mitochondrial function. Nature. 2014;514:628–32.
Keoh LQ, Chiu CF, Ramasamy TS. Metabolic plasticity and cancer stem cell metabolism: exploring the glycolysis-OXPHOS switch as a mechanism for resistance and tumorigenesis. Stem Cell Rev Rep. 2025;21:2446–68.
Elgendy SM, Alyammahi SK, Alhamad DW, Abdin SM, Omar HA. Ferroptosis: an emerging approach for targeting cancer stem cells and drug resistance. Crit Rev Oncol Hematol. 2020;155:103095.
Shan K, Feng N, Zhu D, Qu H, Fu G, Li J, et al. Free docosahexaenoic acid promotes ferroptotic cell death via lipoxygenase dependent and independent pathways in cancer cells. Eur J Nutr. 2022;61:4059–75.
Dhimolea E, de Matos Simoes R, Kansara D, Al’Khafaji A, Bouyssou J, Weng X, et al. An embryonic diapause-like adaptation with suppressed Myc activity enables tumor treatment persistence. Cancer Cell. 2021;39:240–56.e211.
Russo M, Chen M, Mariella E, Peng H, Rehman SK, Sancho E, et al. Cancer drug-tolerant persister cells: from biological questions to clinical opportunities. Nat Rev Cancer. 2024;24:694–717.
Alcorta A, Lopez-Gomez L, Capasso R, Abalo R. Vitamins and fatty acids against chemotherapy-induced intestinal mucositis. Pharmacol Ther. 2024;261:108689.
Sztolsztener K, Harasim-Symbor E, Chabowski A, Konstantynowicz-Nowicka K. Cannabigerol as an anti-inflammatory agent altering the level of arachidonic acid derivatives in the colon tissue of rats subjected to a high-fat high-sucrose diet. Biomed Pharmacother. 2024;178:117286.
Aldoori J, Zulyniak MA, Toogood GJ, Hull MA. Plasma n-3 polyunsaturated fatty acid levels and colorectal cancer risk in the UK Biobank: evidence of nonlinearity, as well as tumor site- and sex-specificity. Cancer Epidemiol Biomarkers Prev. 2025;34:394–404.
Browning LM, Walker CG, Mander AP, West AL, Madden J, Gambell JM, et al. Incorporation of eicosapentaenoic and docosahexaenoic acids into lipid pools when given as supplements providing doses equivalent to typical intakes of oily fish. Am J Clin Nutr. 2012;96:748–758.
Agagunduz D, Cocozza E, Cemali O, Bayazit AD, Nani MF, Cerqua I, et al. Understanding the role of the gut microbiome in gastrointestinal cancer: a review. Front Pharmacol. 2023;14:1130562.
Hajjar R, Mars RAT, Kashyap PC. Harnessing the microbiome for cancer therapy. Nat Rev Microbiol. 2026:1–16.
Lazzari L, Corti G, Picco G, Isella C, Montone M, Arcella P, et al. Patient-derived xenografts and matched cell lines identify pharmacogenomic vulnerabilities in colorectal cancer. Clin Cancer Res. 2019;25:6243–59.
Kleih M, Bopple K, Dong M, Gaissler A, Heine S, Olayioye MA, et al. Direct impact of cisplatin on mitochondria induces ROS production that dictates cell fate of ovarian cancer cells. Cell Death Dis. 2019;10:851.
Acknowledgements
This work was supported by AIRC (Associazione Italiana per la Ricerca sul Cancro) grant IG-23211 to LP and MFAG-25040 to AP; FPRC 5 × 1000 Ministero della Salute 2022 CARESS to AP; MUR (Dipartimenti di Eccellenza DM 11/05/2017 n262) to the Department of Oncology, University of Turin (2023–2027 14586 DIORAMA); MUR PRIN2022A93K7S_003 to LP and PRIN2022ECBA39 to VM; Italian Ministry of Health, Ricerca Corrente 2025. VCL was supported by MSCA program fellowship and by Fondazione Veronesi fellowship. We are grateful to Jessica Enriquez for her assistance with microscopic imaging and to Donatella Valdembri for providing antibodies and for her valuable suggestions in the immunofluorescence analysis.
Author information
Authors and Affiliations
Contributions
MVM, VM, LdB, and LP conceived the study; LdB, MVM, EM, VCL, and VM performed cells and organoids experiments; BP, LdB and VM performed immunofluorescence, confocal microscopy and FACS experiments; AP, BP, LdB, and VM performed imaging analysis; AB, LT, and AP gave conceptual advice; LP, LdB, and VM supervised the project and interpreted the results. LP drafted the initial manuscript; MVM, LdB, VM, AP, AB and LT revised and edited the final version. LP, AP, and VM acquired funding. All authors reviewed and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
LT has received research grants from Menarini, Merck KGaA, Merus, Pfizer, Servier and Symphogen outside the submitted work. The other authors declare no competing interests.
Ethics approval
All methods were performed in accordance with the relevant guidelines and regulations. Human PDTOs and intestinal organoids were derived from CRC liver metastases and from healthy intestine. These studies were approved respectively, by medical ethical committees of Fondazione del Piemonte per l’Oncologia FPO - IRCCS (PROFILING protocol No. 001-IRCC-00IIS-10, version 11.0, updated July 13, 2022) and by ethical committee of each participating center to the ALFAOMEGA Master Observational Trial (NCT04120935). All patients provided written informed consent.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Edited by Professor Dagmar Kulms
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
di Blasio, L., Vara-Messler, M., Peracino, B. et al. Omega-3 fatty acid DHA induces ferroptosis in colorectal cancer patient-derived organoids and drug-tolerant cells. Cell Death Dis (2026). https://doi.org/10.1038/s41419-026-08744-8
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41419-026-08744-8