Abstract
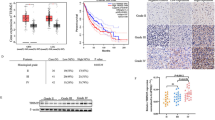
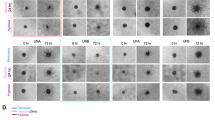
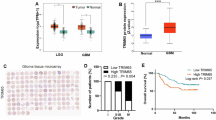
Glioblastoma (GBM) frequently activates hypoxia signaling even under normoxic conditions, yet the mechanism sustaining hypoxia-inducible factor-1α (HIF-1α) stability remains unclear. Here, we identify the E3 ubiquitin ligase TRIM25 as a key driver of this phenomenon. TRIM25, aberrantly upregulated in GBM, directly binds HIF-1α and catalyzes K11/K29-linked polyubiquitination at lysine 532 of hydroxylated HIF-1α, preventing its canonical proteasomal degradation. This non-canonical ubiquitin modification stabilizes HIF-1α despite normal oxygen availability and sustains a pseudohypoxic transcriptional program in GBM cells. Functional studies in GBM cell lines, patient-derived cultures, and tumor models demonstrate that TRIM25-mediated HIF-1α stabilization promotes tumor proliferation, invasion, and angiogenic potential. Importantly, small-molecule screening identified T7117 as an inhibitor that disrupts the TRIM25–HIF-1α interaction, suppresses tumor growth, and enhances temozolomide efficacy. Together, our findings uncover a previously unrecognized ubiquitin mechanism that stabilizes hydroxylated HIF-1α under normoxia, revealing the TRIM25–HIF-1α axis as a driver of GBM pseudohypoxia and a potential therapeutic target.
Similar content being viewed by others
Data availability
The RNA-seq dataset was deposited in the Sequence Read Archive (SRA) repository at NCBI under the accession number PRJNA1347646. The datasets used and analyzed during the current study are available from the corresponding author upon reasonable request.
Code availability
Public R packages used in this study include DESeq2 and edgeR. In-house Perl scripts for raw FASTQ sequencing data trimming are available from the corresponding author upon reasonable request, with no restrictions on access.
References
Grochans S, Cybulska AM, Siminska D, Korbecki J, Kojder K, Chlubek D, et al. Epidemiology of Glioblastoma Multiforme: Literature Review. Cancers 14. (2022).
Schaff LR, Mellinghoff IK. Glioblastoma and other primary brain malignancies in adults: a review. JAMA. 2023;329:574–587.
Weller M, van den Bent M, Preusser M, Le Rhun E, Tonn JC, Minniti G, et al. EANO guidelines on the diagnosis and treatment of diffuse gliomas of adulthood. Nat Rev Clin Oncol. 2021;18:170–186.
Stupp R, Hegi ME, Mason WP, van den Bent MJ, Taphoorn MJ, Janzer RC, et al. Effects of radiotherapy with concomitant and adjuvant temozolomide versus radiotherapy alone on survival in glioblastoma in a randomised phase III study: 5-year analysis of the EORTC-NCIC trial. Lancet Oncol. 2009;10:459–466.
Liao C, Liu X, Zhang C, Zhang Q. Tumor hypoxia: From basic knowledge to therapeutic implications. Semin Cancer Biol. 2023;88:172–186.
Beckers C, Pruschy M, Vetrugno I. Tumor hypoxia and radiotherapy: A major driver of resistance even for novel radiotherapy modalities. Semin Cancer Biol. 2024;98:19–30.
Zhang Y, Zhang B, Lv C, Zhang N, Xing K, Wang Z, et al. Single-cell RNA sequencing identifies critical transcription factors of tumor cell invasion induced by hypoxia microenvironment in glioblastoma. Theranostics. 2023;13:3744–3760.
Sattiraju A, Kang S, Giotti B, Chen Z, Marallano VJ, Brusco C, et al. Hypoxic niches attract and sequester tumor-associated macrophages and cytotoxic T cells and reprogram them for immunosuppression. Immunity. 2023;56:1825–1843.e1826.
de Oliveira KG, Bang-Rudenstam A, Beyer S, Boukredine A, Talbot H, Governa V, et al. Decoding of the surfaceome and endocytome in primary glioblastoma cells identifies potential target antigens in the hypoxic tumor niche. Acta Neuropathol Commun. 2024;12:35.
Yin H, Liu Y, Dong Q, Wang H, Yan Y, Wang X, et al. The mechanism of extracellular CypB promotes glioblastoma adaptation to a glutamine-deprived microenvironment. Cancer Lett. 2024;597:216862.
Chung YJ, Hoare Z, Baark F, Yu CS, Guo J, Fuller W, et al. Elevated Na is a dynamic and reversible modulator of mitochondrial metabolism in the heart. Nat Commun. 2024;15:4277.
Zhang J, Yao M, Xia S, Zeng F, Liu Q. Systematic and comprehensive insights into HIF-1 stabilization under normoxic conditions: implications for cellular adaptation and therapeutic strategies in cancer. Cell Mol Biol Lett. 2025;30:2.
Rendina BP, Ahmad F, Akerman L, Mills I, Lee NC, Chen C, et al. Post-transcriptional control of HIF-1alpha by MBNL1 restrains hypoxia-driven stemness in GBM. Neoplasia. 2026;72:101262.
Semenza GL. Targeting HIF-1 for cancer therapy. Nat Rev Cancer. 2003;3:721–732.
Lee JW, Bae SH, Jeong JW, Kim SH, Kim KW. Hypoxia-inducible factor (HIF-1)alpha: its protein stability and biological functions. Exp Mol Med. 2004;36:1–12.
Schofield CJ, Ratcliffe PJ. Oxygen sensing by HIF hydroxylases. Nat Rev Mol Cell Biol. 2004;5:343–354.
Paredes F, Williams HC, San Martin A. Metabolic adaptation in hypoxia and cancer. Cancer Lett. 2021;502:133–142.
Chen T, Li K, Liu Z, Liu J, Wang Y, Sun R, et al. WDR5 facilitates EMT and metastasis of CCA by increasing HIF-1alpha accumulation in Myc-dependent and independent pathways. Mol Ther. 2021;29:2134–2150.
Dong S, Liang S, Cheng Z, Zhang X, Luo L, Li L, et al. ROS/PI3K/Akt and Wnt/beta-catenin signalings activate HIF-1alpha-induced metabolic reprogramming to impart 5-fluorouracil resistance in colorectal cancer. J Exp Clin Cancer Res. 2022;41:15.
Selak MA, Armour SM, MacKenzie ED, Boulahbel H, Watson DG, Mansfield KD, et al. Succinate links TCA cycle dysfunction to oncogenesis by inhibiting HIF-alpha prolyl hydroxylase. Cancer Cell. 2005;7:77–85.
Yang JX, Chuang YC, Tseng JC, Liu YL, Lai CY, Lee AY, et al. Tumor-promoting effect of PDLIM2 downregulation involves mitochondrial ROS, oncometabolite accumulations and HIF-1alpha activation. J Exp Clin Cancer Res. 2024;43:169.
Lawson H, Holt-Martyn JP, Dembitz V, Kabayama Y, Wang LM, Bellani A, et al. The selective prolyl hydroxylase inhibitor IOX5 stabilizes HIF-1alpha and compromises development and progression of acute myeloid leukemia. Nat Cancer. 2024;5:916–937.
Tan S, Ming Y, Guo J, Liu W, Ma H, Liu Y, et al. A circRNA promotes esophageal squamous cell carcinoma progression by inhibiting TRIM25-mediated degradation of IGF2BP family members. Mol Cancer. 2025;24:243.
Wang J, Huang Z, Ji L, Chen C, Wan Q, Xin Y, et al. SHF acts as a novel tumor suppressor in glioblastoma multiforme by disrupting STAT3 dimerization. Adv Sci. 2022;9:e2200169.
Jiao J, Zhang R, Li Z, Yin Y, Fang X, Ding X, et al. Nuclear Smad6 promotes gliomagenesis by negatively regulating PIAS3-mediated STAT3 inhibition. Nat Commun. 2018;9:2504.
Liu Y, Ni K, Zhao S, Zhao J, Zhong M, Cheng C, et al. CBLB regulates MAPK-P38 pathway via MAP3K9 ubiquitination to inhibit GBM cell invasion and migration. J Cell Physiol. 2025;240:e70037.
Paltoglou S, Roberts BJ. HIF-1alpha and EPAS ubiquitination mediated by the VHL tumour suppressor involves flexibility in the ubiquitination mechanism, similar to other RING E3 ligases. Oncogene. 2007;26:604–609.
Tanimoto K, Makino Y, Pereira T, Poellinger L. Mechanism of regulation of the hypoxia-inducible factor-1 alpha by the von Hippel-Lindau tumor suppressor protein. EMBO J. 2000;19:4298–4309.
Wang X, Wu R, Zhai P, Liu Z, Xia R, Zhang Z, et al. Hypoxia promotes EV secretion by impairing lysosomal homeostasis in HNSCC through negative regulation of ATP6V1A by HIF-1alpha. J Extracell Vesicles. 2023;12:e12310.
McAleese CE, Choudhury C, Butcher NJ, Minchin RF. Hypoxia-mediated drug resistance in breast cancers. Cancer Lett. 2021;502:189–199.
Luo YW, Fang Y, Zeng HX, Ji YC, Wu MZ, Li H, et al. HIF1alpha counteracts TGFbeta1-driven TSP1 expression in endothelial cells to stimulate angiogenesis in the hypoxic tumor microenvironment. Cancer Res. 2025;85:69–83.
Liu Z, Wang Y, Dou C, Xu M, Sun L, Wang L, et al. Hypoxia-induced up-regulation of VASP promotes invasiveness and metastasis of hepatocellular carcinoma. Theranostics. 2018;8:4649–4663.
Song H, Qiu Z, Wang Y, Xi C, Zhang G, Sun Z, et al. HIF-1alpha/YAP signaling rewrites glucose/iodine metabolism program to promote papillary thyroid cancer progression. Int J Biol Sci. 2023;19:225–241.
Hao S, Li F, Jiang P, Gao J. Effect of chronic intermittent hypoxia-induced HIF-1alpha/ATAD2 expression on lung cancer stemness. Cell Mol Biol Lett. 2022;27:44.
Pugh CW, Ratcliffe PJ. The von Hippel-Lindau tumor suppressor, hypoxia-inducible factor-1 (HIF-1) degradation, and cancer pathogenesis. Semin Cancer Biol. 2003;13:83–89.
Morreale FE, Walden H. Types of Ubiquitin ligases. Cell. 2016;165:248–248.e241.
Zhu S, Zhang X, Liu W, Zhou Z, Xiong S, Li J, et al. Ubiquitination in cancer: mechanisms and therapeutic opportunities. Cancer Commun. 2025;45:1128–1161.
Komander D, Rape M. The ubiquitin code. Annu Rev Biochem. 2012;81:203–229.
Sun Y, Zhang Z, Li H, Bu X, Chen L, Wang X, et al. Destruction of VISTA by TRIM25 ablation in T cells potentiates cancer immunotherapy. Cell Res. 2025;35:1003–1020.
Li Y, Wu S, Tian X, Kong C, Hong W, Xiao T, et al. The structural basis of TRIM25-mediated regulation of RIG-I. J Biol Chem. 2025;301:108367.
Zhou L, Wang Z, Huang Y, Zhang X, Jiang H, Guo Z, et al. NLRP12 decreases TRIM25-mediated HK2 degradation to promote glycolysis and H3K18la in gastric cancer. Cell Death Dis. 2025;16:615.
Chen Y, Xu X, Ding K, Tang T, Cai F, Zhang H, et al. TRIM25 promotes glioblastoma cell growth and invasion via regulation of the PRMT1/c-MYC pathway by targeting the splicing factor NONO. J Exp Clin Cancer Res. 2024;43:39.
Zhu M, Wu Y, Ou H, Liu X, Wang Y, Liu X, et al. Targeting TRIM25 as a therapeutic strategy to enhance ferroptosis in glioblastoma cells. J Nanobiotechnol. 2026;24:20.
Wei J, Wang L, Zhang Y, Sun T, Zhang C, Hu Z, et al. TRIM25 promotes temozolomide resistance in glioma by regulating oxidative stress and ferroptotic cell death via the ubiquitination of Keap1. Oncogene. 2023;42:2103–2112.
Yan Y, Zhou S, Chen X, Yi Q, Feng S, Zhao Z, et al. Suppression of ITPKB degradation by Trim25 confers TMZ resistance in glioblastoma through ROS homeostasis. Signal Transduct Target Ther. 2024;9:58.
Ma Q, Jiang H, Ma L, Zhao G, Xu Q, Guo D, et al. The moonlighting function of glycolytic enzyme enolase-1 promotes choline phospholipid metabolism and tumor cell proliferation. Proc Natl Acad Sci USA. 2023;120:e2209435120.
Bratkovic D, Margvelashvili L, Tchan MC, Nisbet J, Smith N. PTC923 (sepiapterin) lowers elevated blood phenylalanine in subjects with phenylketonuria: a phase 2 randomized, multi-center, three-period crossover, open-label, active controlled, all-comers study. Metabolism. 2022;128:155116.
Martinez-Pizarro A, Pico S, Lopez-Marquez A, Rodriguez-Lopez C, Montalvo E, Alvarez M, et al. PAH deficient pathology in humanized c.1066-11G>A phenylketonuria mice. Hum Mol Genet. 2024;33:1074–1089.
Czechowicz P, Wiech-Walow A, Slawski J, Collawn JF, Bartoszewski R. Old drugs, new challenges: reassigning drugs for cancer therapies. Cell Mol Biol Lett. 2025;30:27.
Savander S, Curabaz NN, Abbasi AM, Khan A, Saeed K, Tanoli Z Data-driven strategies for drug repurposing: insights, recommendations, and case studies. Brief Bioinform 2025, 26.
Acknowledgements
The authors thank the MogoEdit group (https://www.mogoedit.com) for the assistance in language editing.
Funding
J.J. discloses support for the research of this work from the Natural Science Foundation of China (NSFC) (Grant numbers: 82472723 and 82172955). J. Shao discloses support for the research of this work from the Key Project of the Jiangsu Provincial Health Commission (Grant number: ZD2022038). H. H. discloses support for the research of this work from the 2024 Jiangsu Province Graduate Student Practice Program (Grant number: SJCX24_0728). Other authors declare no relevant funding.
Author information
Authors and Affiliations
Contributions
HH, KN, CL and MC contributed equally to this work. HH, KN, CL, and MC contributed to the design of the study, analyzed data, performed all experiments, prepared figures, and edited the manuscript. YL, JZ, YS and YC performed computational analyses. JS, JShao, Y L, JW and JJ conceptualized and coordinated the study, analyzed the data, and wrote the paper. All authors read and edited the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
All methods and experimental protocols in this study were performed in accordance with the relevant guidelines and regulations (including the Declaration of Helsinki for human studies and the ARRIVE guidelines for animal research). For human-related research, approval was obtained from the Institutional Review Board and Ethics Committee of the Affiliated Wuxi People’s Hospital of Nanjing Medical University (Reference number: (2023)109). Written informed consent was obtained from all participants prior to their inclusion in the study. Furthermore, for any identifiable images originating from human research participants, the authors have obtained separate written informed consent for publication of these images. For animal-related research, all procedures involving live vertebrates were approved by the Ethics Committee of the Affiliated Wuxi People’s Hospital of Nanjing Medical University (Reference number: (2023)114) and were performed in strict compliance with the institutional animal care regulations and the ARRIVE guidelines.
Publication ethics
The authors declare that the manuscript, or any part of it, has not been previously published or submitted concurrently to any other journal.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Edited by Professor Bertrand Joseph
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Huang, H., Ni, K., Li, C. et al. TRIM25 promotes glioblastoma progression by stabilizing HIF-1α expression in normoxia through K11/K29 polyubiquitination. Cell Death Dis (2026). https://doi.org/10.1038/s41419-026-08757-3
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41419-026-08757-3