Abstract
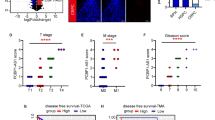
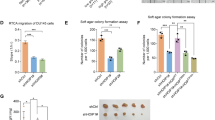
Castrate resistant prostate cancer (CRPC) is often driven by constitutively active androgen receptor and AR splicing variants that become resistant to established hormonal therapy strategies such as enzalutamide. Deubiquitinating enzymes (DUBs) play crucial roles in cancer development, progression, and metastasis by epigenetic modification. Hence, targeting DUBs might prove to be a valid strategy for developing novel anti-cancer therapeutics. Here, we reveal that the deubiquitinating enzyme USP13 is up-regulated in PCa tissues and correlates with prostate cancer progression. USP13 silencing inhibits prostate cancer cell growth in vitro and in vivo. Mechanically, USP13 directly interacts with PCMT1 and removes polyubiquitination of PCMT1 to maintain its stability, which promotes PCa cell proliferation and enzalutamide resistance. Depletion of USP13 promoted PCa cells sensitive to enzalutamide. Clinically, USP13 was significantly up-regulated in prostate cancer tissues and positively associated with PCMT1 expression. Notably, inhibition of USP13 significantly decreases prostate tumor growth and improves enzalutamide treatments through PCMT1 suppression. Our studies demonstrate that inhibition of USP13 can offer a viable therapeutic option to overcome enzalutamide resistance in prostate cancer patients with USP13/PCMT1-overexpression.
Similar content being viewed by others
Acknowledgements
This work is supported by the National Natural Science Foundation of P.R. China (grant no. 82460497), the National Natural Science Foundation of P.R. China (grant no. 82460547), the Jiangxi Provincial Natural Science Foundation for Distinguished Young Scholars (grant no. 20232ACB216014), the Natural Science Foundation of Jiangxi (grant no. 20242BAB26142), the Clinical Research Cultivation Program of First Clinical Hospital of Nanchang University (YFYLCYJPY202313), the Science and Technology Plan Project of Health Commission of Jiangxi Province (grant no. 202210361), the Science and Technology Plan Project of Health Commission of Jiangxi Province (grant no. 202410018), and the Foundation of Jiangxi Provincial Key Laboratory of Urinary System Diseases (Grant No. 2024SSY06111).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
The animal experiments performed here have been approved by the Animal Protection and Use Committee of The First Affiliated Hospital of Nanchang University (approval number: CDYFY-IACUC-202407QR186). Clinic tissue sample collection was approved by the Internal Review and Ethics Boards of the First Affiliated Hospital of Nanchang University (Registration number: (2024) CDYFYYLK(10-026)). Informed consent was obtained from all participants, and all methods were performed in accordance with relevant guidelines and regulations.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Edited by Dr Gerry Melino
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Wang, Z., Liu, X., Li, Z. et al. USP13 promotes enzalutamide resistance by catalyzing depolyubiquitination of PCMT1 in prostate cancer. Cell Death Dis (2026). https://doi.org/10.1038/s41419-026-08824-9
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41419-026-08824-9