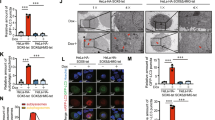
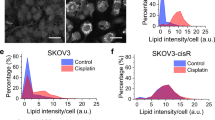
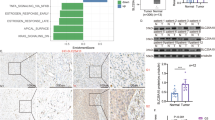
Abstract
Cisplatin resistance remains a major cause of chemotherapy failure in cervical cancer. Although our previous work identified that SOX4 promotes cisplatin resistance in cervical cancer cells, the underlying mechanism has not been fully elucidated. Here, we demonstrated that SOX4 not only induces resistance to cisplatin but also to oxaliplatin and carboplatin, suggesting its potential role as a multidrug resistance gene. Overexpression of SOX4 markedly suppressed glycolytic activity in cervical cancer cells and induced cisplatin resistance by inhibiting both the intrinsic and extrinsic apoptotic pathways. Rescue and neutralization experiments further indicated that SOX4 upregulates SIRT1, which subsequently represses the expression of GLUT1 on the cell membrane. This suppression leads to diminished cellular glucose uptake, resulting in decreased glycolysis and overall metabolic activity. Given that cisplatin preferentially targets highly proliferating cells, SOX4-driven metabolic deceleration enables cervical cancer cells to evade cisplatin-mediated cytotoxicity. Together, these findings demonstrate that SOX4 enhances cisplatin resistance in cervical cancer through SIRT1-upregulated suppression of glycolysis.
Similar content being viewed by others
Data availability
All data generated or analysed during this study are included in this published article and its supplementary information files.
References
Wang X, Song Y, Wei X, Wang G, Sun R, Wang M, et al. Prevalence and distribution of human papillomavirus genotypes among women attending gynecology clinics in northern Henan Province of China [J]. Virol J. 2022;19:6.
Bray F, Laversanne M, Sung H, Ferlay J, Siegel RL, Soerjomataram I, et al. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries [J]. CA Cancer J Clin. 2024;74:229–63.
Petca A, Borislavschi A, Zvanca ME, Petca RC, Sandru F, Dumitrascu MC. Non-sexual HPV transmission and role of vaccination for a better future (Review) [J]. Exp Ther Med. 2020;20:186.
Moreno CS. SOX4: The unappreciated oncogene [J]. Semin Cancer Biol. 2020;67:57–64.
Pan Q, Meng X, Li J, Qin X, Chen H, Li Y. CircSAMD11 facilitates progression of cervical cancer via regulating miR-503/SOX4 axis through Wnt/beta-catenin pathway [J]. Clin Exp Pharmacol Physiol. 2022;49:175–87.
Mehta GA, Angus SP, Khella CA, Tong K, Khanna P, Dixon SAH, et al. SOX4 and SMARCA4 cooperatively regulate PI3k signaling through transcriptional activation of TGFBR2 [J]. NPJ Breast Cancer. 2021;7:40.
Bagati A, Kumar S, Jiang P, Pyrdol J, Zou AE, Godicelj A, et al. Integrin alphavbeta6-TGFbeta-SOX4 pathway drives immune evasion in triple-negative breast cancer [J]. Cancer Cell. 2021;39:54–67 e9.
Yuan LY, Zhou M, Lv H, Qin X, Zhou J, Mao X, et al. Involvement of NEAT1/miR-133a axis in promoting cervical cancer progression via targeting SOX4 [J]. J Cell Physiol. 2019;234:18985–93.
Bai Y, Li X. hsa_circ_0008285 facilitates the progression of cervical cancer by targeting miR-211-5p/SOX4 axis [J]. Cancer Manag Res. 2020;12:3927–36.
Chen X, Yang J, Wang Y. LncRNA JPX promotes cervical cancer progression by modulating miR-25-3p/SOX4 axis [J]. Cancer Cell Int. 2020;20:441.
Xu Y, Zhu H, Ma H, Yuan L, Hu Q, Yang L. LINC01305 inhibits malignant progression of cervical cancer via miR-129-5p/Sox4 axis [J]. Am J Transl Res. 2020;12:7581–92.
Zhang L, Wu X, Li Y, Teng X, Zou L, Yu B. LncRNA SNHG5 promotes cervical cancer progression by regulating the miR-132/SOX4 pathway [J]. Autoimmunity. 2021;54:88–96.
Jia C, Chen F, Li W, Zhu X, Wang Y, Guo H, et al. CircCCNB1 knockdown blocks the progression of cervical cancer by acting as competing endogenous RNA in the miR-370-3p/SOX4 pathway [J]. Ann Clin Lab Sci. 2023;53:94–105.
Deng X, Wang Y, Guo H, Wang Q, Rao S, Wu H. Pan-cancer analysis and experimental validation of SOX4 as a potential diagnosis, prognosis, and immunotherapy biomarker. Cancers. 2023;15:5235.
Hanahan D. Hallmarks of cancer: new dimensions [J]. Cancer Discov. 2022;12:31–46.
Warburg O. On the origin of cancer cells [J]. Science. 1956;123:309–14.
Lu Y, Wang L, Ding W, Wang D, Wang X, Luo Q, et al. Ammonia mediates mitochondrial uncoupling and promotes glycolysis via HIF-1 activation in human breast cancer MDA-MB-231 cells [J]. Biochem Biophys Res Commun. 2019;519:153–9.
Xue YN, Yu BB, Li JL, Guo R, Zhang LC, Sun LK, et al. Zinc and p53 disrupt mitochondrial binding of HK2 by phosphorylating VDAC1 [J]. Exp Cell Res. 2019;374:249–58.
Pranzini E, Pardella E, Paoli P, Fendt SM, Taddei ML. Metabolic reprogramming in anticancer drug resistance: a focus on amino acids [J]. Trends Cancer. 2021;7:682–99.
Cao Y. Adipocyte and lipid metabolism in cancer drug resistance [J]. J Clin Invest. 2019;129:3006–17.
Yu T, Yang G, Hou Y, Tang X, Wu C, Wu XA, et al. Cytoplasmic GPER translocation in cancer-associated fibroblasts mediates cAMP/PKA/CREB/glycolytic axis to confer tumor cells with multidrug resistance [J]. Oncogene. 2017;36:2131–45.
Giacomini I, Ragazzi E, Pasut G, Montopoli M. The pentose phosphate pathway and its involvement in cisplatin resistance. Int J Mol Sci. 2020;21:937.
Wangpaichitr M, Theodoropoulos G, Nguyen DJM, Wu C, Spector SA, Feun LG, et al. Cisplatin resistance and redox-metabolic vulnerability: a second alteration. Int J Mol Sci. 2021;22:7379.
Liang XJ, Finkel T, Shen DW, Yin JJ, Aszalos A, Gottesman MM. SIRT1 contributes in part to cisplatin resistance in cancer cells by altering mitochondrial metabolism [J]. Mol Cancer Res. 2008;6:1499–506.
Liu C, Jin Y, Fan Z. The mechanism of Warburg effect-induced chemoresistance in cancer [J]. Front Oncol. 2021;11:698023.
Zwaans BM, Lombard DB. Interplay between sirtuins, MYC and hypoxia-inducible factor in cancer-associated metabolic reprogramming. Dis Model Mech. 2014;7:1023–32.
Wu QJ, Zhang TN, Chen HH, Yu XF, Lv JL, Liu YY, et al. The sirtuin family in health and disease [J]. Signal Transduct Target Ther. 2022;7:402.
Yuan J, Ofengeim D. A guide to cell death pathways [J]. Nat Rev Mol Cell Biol. 2024;25:379–95.
Cocetta V, Ragazzi E, Montopoli M. Links between cancer metabolism and cisplatin resistance [J]. Int Rev Cell Mol Biol. 2020;354:107–64.
Chelakkot C, Chelakkot VS, Shin Y, Song K. Modulating glycolysis to improve cancer therapy. Int J Mol Sci. 2023;24:2606.
Tan Y, Li J, Zhao G, Huang KC, Cardenas H, Wang Y, et al. Metabolic reprogramming from glycolysis to fatty acid uptake and beta-oxidation in platinum-resistant cancer cells [J]. Nat Commun. 2022;13:4554.
Sun R, Jiang B, Qi H, Zhang X, Yang J, Duan J, et al. SOX4 contributes to the progression of cervical cancer and the resistance to the chemotherapeutic drug through ABCG2 [J]. Cell Death Dis. 2015;6:e1990.
Liu Y, Liu L, Mou ZX. TMEM45A affects proliferation, apoptosis, epithelial-mesenchymal transition, migration, invasion and cisplatin resistance of HPV-positive cervical cancer cell lines [J]. Biochem Genet. 2022;60:173–90.
Grimm D, Bauer J, Wise P, Kruger M, Simonsen U, Wehland M, et al. The role of SOX family members in solid tumours and metastasis [J]. Semin Cancer Biol. 2020;67:122–53.
David CJ, Huang YH, Chen M, Su J, Zou Y, Bardeesy N, et al. TGF-beta tumor suppression through a lethal EMT [J]. Cell. 2016;164:1015–30.
Li L, Liu J, Xue H, Li C, Liu Q, Zhou Y, et al. A TGF-beta-MTA1-SOX4-EZH2 signaling axis drives epithelial-mesenchymal transition in tumor metastasis [J]. Oncogene. 2020;39:2125–39.
Bilir B, Osunkoya AO, Wiles WGT, Sannigrahi S, Lefebvre V, Metzger D, et al. SOX4 is essential for prostate tumorigenesis initiated by PTEN ablation [J]. Cancer Res. 2016;76:1112–21.
Lai YH, Cheng J, Cheng D, Feasel ME, Beste KD, Peng J, et al. SOX4 interacts with plakoglobin in a Wnt3a-dependent manner in prostate cancer cells [J]. BMC Cell Biol. 2011;12:50.
Lee, A, Ahn K, S, Yoon G, J, Kim H, S A. Sox4 stimulates ss-catenin activity through induction of CK2 [J]. Oncol Rep. 2011;25:559–65.
Scharer CD, Mccabe CD, Ali-Seyed M, Berger MF, Bulyk ML, Moreno CS. Genome-wide promoter analysis of the SOX4 transcriptional network in prostate cancer cells [J]. Cancer Res. 2009;69:709–17.
Sinner D, Kordich JJ, Spence JR, Opoka R, Rankin S, Lin SC, et al. Sox17 and Sox4 differentially regulate beta-catenin/T-cell factor activity and proliferation of colon carcinoma cells [J]. Mol Cell Biol. 2007;27:7802–15.
Sharma A, Thacker G, Mishra M, Singh AK, Upadhyay V, Sanyal S, et al. SOX4-mediated FBW7 transcriptional upregulation confers Tamoxifen resistance in ER+ breast cancers via GATA3 downregulation [J]. Life Sci. 2022;303:120682.
Pan S, Bao D, Li Y, Liu D, Quan S, Wang R. SOX4 induces drug resistance of colorectal cancer cells by downregulating CYLD through transcriptional activation of microRNA-17 [J]. J Biochem Mol Toxicol. 2022;36:e22910.
Galluzzi L, Senovilla L, Vitale I, Michels J, Martins I, Kepp O, et al. Molecular mechanisms of cisplatin resistance [J]. Oncogene. 2012;31:1869–83.
Chen J, Cao L, Li Z, Li Y. SIRT1 promotes GLUT1 expression and bladder cancer progression via regulation of glucose uptake [J]. Hum Cell. 2019;32:193–201.
Kim HS, Xiao C, Wang RH, Lahusen T, Xu X, Vassilopoulos A, et al. Hepatic-specific disruption of SIRT6 in mice results in fatty liver formation due to enhanced glycolysis and triglyceride synthesis [J]. Cell Metab. 2010;12:224–36.
Funding
This work was supported by the Key Scientific and Technological Projects of Henan Province (Grant No. 252102311135); the Key Scientific Research Projects of Higher Education Institutions in Henan Province (Grant No. 25A320018, 25A320020); the Natural Science Foundation of Henan (Grant No. 242300420119, 242300420123); the 111 Project (Grant No. D20036).
Author information
Authors and Affiliations
Contributions
RLS was responsible for funding acquisition, project administration, study design and experimental operation. HFG performed experimental operation, data analysis and manuscript drafting. RZ and HYL conducted experimental operation and literature investigation. HJY revised the manuscript. XFZ provided resources and materials. XLQ provided funding support. JL was responsible for study design. QZW conducted final review and editing, and provided funding support.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Sun, R., Gong, H., Zhao, R. et al. SOX4 induces cisplatin resistance in cervical cancer cells by inhibiting aerobic glycolysis. Cell Death Discov. (2026). https://doi.org/10.1038/s41420-026-02954-x
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41420-026-02954-x