Abstract
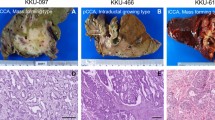
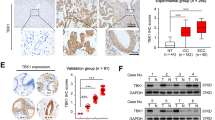
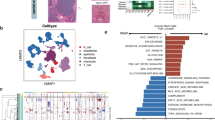
Intrahepatic cholangiocarcinoma (CCA) is a highly aggressive malignancy arising from the intrahepatic biliary epithelium with insidious onset and dismal clinical outcomes. The lack of reliable early diagnostic markers and effective therapeutic targets underscores the urgent need for novel intervention strategies. Integrated evaluation of public transcriptomic datasets and local validation cohort with survival analysis were performed to assess expression pattern and prognostic significance of cystatin SN (CST1) in CCA. Functional characterization was performed via gain- and loss-of-function experiments in HuCCT1 and RBE cells, complemented by murine orthotopic liver implantation and pulmonary metastasis models. We found that CST1 was significantly upregulated in human CCA tissues. Elevated CST1 expression predicted unfavorable prognosis in CCA patients. Subsequently functional studies revealed that overexpression of CST1 suppressed cellular senescence markers, as evidenced by decreased senescence-associated β-galactosidase activity and downregulated senescence-associated secretory phenotype factors (IL-6, CCL20). Concomitantly, CST1 overexpression enhanced cell proliferation, migration, invasion, and in vivo metastatic capacity. Integrated multi-omics profiling identified CST1-mediated suppression of pyrimidine metabolism through TYMS downregulation. However, exogenous thymidine supplementation failed to rescue proliferation defects upon CST1 knockdown, indicating that CST1-promoted tumor growth is independent of pyrimidine metabolism. Mechanistically, NFATC2 transcriptionally activates CST1, which subsequently abrogates senescence through SOX4 stabilization; ectopic SOX4 expression rescues senescence induced by CST1 depletion. These findings establish CST1 as a promising therapeutic target and provide mechanistic insights for CCA intervention strategies.
Similar content being viewed by others
Data availability
The datasets used during the current study are available from the corresponding author on reasonable request.
References
Banales JM, Marin JJG, Lamarca A, Rodrigues PM, Khan SA, Roberts LR, et al. Cholangiocarcinoma 2020: the next horizon in mechanisms and management. Nat Rev Gastroenterol Hepatol. 2020;17:557–88.
Campisi J, d’Adda di Fagagna F. Cellular senescence: when bad things happen to good cells. Nat Rev Mol Cell Biol. 2007;8:729–40.
d’Adda di Fagagna F. Living on a break: cellular senescence as a DNA-damage response. Nat Rev Cancer. 2008;8:512–22.
Ewald JA, Desotelle JA, Wilding G, Jarrard DF. Therapy-induced senescence in cancer. J Natl Cancer Inst. 2010;102:1536–46.
Hanahan D, Weinberg RA. The hallmarks of cancer. Cell. 2000;100:57–70.
Abrahamson M, Alvarez-Fernandez M, Nathanson CM Cystatins. Biochem Soc Symp. 2003:179–99.
Liu Y, Yao J. Research progress of cystatin SN in cancer. Onco Targets Ther. 2019;12:3411–9.
Kim JT, Lee SJ, Kang MA, Park JE, Kim BY, Yoon DY, et al. Cystatin SN neutralizes the inhibitory effect of cystatin C on cathepsin B activity. Cell Death Dis. 2013;4:e974.
Li D, Wang Y, Dong C, Chen T, Dong A, Ren J, et al. CST1 inhibits ferroptosis and promotes gastric cancer metastasis by regulating GPX4 protein stability via OTUB1. Oncogene. 2023;42:83–98.
Liu Y, Ma H, Wang Y, Du X, Yao J, Cystatin SN. Affects cell proliferation by regulating the ERalpha/PI3K/AKT/ERalpha loopback pathway in breast cancer. Onco Targets Ther. 2019;12:11359–69.
Liu Y, Liao L, An C, Wang X, Li Z, Xu Z, et al. alpha-Enolase lies downstream of mTOR/HIF1alpha and promotes thyroid carcinoma progression by regulating CST1. Front Cell Dev Biol. 2021;9:670019.
Yang J, Luo G, Li C, Zhao Z, Ju S, Li Q, et al. Cystatin SN promotes epithelial-mesenchymal transition and serves as a prognostic biomarker in lung adenocarcinoma. BMC Cancer. 2022;22:589.
Cui Y, Sun D, Song R, Zhang S, Liu X, Wang Y, et al. Upregulation of cystatin SN promotes hepatocellular carcinoma progression and predicts a poor prognosis. J Cell Physiol. 2019;234:22623–34.
Oh SS, Park S, Lee KW, Madhi H, Park SG, Lee HG, et al. Extracellular cystatin SN and cathepsin B prevent cellular senescence by inhibiting abnormal glycogen accumulation. Cell Death Dis. 2017;8:e2729.
Polette M, Nawrocki-Raby B, Gilles C, Clavel C, Birembaut P. Tumour invasion and matrix metalloproteinases. Crit Rev Oncol Hematol. 2004;49:179–86.
Siddiqui A, Ceppi P. A non-proliferative role of pyrimidine metabolism in cancer. Mol Metab. 2020;35:100962.
Gandhi M, Gross M, Holler JM, Coggins SA, Patil N, Leupold JH, et al. The lncRNA lincNMR regulates nucleotide metabolism via a YBX1 - RRM2 axis in cancer. Nat Commun. 2020;11:3214.
Kim J, Hu Z, Cai L, Li K, Choi E, Faubert B, et al. CPS1 maintains pyrimidine pools and DNA synthesis in KRAS/LKB1-mutant lung cancer cells. Nature. 2017;546:168–72.
Rabinovich S, Adler L, Yizhak K, Sarver A, Silberman A, Agron S, et al. Diversion of aspartate in ASS1-deficient tumours fosters de novo pyrimidine synthesis. Nature. 2015;527:379–83.
Han R, Huang S, Bao Y, Liu X, Peng X, Chen Z, et al. Upregulation of SOX4 antagonizes cellular senescence in esophageal squamous cell carcinoma. Oncol Lett. 2016;12:1367–72.
Zhao W, Zhao J, Li K, Hu Y, Yang D, Tan B, et al. Oncogenic role of the NFATC2/NEDD4/FBP1 axis in cholangiocarcinoma. Lab Invest. 2023;103:100193.
Mohamed MM, Sloane BF. Cysteine cathepsins: multifunctional enzymes in cancer. Nat Rev Cancer. 2006;6:764–75.
Bell-McGuinn KM, Garfall AL, Bogyo M, Hanahan D, Joyce JA. Inhibition of cysteine cathepsin protease activity enhances chemotherapy regimens by decreasing tumor growth and invasiveness in a mouse model of multistage cancer. Cancer Res. 2007;67:7378–85.
Premzl A, Zavasnik-Bergant V, Turk V, Kos J. Intracellular and extracellular cathepsin B facilitate invasion of MCF-10A neoT cells through reconstituted extracellular matrix in vitro. Exp Cell Res. 2003;283:206–14.
Sever N, Filipic M, Brzin J, Lah TT. Effect of cysteine proteinase inhibitors on murine B16 melanoma cell invasion in vitro. Biol Chem. 2002;383:839–42.
Huang G, Lu L, You Y, Li J, Zhang K. Knockdown of ENO1 promotes autophagy dependent-ferroptosis and suppresses glycolysis in breast cancer cells via the regulation of CST1. Drug Dev Res. 2024;85:e70004.
Qiang J, Zhao C, Shi LQ, Sun SR, Wang HK, Liu SL, et al. BRD9 promotes the progression of gallbladder cancer via CST1 upregulation and interaction with FOXP1 through the PI3K/AKT pathway and represents a therapeutic target. Gene Ther. 2024;31:594–606.
Zhang L, Chen X, Wang J, Chen M, Chen J, Zhuang W, et al. Cysteine protease inhibitor 1 promotes metastasis by mediating an oxidative phosphorylation/MEK/ERK axis in esophageal squamous carcinoma cancer. Sci Rep. 2024;14:4985.
Jiang J, Liu HL, Tao L, Lin XY, Yang YD, Tan SW, et al. Let‑7d inhibits colorectal cancer cell proliferation through the CST1/p65 pathway. Int J Oncol. 2018;53:781–90.
Thanasai J, Limpaiboon T, Jearanaikoon P, Sripa B, Pairojkul C, Tantimavanich S, et al. Effects of thymidine phosphorylase on tumor aggressiveness and 5-fluorouracil sensitivity in cholangiocarcinoma. World J Gastroenterol. 2010;16:1631–8.
Wu X, Chen D, Li M, Liang G, Ye H. UCK2 promotes intrahepatic cholangiocarcinoma progression and desensitizes cisplatin treatment by PI3K/AKT/mTOR/autophagic axis. Cell Death Discov. 2024;10:375.
Kim J, Bae DH, Kim JH, Song KS, Kim YS, Kim SY. HOXC10 overexpression promotes cell proliferation and migration in gastric cancer. Oncol Rep. 2019;42:202–12.
Luan X, Peng X, Hui G, Wei Z. TFAP2A promotes cell progression and suppresses ferroptosis in lung adenocarcinoma via activating transcription of CST1. J Biochem Mol Toxicol. 2025;39:e70087.
Zhang D, Li H, Jiang X, Cao L, Wen Z, Yang X, et al. Role of AP-2alpha and MAPK7 in the regulation of autocrine TGF-beta/miR-200b signals to maintain epithelial-mesenchymal transition in cholangiocarcinoma. J Hematol Oncol. 2017;10:170.
Sadik H, Korangath P, Nguyen NK, Gyorffy B, Kumar R, Hedayati M, et al. HOXC10 expression supports the development of chemotherapy resistance by fine tuning DNA repair in breast cancer cells. Cancer Res. 2016;76:4443–56.
Li S, Zhang W, Wu C, Gao H, Yu J, Wang X, et al. HOXC10 promotes proliferation and invasion and induces immunosuppressive gene expression in glioma. FEBS J. 2018;285:2278–91.
Acknowledgements
Not applicable.
Funding
This research was supported by the Natural Science Foundation of Shandong Province (grant number ZR2021MH069).
Author information
Authors and Affiliations
Contributions
WZ: Conceptualization, formal analysis, writing-original draft, funding acquisition; JZ: Writing-review & editing, Resources, Validation; KL, JS, LC, and GY: Investigation.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
Clinical experimental protocols were approved by the Ethical and Scientific Committees of Affiliated Hospital of Qingdao University. All animal works were performed following National Research Council: Guide for the Care and Use of Laboratory Animals, and approved by the Animal Experimentation Ethics Committee of Affiliated Hospital of Qingdao University.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zhao, W., Zhao, J., Li, K. et al. NFATC2-mediated CST1 upregulation drives cholangiocarcinoma growth and metastasis. Cell Death Discov. (2026). https://doi.org/10.1038/s41420-026-03036-8
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41420-026-03036-8