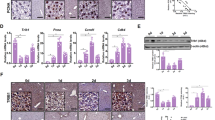
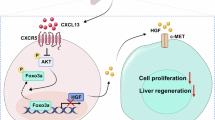
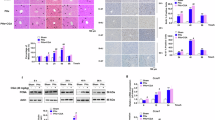
Abstract
Following extensive liver resections, diminished liver regeneration impairs the maintenance or restoration of sufficient functional liver mass. Currently, effective therapies to restore liver regeneration are lacking, rendering liver transplantation the sole treatment option for end-stage liver disease. Therefore, it is imperative to elucidate the regulatory mechanisms underlying liver regeneration. In this study, we employed a multi-omics approach integrating Hi-C, RNA-seq, and ATAC-seq to dissect the early regulatory mechanisms of liver regeneration in rats and mice. Our results indicate that immune and inflammatory processes are markedly enriched during the early phase of regeneration, accompanied by upregulation of glucocorticoids (GCs) and their receptor (GR). First, the expression dynamics of the GC-related circadian gene Nr1d1 and its regulatory network—including Nfκbiα, Arntl, Clock, and Rora—align with chromatin reorganization, leading us to propose that the GC–GR–Nr1d1 axis is involved in maintaining liver homeostasis. Second, the GR-regulated FoxO family is significantly enriched, and the FoxO-associated gene Klf2 exhibits coordinated changes in expression, chromatin accessibility, and chromatin structure. Functional experiments demonstrate that Klf2 negatively regulates hepatocyte proliferation. Hence, we propose the GC–GR–FoxOs–Klf2 axis acts as a checkpoint in hepatocyte proliferation, preventing premature activation of proliferation- and cell cycle-related genes and ensuring orderly and efficient liver regeneration. Our findings on the role of GCs in liver regeneration may further support their future therapeutic application in liver diseases such as liver fibrosis, alcoholic cirrhosis, and hepatocellular carcinoma (HCC).
Similar content being viewed by others
Data availability
Rat all sequencing raw data (Hi-C, RNA-seq and ATAC-seq) generated in this study have been submitted to NCBI with a BioProject accession number of PRJNA1104701 (http://www.ncbi.nlm.nih.gov/bioproject/1104701). Mouse Hi-C raw data have been submitted to NCBI with a BioProject accession number of PRJNA1417570 (http://www.ncbi.nlm.nih.gov/bioproject/1417570). Mouse RNA-seq raw data have been submitted to NCBI with a BioProject accession number of PRJNA1417571 (http://www.ncbi.nlm.nih.gov/bioproject/1417571).
References
Matchett KP, Wilson-Kanamori JR, Portman JR, Kapourani CA, Fercoq F, May S, et al. Multimodal decoding of human liver regeneration. Nature. 2024;630:158–65.
Michalopoulos GK, Bhushan B. Liver regeneration: biological and pathological mechanisms and implications. Nat Rev Gastroenterol Hepatol. 2020;18:40–55.
Arechederra M, Berasain C, Avila MA, Fernandez-Barrena MG. Chromatin dynamics during liver regeneration. Semin Cell Dev Biol. 2020;97:38–46.
Bangru S, Kalsotra A. Cellular and molecular basis of liver regeneration. Semin Cell Dev Biol. 2020;100:74–87.
Mukhopadhyay B, Cinar R, Yin S, Liu J, Tam J, Godlewski G, et al. Hyperactivation of anandamide synthesis and regulation of cell-cycle progression via cannabinoid type 1 (CB1) receptors in the regenerating liver. Proc Natl Acad Sci USA. 2011;108:6323–8.
Polyzos SA, Targher G. Role of glucocorticoids in metabolic dysfunction-associated steatotic liver disease. Curr Obes Rep. 2024;13:242–55.
Ye C, Li W, Li L, Zhang K. Glucocorticoid treatment strategies in liver failure. Front Immunol. 2022;13:846091.
Auger JP, Zimmermann M, Faas M, Stifel U, Chambers D, Krishnacoumar B, et al. Metabolic rewiring promotes anti-inflammatory effects of glucocorticoids. Nature. 2024;629:184–92.
Bazwinsky-Wutschke I, Zipprich A, Dehghani F. Endocannabinoid system in hepatic glucose metabolism, fatty liver disease, and cirrhosis. Int J Mol Sci. 2019;20:2516.
Okabe T, Chavan R, Fonseca Costa SS, Brenna A, Ripperger JA, Albrecht U. REV-ERBalpha influences the stability and nuclear localization of the glucocorticoid receptor. J Cell Sci. 2016;129:4143–54.
Chen L, Xia S, Wang F, Zhou Y, Wang S, Yang T, et al. m(6)A methylation-induced NR1D1 ablation disrupts the HSC circadian clock and promotes hepatic fibrosis. Pharm Res. 2023;189:106704.
Kim YH, Marhon SA, Zhang Y, Steger DJ, Won K-J, Lazar MA. Rev-erbα dynamically modulates chromatin looping to control circadian gene transcription. Science. 2018;359:1274–7.
Inés Pineda Torra VT, Delaunay F, Saladin R, Laudet V, Fruchart J-C, Kosykh V, et al. Circadian and glucocorticoid regulation of rev-erbα expression in liver. Endocrinology. 2000;141:3799–806.
Murayama Y, Yahagi N, Takeuchi Y, Aita Y, Mehrazad Saber Z, Wada N, et al. Glucocorticoid receptor suppresses gene expression of Rev-erbalpha (Nr1d1) through interaction with the CLOCK complex. FEBS Lett. 2019;593:423–32.
Mukhopadhyay B, Holovac K, Schuebel K, Mukhopadhyay P, Cinar R, Iyer S, et al. The endocannabinoid system promotes hepatocyte progenitor cell proliferation and maturation by modulating cellular energetics. Cell Death Discov. 2023;9:104.
Santos BF, Grenho I, Martel PJ, Ferreira BI, Link W. FOXO family isoforms. Cell Death Dis. 2023;14:702.
Guo X, Peng K, He Y, Xue L. Mechanistic regulation of FOXO transcription factors in the nucleus. Biochim Biophys Acta Rev Cancer. 2024;1879:189083.
Nakae J, Oki M, Cao Y. The FoxO transcription factors and metabolic regulation. FEBS Lett. 2008;582:54–67.
Orea-Soufi A, Paik J, Braganca J, Donlon TA, Willcox BJ, Link W. FOXO transcription factors as therapeutic targets in human diseases. Trends Pharm Sci. 2022;43:1070–84.
Krafczyk N, Klotz LO. FOXO transcription factors in antioxidant defense. IUBMB Life. 2022;74:53–61.
Cheng Z. FoxO transcription factors in mitochondrial homeostasis. Biochem J. 2022;479:525–36.
Sanchez AM, Candau RB, Bernardi H. FoxO transcription factors: their roles in the maintenance of skeletal muscle homeostasis. Cell Mol Life Sci. 2014;71:1657–71.
Calissi G, Lam EW, Link W. Therapeutic strategies targeting FOXO transcription factors. Nat Rev Drug Discov. 2021;20:21–38.
Jiramongkol Y, Lam EW. FOXO transcription factor family in cancer and metastasis. Cancer Metastasis Rev. 2020;39:681–709.
Yadav RK, Chauhan AS, Zhuang L, Gan B. FoxO transcription factors in cancer metabolism. Semin Cancer Biol. 2018;50:65–76.
Murtaza G, Khan AK, Rashid R, Muneer S, Hasan SMF, Chen J. FOXO transcriptional factors and long-term living. Oxid Med Cell Longev. 2017;2017:3494289.
Dong XC. FOXO transcription factors in non-alcoholic fatty liver disease. Liver Res. 2017;1:168–73.
Pan X, Zhang Y, Kim HG, Liangpunsakul S, Dong XC. FOXO transcription factors protect against the diet-induced fatty liver disease. Sci Rep. 2017;7:44597.
Salih DA, Brunet A. FoxO transcription factors in the maintenance of cellular homeostasis during aging. Curr Opin Cell Biol. 2008;20:126–36.
Klotz LO, Sanchez-Ramos C, Prieto-Arroyo I, Urbanek P, Steinbrenner H, Monsalve M. Redox regulation of FoxO transcription factors. Redox Biol. 2015;6:51–72.
Kim S, Koh H. Role of FOXO transcription factors in crosstalk between mitochondria and the nucleus. J Bioenerg Biomembr. 2017;49:335–41.
Hornsveld M, Smits LMM, Meerlo M, van Amersfoort M, Groot Koerkamp MJA, van Leenen D, et al. FOXO transcription factors both suppress and support breast cancer progression. Cancer Res. 2018;78:2356–69.
Mukhopadhyay B, Schuebel K, Mukhopadhyay P, Cinar R, Godlewski G, Xiong K, et al. Cannabinoid receptor 1 promotes hepatocellular carcinoma initiation and progression through multiple mechanisms. Hepatology. 2015;61:1615–26.
Liang CQ, Zhou DC, Peng WT, Chen WY, Wu HY, Zhou YM, et al. FoxO3 restricts liver regeneration by suppressing the proliferation of hepatocytes. NPJ Regen Med. 2022;7:33.
Kuo T, Liu PH, Chen TC, Lee RA, New J, Zhang D, et al. Transcriptional regulation of FoxO3 gene by glucocorticoids in murine myotubes. Am J Physiol Endocrinol Metab. 2016;310:E572–85.
Qin W, Pan J, Qin Y, Lee DN, Bauman WA, Cardozo C. Identification of functional glucocorticoid response elements in the mouse FoxO1 promoter. Biochem Biophys Res Commun. 2014;450:979–83.
Riascos-Bernal DF, Sibinga NE. Neutrophil extracellular traps in cardiac hypertrophy: a KLF2 perspective. J Clin Invest. 2022;132:e147191.
Shao D, Villet O, Zhang Z, Choi SW, Yan J, Ritterhoff J, et al. Glucose promotes cell growth by suppressing branched-chain amino acid degradation. Nat Commun. 2018;9:2935.
Maity J, Deb M, Greene C, Das H. KLF2 regulates dental pulp-derived stem cell differentiation through the induction of mitophagy and altering mitochondrial metabolism. Redox Biol. 2020;36:101622.
Serr I, Furst RW, Ott VB, Scherm MG, Nikolaev A, Gokmen F, et al. miRNA92a targets KLF2 and the phosphatase PTEN signaling to promote human T follicular helper precursors in T1D islet autoimmunity. Proc Natl Acad Sci USA. 2016;113:E6659–68.
Salter AI, Ivey RG, Kennedy JJ, Voillet V, Rajan A, Alderman EJ, et al. Phosphoproteomic analysis of chimeric antigen receptor signaling reveals kinetic and quantitative differences that affect cell function. Sci Signal. 2018;11:eaat6753.
Sangwung P, Zhou G, Nayak L, Chan ER, Kumar S, Kang DW, et al. KLF2 and KLF4 control endothelial identity and vascular integrity. JCI Insight. 2017;2:e91700.
Guixe-Muntet S, de Mesquita FC, Vila S, Hernandez-Gea V, Peralta C, Garcia-Pagan JC, et al. Cross-talk between autophagy and KLF2 determines endothelial cell phenotype and microvascular function in acute liver injury. J Hepatol. 2017;66:86–94.
Li Y, Tu S, Zeng Y, Zhang C, Deng T, Luo W, et al. KLF2 inhibits TGF-beta-mediated cancer cell motility in hepatocellular carcinoma. Acta Biochim Biophys Sin (Shanghai). 2020;52:485–94.
Manavski Y, Abel T, Hu J, Kleinlutzum D, Buchholz CJ, Belz C, et al. Endothelial transcription factor KLF2 negatively regulates liver regeneration via induction of activin A. Proc Natl Acad Sci USA. 2017;114:3993–8.
Caldez MJ, Van Hul N, Koh HWL, Teo XQ, Fan JJ, Tan PY, et al. Metabolic remodeling during liver regeneration. Dev Cell. 2018;47:425–38.
Liu R, Scimeca M, Sun Q, Melino G, Mauriello A, Shao C, et al. Harnessing metabolism of hepatic macrophages to aid liver regeneration. Cell Death Dis. 2023;14:574.
Wang X, Menezes CJ, Jia Y, Xiao Y, Venigalla SSK, Cai F, et al. Metabolic inflexibility promotes mitochondrial health during liver regeneration. Science. 2024;384:eadj4301.
Pervouchine D, Popov Y, Berry A, Borsari B, Frankish A, Guigo R. Integrative transcriptomic analysis suggests new autoregulatory splicing events coupled with nonsense-mediated mRNA decay. Nucleic Acids Res. 2019;47:5293–306.
Dai Z, Dai X. Nuclear colocalization of transcription factor target genes strengthens coregulation in yeast. Nucleic Acids Res. 2012;40:27–36.
Mukhopadhyay B, Marietta C, Shen PH, Oiseni A, Mirshahi F, Mazzu M, et al. A patient-based iPSC-derived hepatocyte model of alcohol-associated cirrhosis reveals bioenergetic insights into disease pathogenesis. Nat Commun. 2024;15:2869.
Kar A, Ghosh P, Gautam A, Chowdhury S, Basak D, Sarkar I, et al. CD38-RyR2 axis-mediated signaling impedes CD8(+) T cell response to anti-PD1 therapy in cancer. Proc Natl Acad Sci USA. 2024;121:e2315989121.
Liu R, Cui J, Sun Y, Xu W, Wang Z, Wu M, et al. Autophagy deficiency promotes M1 macrophage polarization to exacerbate acute liver injury via ATG5 repression during aging. Cell Death Discov. 2021;7:397.
Zheng G, Pang S, Wang J, Wang F, Wang Q, Yang L, et al. Glucocorticoid receptor-mediated Nr1d1 chromatin circadian misalignment in stress-induced irritable bowel syndrome. iScience. 2023;26:107137.
Papa S, Zazzeroni F, Fu YX, Bubici C, Alvarez K, Dean K, et al. Gadd45beta promotes hepatocyte survival during liver regeneration in mice by modulating JNK signaling. J Clin Invest. 2008;118:1911–23.
Hu Z, Han Y, Liu Y, Zhao Z, Ma F, Cui A, et al. CREBZF as a key regulator of STAT3 pathway in the control of liver regeneration in mice. Hepatology. 2020;71:1421–36.
Lopez-Luque J, Caballero-Diaz D, Martinez-Palacian A, Roncero C, Moreno-Caceres J, Garcia-Bravo M, et al. Dissecting the role of epidermal growth factor receptor catalytic activity during liver regeneration and hepatocarcinogenesis. Hepatology. 2016;63:604–19.
Chen T, Oh S, Gregory S, Shen X, Diehl AM. Single-cell omics analysis reveals functional diversification of hepatocytes during liver regeneration. JCI Insight. 2020;5:e141024.
Ye B, Shen W, Zhang C, Yu M, Ding X, Yin M, et al. The role of ZNF143 overexpression in rat liver cell proliferation. BMC Genom. 2022;23:483.
Grandi FC, Modi H, Kampman L, Corces MR. Chromatin accessibility profiling by ATAC-seq. Nat Protoc. 2022;17:1518–52.
Dixon JR, Selvaraj S, Yue F, Kim A, Li Y, Shen Y, et al. Topological domains in mammalian genomes identified by analysis of chromatin interactions. Nature. 2012;485:376–80.
Funding
This work was supported by the National Science and Technology Major Project (2018ZX10305410) to ZHZ, Henan Natural Science Foundation (242300421330) to BYY and the Key Scientific Research Projects of Henan Higher Education (24A180014) to B.Y.Y.
Author information
Authors and Affiliations
Contributions
ZHZ conceived and designed the project. BYY, MJY, XJG, QPJ, PL, and YZ carried out the experiments; DJX and WLS carried out the data analysis and interpreted all bioinformatic data. BYY and ZHZ contributed to results interpretation and wrote the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
All animal experiments were conducted in accordance with procedures approved by the ethics committee at Henan Normal University, Henan, China, and conformed to the relevant regulatory standards. All animal studies were completed in the experimental animal center of Henan Normal University, China (license number: HNSD-2025-07-07, licensed by the Ministry of Science and Technology of China).
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ye, B., Xie, D., Shen, W. et al. Dual control of liver regeneration by Nr1d1 homeostasis and Klf2 checkpoint. Cell Death Discov. (2026). https://doi.org/10.1038/s41420-026-03039-5
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41420-026-03039-5