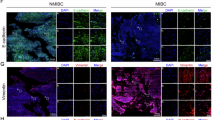
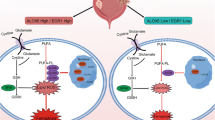
Abstract
Ferroptosis, a form of regulated cell death, plays a pivotal role in the development and treatment of cancer because of its impact on tumor cell proliferation, differentiation, and resistance to chemotherapy. NT5DC2, a gene associated with ferroptosis, has been identified as a key facilitator of cellular proliferation and metastasis in several cancers. In this study, we found that NT5DC2 is highly expressed in bladder cancer tissues compared with normal tissues and that its expression is correlated with the poor prognosis of bladder cancer patients. Functionally, we demonstrated that NT5DC2 suppresses ferroptosis in bladder cancer cells and promotes malignant tumor progression. Mechanistically, NT5DC2 interacts with ACSL3 and hampers its ubiquitination, thereby improving the stability of ACSL3, a crucial ferroptosis suppressing protein induced by oleic acid in lymph nodes. In addition, rescue assay results indicated that ACSL3 mediated the roles of NT5DC2 in suppressing ferroptosis of bladder cancer cells. Furthermore, we found that the upregulation of ACSL3 by oleic acid treatment was mediated by NT5DC2 as manifested by the observation that silencing of NT5DC2 abrogates this regulatory effect of oleic acid treatment. Collectively, our findings suggest that NT5DC2/ACSL3 plays a critical role in bladder cancer progression and ferroptosis regulation, suggesting that NT5DC2/ACSL3 is a potential therapeutic target for bladder cancer treatment.
Similar content being viewed by others
Data availability
All the data produced or analyzed in this study are available within the published article and its supplementary information files.
References
Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, et al. Global Cancer Statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. Cancer J Clin. 2021;71:209–49.
Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68:394–424.
Haas M, Knobloch C, Mayr R, Gierth M, Pickl C, Engelmann S, et al. Short-term and long-term morbidity after radical cystectomy in patients with NMIBC and comparison with MIBC: identifying risk factors for severe short-term complications. Urol Int. 2023;107:246–56.
Lenis AT, Lec PM, Chamie K, Mshs MD. Bladder cancer: a review. Jama. 2020;324:1980–91.
Lopez-Beltran A, Cookson MS, Guercio BJ, Cheng L. Advances in diagnosis and treatment of bladder cancer. Bmj. 2024;384:e076743.
van Hoogstraten LMC, Vrieling A, van der Heijden AG, Kogevinas M, Richters A, Kiemeney LA. Global trends in the epidemiology of bladder cancer: challenges for public health and clinical practice. Nat Rev Clin Oncol. 2023;20:287–304.
Mou Y, Wang J, Wu J, He D, Zhang C, Duan C, et al. Ferroptosis, a new form of cell death: opportunities and challenges in cancer. J Hematol Oncol. 2019;12:34.
Dixon SJ, Pratt DA. Ferroptosis: a flexible constellation of related biochemical mechanisms. Mol Cell. 2023;83:1030–42.
Peng Z, Ding YN, Yang ZM, Li XJ, Zhuang Z, Lu Y, et al. Corrigendum to “Neuron-targeted liposomal coenzyme Q10 attenuates neuronal ferroptosis after subarachnoid hemorrhage by activating the ferroptosis suppressor protein 1/coenzyme Q10 system” Acta Biomaterialia 2024, 179:325-339. Acta Biomater. 2024;182:301–2.
Li D, Wang Y, Dong C, Chen T, Dong A, Ren J, et al. CST1 inhibits ferroptosis and promotes gastric cancer metastasis by regulating GPX4 protein stability via OTUB1. Oncogene. 2023;42:83–98.
Li H, Yu K, Hu H, Zhang X, Zeng S, Li J, et al. METTL17 coordinates ferroptosis and tumorigenesis by regulating mitochondrial translation in colorectal cancer. Redox Biol. 2024;71:103087.
Yuan S, Xi S, Weng H, Guo MM, Zhang JH, Yu ZP, et al. YTHDC1 as a tumor progression suppressor through modulating FSP1-dependent ferroptosis suppression in lung cancer. Cell Death Differ. 2023;30:2477–90.
Nakashima A, Yamaguchi H, Kodani Y, Kaneko YS, Kawata M, Nagasaki H, et al. Identification by nano-LC-MS/MS of NT5DC2 as a protein binding to tyrosine hydroxylase: Down-regulation of NT5DC2 by siRNA increases catecholamine synthesis in PC12D cells. Biochem Biophys Res Commun. 2019;516:1060–5.
Singgih EL, van der Voet M, Schimmel-Naber M, Brinkmann EL, Schenck A, Franke B. Investigating cytosolic 5’-nucleotidase II family genes as candidates for neuropsychiatric disorders in Drosophila (114/150 chr). Transl Psychiatry. 2021;11:55.
Jordheim LP. Expanding the clinical relevance of the 5’-nucleotidase cN-II/NT5C2. Purinergic Signal. 2018;14:321–9.
Huang Z, Xu E, Ma X, Wang Y, Zhu J, Zhu K, et al. Low NT5DC2 expression predicts favorable prognosis and suppresses soft tissue sarcoma progression via ECM-receptor interaction pathway. Transl Oncol. 2024;44:101937.
Sang R, Yu X, Xia H, Qian X, Yong J, Xu Y, et al. NT5DC2 knockdown suppresses progression, glycolysis, and neuropathic pain in triple-negative breast cancer by blocking the EGFR pathway. Mol Carcinog. 2024;63:785–96.
Jin X, Liu X, Zhang Z, Xu L. NT5DC2 suppression restrains progression towards metastasis of non-small-cell lung cancer through regulation p53 signaling. Biochem Biophys Res Commun. 2020;533:354–61.
Padanad MS, Konstantinidou G, Venkateswaran N, Melegari M, Rindhe S, Mitsche M, et al. Fatty acid oxidation mediated by Acyl-CoA synthetase long chain 3 is required for mutant KRAS lung tumorigenesis. Cell Rep. 2016;16:1614–28.
Yang Y, Zhu T, Wang X, Xiong F, Hu Z, Qiao X, et al. ACSL3 and ACSL4, distinct roles in ferroptosis and cancers. Cancers. 2022;14:5896.
Magtanong L, Ko PJ, To M, Cao JY, Forcina GC, Tarangelo A, et al. Exogenous monounsaturated fatty acids promote a ferroptosis-resistant cell state. Cell Chem Biol. 2019;26:420–32.e9.
Ubellacker JM, Tasdogan A, Ramesh V, Shen B, Mitchell EC, Martin-Sandoval MS, et al. Lymph protects metastasizing melanoma cells from ferroptosis. Nature. 2020;585:113–8.
Xie Y, Wang B, Zhao Y, Tao Z, Wang Y, Chen G, et al. Mammary adipocytes protect triple-negative breast cancer cells from ferroptosis. J Hematol Oncol. 2022;15:72.
Han S, Guo J, Kong C, Li J, Lin F, Zhu J, et al. ANKRD1 aggravates renal ischaemia‒reperfusion injury via promoting TRIM25-mediated ubiquitination of ACSL3. Clin Transl Med. 2024;14:e70024.
Liu H, Du J, Chao S, Li S, Cai H, Zhang H, et al. Fusobacterium nucleatum promotes colorectal cancer cell to acquire stem cell-like features by manipulating lipid droplet-mediated numb degradation. Adv Sci (Weinh). 2022;9:e2105222.
Tong X, Tang R, Xiao M, Xu J, Wang W, Zhang B, et al. Targeting cell death pathways for cancer therapy: recent developments in necroptosis, pyroptosis, ferroptosis, and cuproptosis research. J Hematol Oncol. 2022;15:174.
Li H, Sun Y, Yao Y, Ke S, Zhang N, Xiong W, et al. USP8-governed GPX4 homeostasis orchestrates ferroptosis and cancer immunotherapy. Proc Natl Acad Sci USA. 2024;121:e2315541121.
Liu J, Lu X, Zeng S, Fu R, Wang X, Luo L, et al. ATF3-CBS signaling axis coordinates ferroptosis and tumorigenesis in colorectal cancer. Redox Biol. 2024;71:103118.
Zhou Q, Liu T, Qian W, Ji J, Cai Q, Jin Y, et al. HNF4A-BAP31-VDAC1 axis synchronously regulates cell proliferation and ferroptosis in gastric cancer. Cell Death Dis. 2023;14:356.
Li KS, Zhu XD, Liu HD, Zhang SZ, Li XL, Xiao N, et al. NT5DC2 promotes tumor cell proliferation by stabilizing EGFR in hepatocellular carcinoma. Cell Death Dis. 2020;11:335.
Guo S, Ran H, Xiao D, Huang H, Mi L, Wang X, et al. NT5DC2 promotes tumorigenicity of glioma stem-like cells by upregulating fyn. Cancer Lett. 2019;454:98–107.
Cui Y, Wen Y, Lv C, Zhao D, Yang Y, Qiu H, et al. Decreased RNA‑binding protein IGF2BP2 downregulates NT5DC2, which suppresses cell proliferation, and induces cell cycle arrest and apoptosis in diffuse large B‑cell lymphoma cells by regulating the p53 signaling pathway. Mol Med Rep. 2022;26:286.
Jiang X, Stockwell BR, Conrad M. Ferroptosis: mechanisms, biology and role in disease. Nat Rev Mol Cell Biol. 2021;22:266–82.
Chen YC, Oses-Prieto JA, Pope LE, Burlingame AL, Dixon SJ, Renslo AR. Reactivity-based probe of the iron(II)-dependent interactome identifies new cellular modulators of ferroptosis. J Am Chem Soc. 2020;142:19085–93.
Tang Z, Li C, Kang B, Gao G, Li C, Zhang Z. GEPIA: a web server for cancer and normal gene expression profiling and interactive analyses. Nucleic Acids Res. 2017;45:W98–w102.
Ranstam J, Cook JA. Kaplan-Meier curve. Br J Surg. 2017;104:442.
Wen Y, Chen H, Zhang L, Wu M, Zhang F, Yang D, et al. Glycyrrhetinic acid induces oxidative/nitrative stress and drives ferroptosis through activating NADPH oxidases and iNOS, and depriving glutathione in triple-negative breast cancer cells. Free Radic Biol Med. 2021;173:41–51.
Zhang F, Tang X, Fan S, Liu X, Sun J, Ju C, et al. Targeting the p300/NONO axis sensitizes melanoma cells to BRAF inhibitors. Oncogene. 2021;40:4137–50.
Acknowledgements
We appreciate our lab members and collaborators for their thorough review and valuable contributions to enhancing the manuscript.
Funding
This work was supported by the National Natural Science Foundation of China (grant numbers 82360472 to XBL); The Double Thousand Talents Project of Jiangxi Province (grant numbers jxsq2023201077 to XBL); the Jiangxi Provincial Natural Science Foundation of China (grant numbers 20232BAB206092 and 20212ACB206028 to XBL); Nanchang science and Technology Bureau (grant numbers 2022-KJZC-008 and grant numbers 2021-156 to XBL)
Author information
Authors and Affiliations
Contributions
SN: Conceptualization, data curation, formal analysis, methodology, visualization, writing—review and editing; PY: Conceptualization and data curation; YY: Data curation; XT: Investigation; JY: Investigation; FZ: Investigation; KC: Methodology; CJ: Project administration; YZ: Project administration and validation; WB: Validation; LL: Validation; XBL: Conceptualization, funding acquisition, methodology, project administration, resources, supervision, validation, visualization, writing—review and editing. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval and consent to participate
This research protocol was approved by the Ethics Committee of the Third Affiliated Hospital of Nanchang University (No. IIT2025003). All of the animal experiments were reviewed and approved by the Animal Experiment Ethics Committee of Nanchang University (No. NCULAE-20241225004), and the experimental procedures were conducted in accordance with the guidelines.
Consent for publication
All authors have agreed to publish this manuscript.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Niu, S., Yang, P., Yao, Y. et al. NT5DC2 inhibits ferroptosis by stabilizing ACSL3 in bladder cancer. Cell Death Discov. (2026). https://doi.org/10.1038/s41420-026-03091-1
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41420-026-03091-1