Abstract
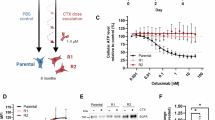
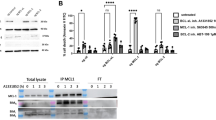
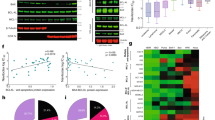
Pre-B-cell leukemia homeobox 1 (PBX1) is a transcription factor involved in diverse cellular processes, but its role in colorectal cancer (CRC) remains incompletely understood. In this study, we show that PBX1 is downregulated in CRC tissues and cell lines. Functional experiments revealed that PBX1 overexpression inhibits proliferation, migration, and invasion, but paradoxically suppresses apoptosis, suggesting a dual regulatory role. Transcriptome and CUT&Tag profiling identified BCL2L1 as a direct transcriptional target of PBX1. PBX1 binds the BCL2L1 promoter and enhances Bcl-xL expression, contributing to apoptotic resistance. BCL2L1 knockdown reversed the anti-apoptotic effects of PBX1 and restored apoptosis levels. Upon 5-fluorouracil (5-FU) treatment, PBX1 overexpression reduced cell viability, while concurrent BCL2L1 knockdown significantly enhanced drug sensitivity. In vivo, xenograft experiments demonstrated that PBX1 overexpression suppressed tumor growth, which was further augmented by BCL2L1 knockdown. These results support the dual role of PBX1 in simultaneously inhibiting tumor growth while promoting cell survival through the BCL2L1–Bcl-xL axis. This regulatory interaction may influence tumor persistence and therapeutic response in CRC.
Similar content being viewed by others
Funding
This research was supported by the Natural Science Foundation of Guangdong Province (Grant No. 2023A1515010434) and the National Natural Science Foundation of China (NSFC, Grant No. 32000456).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
This study was approved by the Ethics Committee of Shantou University Medical College, China (Approval No. SUMC-2020-14). Written informed consent was obtained from all patients for tissue collection and analysis. All methods involving human participants were performed in accordance with the relevant guidelines and regulations. All animal procedures were performed in accordance with institutional guidelines and approved by the Institutional Animal Care and Use Committee of Shantou University Medical College (Approval No. SUMC-2022-289).
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Lin, H., Su, T., Liu, Y. et al. Targeting the PBX1–BCL2L1 axis as a therapeutic strategy in colorectal cancer. Cell Death Discov. (2026). https://doi.org/10.1038/s41420-026-03139-2
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41420-026-03139-2