Abstract
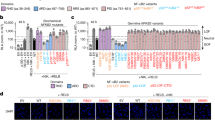
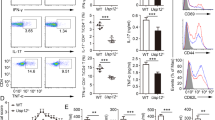
NF-κB, a family of transcription factors regulating diverse biological processes including immune responses, is activated by canonical and noncanonical pathways based on degradation of IκBα and processing of the IκB-like protein p100, respectively. Although p100 responds to noncanonical NF-κB stimuli for processing, it does not undergo degradation, but rather becomes accumulated, along with canonical NF-κB activation. We show here that the stability of p100 is tightly controlled by a deubiquitinase, Otub1. Otub1 deficiency not only promotes signal-induced p100 processing and noncanonical NF-κB activation but also causes steady-state p100 degradation, leading to aberrant NF-κB activation in the canonical pathway. B-cell-conditional deletion of Otub1 results in B-cell hyperplasia, antibody hyper-production, and lupus-like autoimmunity. Otub1-deficient B cells display aberrantly activated phenotypes and overproduce the cytokine IL-6, contributing to autoimmunity induction. Thus, maintenance of p100 stability by Otub1 serves as an unusual mechanism of NF-κB regulation that prevents autoimmunity.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Hayden, M. S. & Ghosh, S. Shared principles in NF-kappaB signaling. Cell 132, 344–362 (2008).
Vallabhapurapu, S. & Karin, M. Regulation and function of NF-kappaB transcription factors in the immune system. Annu. Rev. Immunol. 27, 693–733 (2009).
Sun, S. C. The non-canonical NF-kappaB pathway in immunity and inflammation. Nat. Rev. Immunol. 17, 545–558 (2017).
Hayden, M. S. & Ghosh, S. NF-kappaB, the first quarter-century: remarkable progress and outstanding questions. Genes Dev. 26, 203–234 (2012).
Senftleben, U. et al. Activation of IKKa of a second, evolutionary conserved, NF-kB signaling pathway. Science 293, 1495–1499 (2001).
Sun, S. C. Non-canonical NF-κB signaling pathway. Cell Res. 21, 71–85 (2011).
Xiao, G., Harhaj, E. W. & Sun, S. C. NF-kappaB-inducing kinase regulates the processing of NF-kappaB2 p100. Mol. Cell 7, 401–409 (2001).
Sun, S.-C., Ganchi, P. A., Beraud, C., Ballard, D. W. & Greene, W. C. Autoregulation of the NF-kB transactivator Rel A (p65) by multiple cytoplasmic inhibitors containing ankyrin motifs. Proc. Natl Acad. Sci. USA 91, 1346–1350 (1994).
Basak, S. et al. A fourth IkappaB protein within the NF-kappaB signaling module. Cell 128, 369–381 (2007).
Legarda-Addison, D. & Ting, A. T. Negative regulation of TCR signaling by NF-{kappa}B2/p100. J. Immunol. 178, 7767–7778 (2007).
Liptay, S., Schmid, R. M., Nabel, E. G. & Nabel, G. J. Transcriptional regulation of NF-kappa B2: evidence for kappa B-mediated positive and negative autoregulation. Mol. Cell. Biol. 14, 7693–7703 (1994).
Roux, K. J., Kim, D. I., Raida, M. & Burke, B. A promiscuous biotin ligase fusion protein identifies proximal and interacting proteins in mammalian cells. J. Cell. Biol. 196, 801–810 (2012).
Liao, G., Zhang, M., Harhaj, E. W. & Sun, S. C. Regulation of the NF-kappaB-inducing kinase by tumor necrosis factor receptor-associated factor 3-induced degradation. J. Biol. Chem. 279, 26243–26250 (2004).
Morrison, M. D., Reiley, W., Zhang, M. & SC., S. An atypical tumor necrosis factor (TNF) receptor-associated factor-binding motif of B cell-activating factor belonging to the TNF family (BAFF) receptor mediates induction of the noncanonical NF-kappaB signaling pathway. J. Biol. Chem. 280, 10018–10024 (2005).
Balakirev, M. Y., Tcherniuk, S. O., Jaquinod, M. & Chroboczek, J. Otubains: a new family of cysteine proteases in the ubiquitin pathway. EMBO Rep. 4, 517–522 (2003).
Fong, A. & Sun, S.-C. Genetic evidence for the essential role of beta-transducin repeat-containing protein in the inducible processing of NF-kB2/p100. J. Biol. Chem. 277, 22111–22114 (2002).
Nakada, S. et al. Non-canonical inhibition of DNA damage-dependent ubiquitination by OTUB1. Nature 466, 941–946 (2010).
Wiener, R., Zhang, X., Wang, T. & Wolberger, C. The mechanism of OTUB1-mediated inhibition of ubiquitination. Nature 483, 618–622 (2012).
Arabi, A. et al. Proteomic screen reveals Fbw7 as a modulator of the NF-kappaB pathway. Nat. Commun. 3, 976 (2012).
Busino, L. et al. Fbxw7alpha- and GSK3-mediated degradation of p100 is a pro-survival mechanism in multiple myeloma. Nat. Cell Biol. 14, 375–385 (2012).
Fukushima, H. et al. SCF(Fbw7) modulates the NFkB signaling pathway by targeting NFkB2 for ubiquitination and destruction. Cell Rep. 1, 434–443 (2012).
Kaileh, M. & Sen, R. NF-kappaB function in B lymphocytes. Immunol. Rev. 246, 254–271 (2012).
Hardy, R. R. & Hayakawa, K. Perspectives on fetal derived CD5+ B1 B cells. Eur. J. Immunol. 45, 2978–2984 (2015).
Shinkura, R. et al. Alymphoplasia is caused by a point mutation in the mouse gene encoding Nf-kappa b-inducing kinase. Nat. Genet. 22, 74–77 (1999).
Dejardin, E. The alternative NF-kappaB pathway from biochemistry to biology: pitfalls and promises for future drug development. Biochem. Pharmacol. 72, 1161–1179 (2006).
Brightbill, H. D. et al. Conditional deletion of NF-kappaB-inducing kinase (NIK) in adult mice disrupts mature B cell survival and activation. J. Immunol. 195, 953–964 (2015).
Hahn, M., Macht, A., Waisman, A. & Hovelmeyer, N. NF-kappaB-inducing kinase is essential for B-cell maintenance in mice. Eur. J. Immunol. 46, 732–741 (2016).
Tucker, E. et al. A novel mutation in the Nfkb2 gene generates an NF-kappa B2 “super repressor”. J. Immunol. 179, 7514–7522 (2007).
Liossis, S. C. & Staveri, C. B cell-based treatments in SLE: past experience and current directions. Curr. Rheumatol. Rep. 19, 78 (2017).
Kaul, A. et al. Systemic lupus erythematosus. Nat. Rev. Dis. Primers 2, 16039 (2016).
Eisenberg, R. The chronic graft-versus-host model of systemic autoimmunity. Curr. Dir. Autoimmun. 6, 228–244 (2003).
Bonnefoy, J. Y., Lecoanet-Henchoz, S., Aubry, J. P., Gauchat, J. F. & Graber, P. CD23 and B-cell activation. Curr. Opin. Immunol. 7, 355–359 (1995).
Rickert, R. C. Regulation of B lymphocyte activation by complement C3 and the B cell coreceptor complex. Curr. Opin. Immunol. 17, 237–243 (2005).
Chang, M., Jin, W. & Sun, S. C. Peli1 facilitates TRIF-dependent Toll-like receptor signaling and proinflammatory cytokine production. Nat. Immunol. 10, 1089–1095 (2009).
Barr, T. A. et al. B cell depletion therapy ameliorates autoimmune disease through ablation of IL-6-producing B cells. J. Exp. Med. 209, 1001–1010 (2012).
Arkatkar, T. et al. B cell-derived IL-6 initiates spontaneous germinal center formation during systemic autoimmunity. J. Exp. Med. 214, 3207–3217 (2017).
Li, S. et al. Regulation of virus-triggered signaling by OTUB1- and OTUB2-mediated deubiquitination of TRAF3 and TRAF6. J. Biol. Chem. 285, 4291–4297 (2010).
Goncharov, T. et al. OTUB1 modulates c-IAP1 stability to regulate signalling pathways. EMBO J. 32, 1103–1114 (2013).
Liao, G. & Sun, S. C. Regulation of NF-kappaB2/p100 processing by its nuclear shuttling. Oncogene 22, 4868–4874 (2003).
Rivera-Walsh, I., Cvijic, M. E., Xiao, G. & Sun, S. C. The NF-kappa B signaling pathway is not required for Fas ligand gene induction but mediates protection from activation-induced cell death. J. Biol. Chem. 275, 25222–25230 (2000).
Roux, K. J., Kim, D. I. & Burke, B. BioID: a screen for protein–protein interactions. Curr. Protoc. Protein Sci. 74, Unit 19.23 (2013).
Jin, J. et al. Epigenetic regulation of the expression of Il12 and Il23 and autoimmune inflammation by the deubiquitinase Trabid. Nat. Immunol. 17, 259–268 (2016).
Klarquist, J. & Janssen, E. M. The bm12 inducible model of systemic lupus erythematosus (SLE) in C57BL/6 mice. J. Vis. Exp. 105, e53319 (2015).
Li, Y. et al. Cell intrinsic role of NF-kappaB-inducing kinase in regulating T cell-mediated immune and autoimmune responses. Sci. Rep. 6, 22115 (2016).
Acknowledgements
We thank Genentech Inc. for NIK-flox mice, Walter and Eliza Hall Institute of Medical Research for Nfkb2lym1 mice, and S. Nakada for Otub1 expression vectors. We thank the personnel from the MS Proteomics facility at Baylor College of Medicine for mass spectrometry analysis of the BioID samples. We also thank the personnel from the flow cytometry, DNA analysis, genetically engineered mouse facility, and animal facilities at The MD Anderson Cancer Center for technical assistance. This study was supported by grants from the National Institutes of Health (GM84459, AI64639, and AI057555).
Author information
Authors and Affiliations
Contributions
Y.L. designed and performed the research, prepared the figures, and wrote part of the manuscript; J.-Y.Y., X.X., Z.J., L.Z., J.S., D.L., M.G., X.Z., H.S.L. and X.C. contributed experiments; A.J. performed MS, which was analyzed by S.Y.J. and J.Q.; S.S.W. was involved in the supervision of H.S.L.; and S.-C.S. supervised the work and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Supplementary information
Rights and permissions
About this article
Cite this article
Li, Y., Yang, JY., Xie, X. et al. Preventing abnormal NF-κB activation and autoimmunity by Otub1-mediated p100 stabilization. Cell Res 29, 474–485 (2019). https://doi.org/10.1038/s41422-019-0174-3
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41422-019-0174-3
This article is cited by
-
OTUB1 antagonizes TRIM21 to induce deubiquitination of SPHK1 and promote the progression of hepatocellular carcinoma
Oncogene (2025)
-
NF-κB in inflammation and cancer
Cellular & Molecular Immunology (2025)
-
Deubiquitinase-dependent transcriptional silencing controls inflammation
Cell Research (2025)
-
OTUB1 regulation of ferroptosis and the protective role of ferrostatin-1 in lupus nephritis
Cell Death & Disease (2024)
-
Complex IIa formation and ABC transporters determine sensitivity of OSCC to Smac mimetics
Cell Death & Disease (2024)